Class 7 : Science – ( English ) : Lesson 5. Changes Around Us: Physical and Chemical
EXPLANATION AND ANALYSIS
🧭 Changes are happening around us all the time.
When ice melts ❄️➡️💧, when food cooks 🍳, when leaves fall 🍂, or when iron rusts 🧲➡️🟤, we observe changes in matter. These changes help us understand how materials behave in different situations.
🧠 Observing changes helps us connect science with daily life.
Some changes are temporary 🔄.
Some changes are permanent ⛔.
Science studies these changes to explain nature clearly.
🔍 Changes can be broadly classified into physical changes and chemical changes.
🧪 A physical change is a change in which only the shape, size, or state of a substance changes.
The substance remains the same.
❄️ Ice melting into water is a physical change.
💧 Water freezing back into ice is also a physical change.
🧠 No new substance is formed in a physical change.
These changes are usually reversible 🔁.
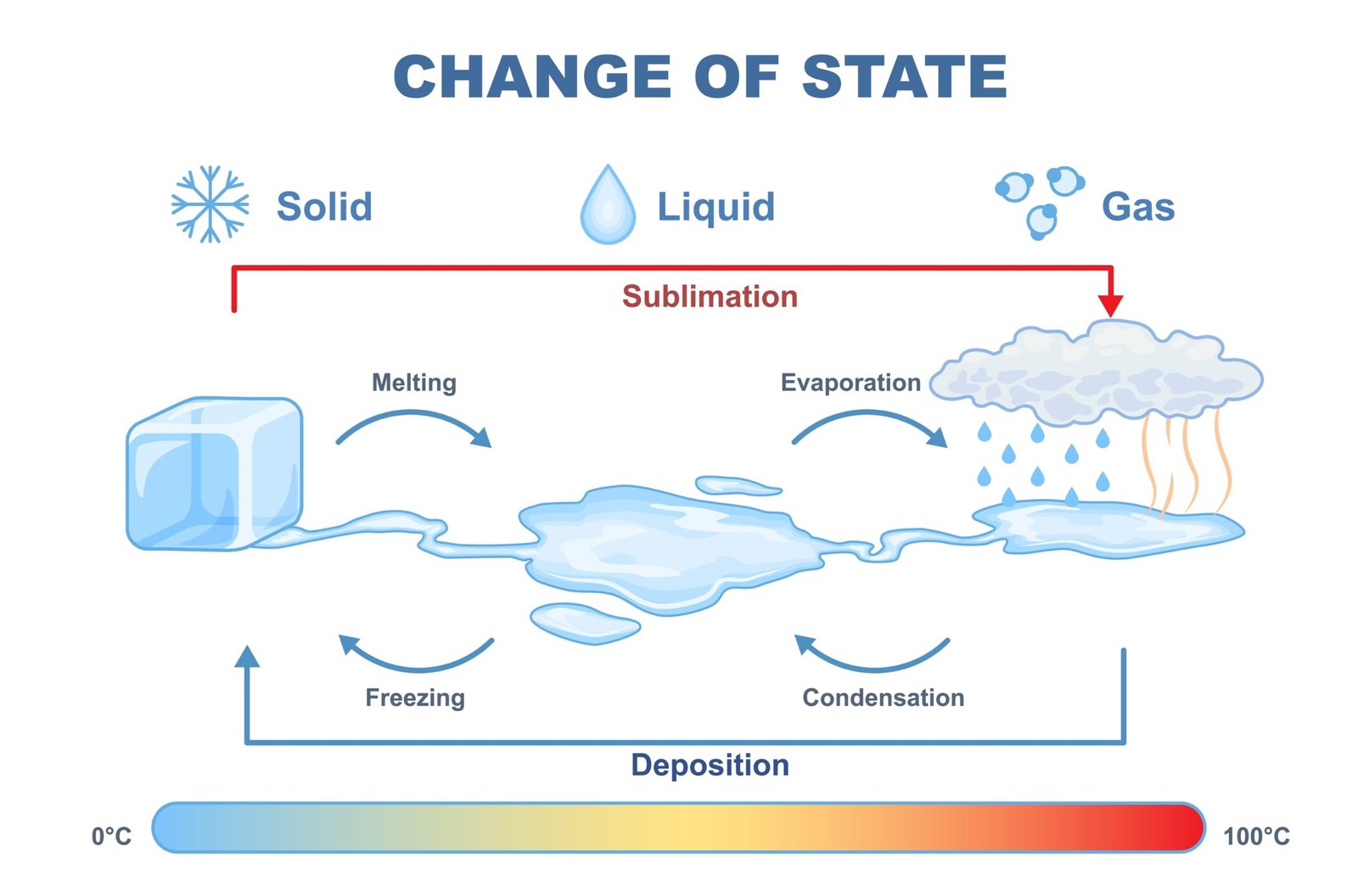
🧭 Changes in state are common physical changes.
Solid ➡️ Liquid ➡️ Gas
🔥 Heating causes melting and evaporation.
❄️ Cooling causes freezing and condensation.
🧠 These changes depend on temperature and pressure.
📦 Change in shape or size is another physical change.
Tearing paper 📄
Breaking chalk 🪨
Stretching rubber band 🧵
🧠 Even after the change, the material remains the same.

🧪 A chemical change is a change in which a new substance is formed.
The original substance cannot be obtained back easily.
🔥 Burning paper produces ash 🟤 and smoke 🌫️.
🧲 Rusting of iron forms rust.
🧠 Chemical changes are usually irreversible ⛔.

🧭 During chemical changes, properties of substances change completely.
Color changes 🎨
Gas formation 💨
Heat or light release ✨
🧠 These signs help identify chemical changes.
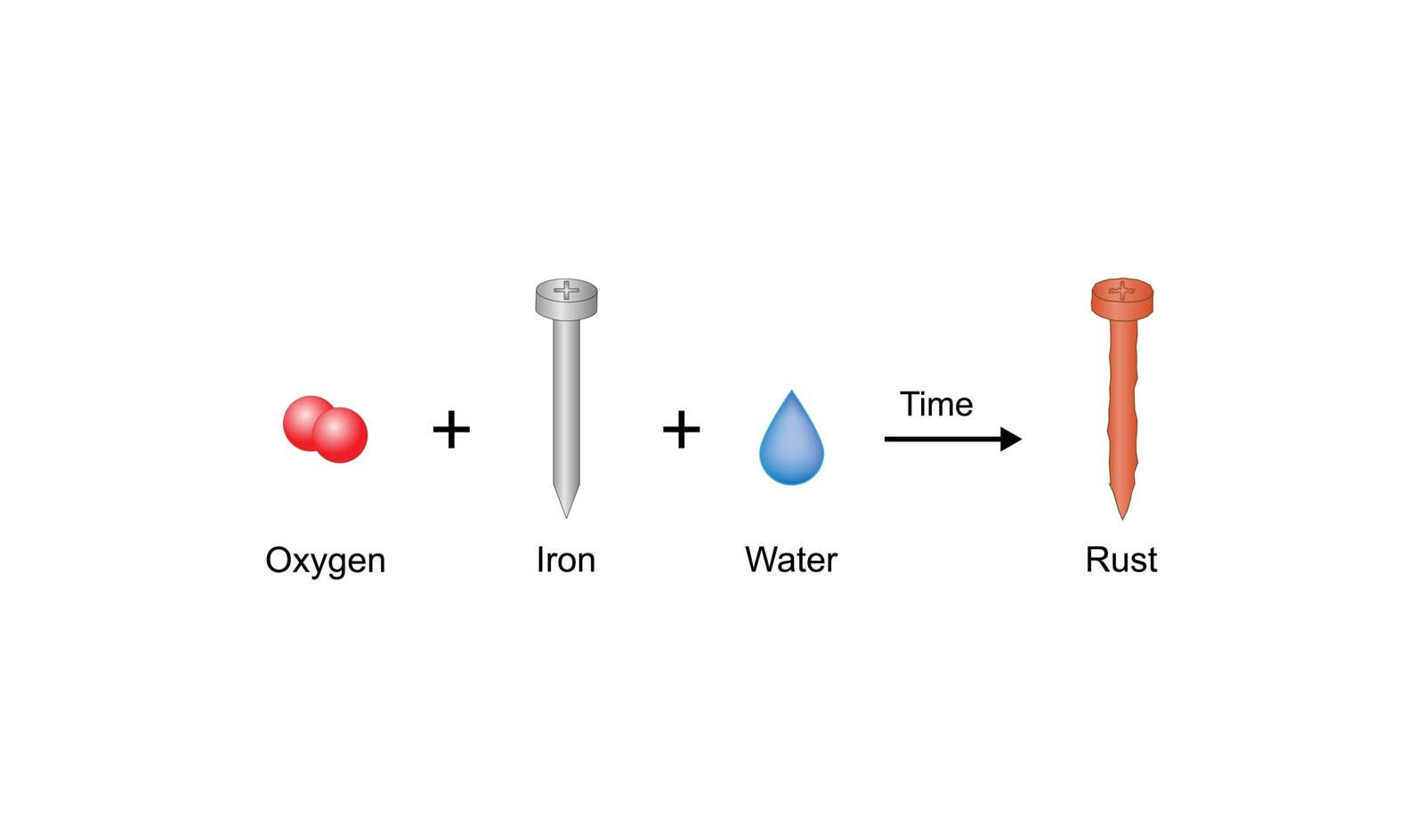
🧪 Rusting is a common chemical change.
Iron reacts with oxygen 🌬️ and moisture 💧.
🧲➡️🟤 Iron + Oxygen + Water → Rust
🧠 Rust weakens iron objects like gates and bridges.
🛠️ Rusting can be prevented.
Painting 🎨
Oiling 🛢️
Galvanization ⚙️
🧠 Prevention saves materials and resources.
🧪 Burning is another chemical change.
Fuel reacts with oxygen to produce heat 🔥 and light ✨.
🧠 Burning helps in cooking 🍳 and heating but must be controlled.
🌱 Photosynthesis is a natural chemical change in plants.
Plants use sunlight ☀️, water 💧, and carbon dioxide 🌬️ to make food.
🧬 This process supports all life on Earth 🌍.
🧭 Some changes are slow, others are fast.
Rusting is slow ⏳.
Burning is fast ⚡.
🧠 Speed of change depends on conditions.
🔄 Some changes are reversible.
Melting and freezing ❄️↔️💧
⛔ Some changes are irreversible.
Cooking food 🍲
Burning paper 🔥
🧠 This classification helps understand material behavior.
🌍 Changes are useful in daily life.
Cooking improves taste 🍲.
Fermentation makes curd 🥣.
Construction uses chemical reactions 🏗️.
🧠 Controlled changes improve quality of life.
🧭 Studying changes develops scientific thinking.
Observation 👀
Comparison 🧩
Reasoning 🧠
🧠 It helps predict results and avoid damage.
📜 Changes are a part of nature.
From weather 🌦️ to growth 🌱, everything involves change.
🚀 Understanding changes prepares students for advanced science.
📝 Summary of the Lesson
Changes occur all around us and can be classified as physical or chemical. Physical changes involve change in shape, size, or state without forming a new substance and are usually reversible. Chemical changes result in the formation of new substances and are often irreversible. Examples of physical changes include melting and tearing, while rusting and burning are chemical changes. Rusting weakens iron but can be prevented using protective methods. Chemical changes like photosynthesis are essential for life. Understanding different types of changes helps us connect science with daily life and use materials wisely.
⚡ Quick Recap
⭐ Changes occur everywhere
⭐ Physical changes are reversible
⭐ Chemical changes form new substances
⭐ Rusting is a chemical change
⭐ Burning releases heat and light
⭐ Some changes are fast, some slow
⭐ Changes affect daily life
——————————————————————————————————————————————————————————————————————————–
TEXTBOOK QUESTIONS
🔒 ❓ Question 1
Which of the following statements are the characteristics of a physical change?
📌 Answer:
✔️ Correct option: (c) (i) and (iii)
🔹 In a physical change, the state of a substance may or may not change.
🔹 No new substance is formed during a physical change.
🔹 Statements (ii) and (iv) describe chemical changes, not physical changes.
🔒 ❓ Question 2
Predict which of the following changes can be reversed and which cannot be reversed. If you are not sure, write that down. Why are you not sure about these?
📌 Answer:
🔵 Reversible changes:
🔹 (ii) Twisting of straight string – can be untwisted.
🔹 (iv) Dissolving sugar in water – sugar can be recovered by evaporation.
🔹 (v) Drawing water from a well – water can be poured back.
🔹 (vii) Boiling water in an open pan – steam can condense back into water.
🔹 (viii) Rolling up a mat – can be unrolled again.
🔴 Irreversible changes:
🔹 (i) Stitching cloth to a shirt – cannot restore original cloth fully.
🔹 (iii) Making idlis from batter – new substance is formed.
🔹 (vi) Ripening of fruits – cannot return to raw stage.
🔹 (ix) Grinding wheat grains to flour – grains cannot be regained.
🔹 (x) Forming of soil from rocks – very slow and irreversible.
🟡 Why some may seem confusing:
🔹 Some changes appear reversible but restoring the original form completely is not practically possible.
🔒 ❓ Question 3
State whether the following statements are True or False. If false, write the correct statement.
📌 Answer:
🔹 (i) Melting of wax is necessary for burning a candle.
✔️ True
🔹 (ii) Collecting water vapour by condensing involves a chemical change.
❌ False
✔️ Correct statement: Condensation of water vapour is a physical change.
🔹 (iii) Converting leaves into compost is a chemical change.
✔️ True
🔹 (iv) Mixing baking soda with lemon juice is a chemical change.
✔️ True
🔒 ❓ Question 4
Fill in the blanks:
📌 Answer:
🔹 (i) Rusting; chemical
🔹 (ii) physical; reversed
🔹 (iii) combustion; chemical
🔹 (iv) magnesium oxide; basic; chemical
🔒 ❓ Question 5
Are the changes of water to ice and water to steam physical or chemical? Explain.
📌 Answer:
🔹 Both changes are physical changes.
🔹 Only the state of water changes, not its chemical composition.
🔹 No new substance is formed, and the changes are reversible.
🔒 ❓ Question 6
Is curdling of milk a physical or chemical change? Justify your statement.
📌 Answer:
🔹 Curdling of milk is a chemical change.
🔹 Milk changes into curd due to formation of new substances.
🔹 The change is irreversible.
🔒 ❓ Question 7
Natural factors like wind and rain help in formation of soil from rocks. Is this a physical or chemical change? Why?
📌 Answer:
🔹 This is mainly a physical change.
🔹 Rocks break into smaller pieces due to weathering.
🔹 No new substance is formed in the process.
🔒 ❓ Question 8
Read the story ‘Eco-friendly Prithvi’ and tick the correct options.
📌 Answer:
🔹 Chopping vegetables, peeling potatoes – Physical changes
🔹 Collecting peels in a clay pot – Physical change
🔹 Decomposition into compost – Chemical change
🔹 Germination and growth of plants – Chemical change
🔒 ❓ Question 9
Classify the following changes into physical, chemical, and both.
📌 Answer:
🔵 Area A (Physical changes):
🔹 Tearing of paper
🔹 Folding of clothes
🔹 Melting of ice
🔴 Area B (Chemical changes):
🔹 Rusting
🔹 Burning of magnesium
🔹 Curdling of milk
🔹 Mixing baking soda with vinegar
🟡 Area C (Both physical and chemical):
🔹 Burning of a candle
🔹 Ripening of fruits
🔒 ❓ Question 10
In which cases does lime water turn milky and why?
📌 Answer:
🔹 Lime water turns milky in cases (a) and (d).
🔹 Carbon dioxide is produced when:
✔️ Vinegar reacts with baking soda
✔️ Lemon juice reacts with baking soda
🔹 CO₂ reacts with lime water to form calcium carbonate, which makes it milky.
——————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS
SECTION 1 — MCQs (5 Questions)
🔒 ❓ Q1. Which of the following is a physical change?
🟢 1️⃣ Rusting of iron
🔵 2️⃣ Burning of paper
🟡 3️⃣ Melting of ice
🟣 4️⃣ Cooking of food
✔️ Answer: 🟡 3️⃣ Melting of ice
🔒 ❓ Q2. Which change produces a new substance?
🟢 1️⃣ Physical change
🔵 2️⃣ Chemical change
🟡 3️⃣ Reversible change
🟣 4️⃣ Temporary change
✔️ Answer: 🔵 2️⃣ Chemical change
🔒 ❓ Q3. Which change is usually irreversible?
🟢 1️⃣ Melting of wax
🔵 2️⃣ Freezing of water
🟡 3️⃣ Rusting of iron
🟣 4️⃣ Stretching of rubber
✔️ Answer: 🟡 3️⃣ Rusting of iron
🔒 ❓ Q4. Which process involves formation of ash?
🟢 1️⃣ Dissolving
🔵 2️⃣ Freezing
🟡 3️⃣ Burning
🟣 4️⃣ Melting
✔️ Answer: 🟡 3️⃣ Burning
🔒 ❓ Q5. Which change can be reversed?
🟢 1️⃣ Burning of coal
🔵 2️⃣ Rusting
🟡 3️⃣ Melting and freezing of water
🟣 4️⃣ Cooking of rice
✔️ Answer: 🟡 3️⃣ Melting and freezing of water
SECTION 2 — Very Short Answer (5 Questions)
🔒 ❓ Q6. Name a change in which no new substance is formed.
📌 ✅ Answer: Physical change
🔒 ❓ Q7. What type of change is rusting?
📌 ✅ Answer: Chemical change
🔒 ❓ Q8. Is melting of ice reversible or irreversible?
📌 ✅ Answer: Reversible
🔒 ❓ Q9. Name one irreversible change.
📌 ✅ Answer: Burning
🔒 ❓ Q10. What is formed during burning of paper?
📌 ✅ Answer: Ash
SECTION 3 — Short Answer (3 Questions)
🔒 ❓ Q11. What is a physical change?
📌 ✅ Answer:
🔹 A physical change is a change in which no new substance is formed.
🔸 Only the shape, size or state of a substance changes.
🔹 Such changes are usually temporary and reversible.
🔒 ❓ Q12. Why is rusting called a chemical change?
📌 ✅ Answer:
🔹 Rusting forms a new substance called rust.
🔸 The original iron cannot be obtained back easily.
🔹 Therefore, rusting is a chemical and irreversible change.
🔒 ❓ Q13. How is a chemical change different from a physical change?
📌 ✅ Answer:
🔹 In a chemical change, a new substance is formed.
🔸 In a physical change, no new substance is formed.
🔹 Chemical changes are mostly irreversible.
SECTION 4 — Long Answer (1 Question)
🔒 ❓ Q14. Explain physical and chemical changes with examples.
📌 ✅ Answer:
🔹 Physical changes involve change in shape, size or state without forming a new substance.
🔸 Melting of ice and stretching of rubber are physical changes.
🔹 Chemical changes produce new substances and are irreversible.
🔸 Rusting of iron and burning of paper are chemical changes.
——————————————————————————————————————————————————————————————————————————–
ADVANCE KNOWLEDGE
🌍 Change: The Only Constant in Nature
Nothing in nature stays exactly the same. Mountains slowly wear down, seasons shift, metals rust, food cooks, and living beings grow. These are not random events—they are changes, driven by energy, time, and interactions between substances.
🧠 Big idea:
Change is how matter responds when conditions or interactions alter.
🧠 Two Great Families of Change
Scientists group changes based on what happens to the substance itself.
🔄 Physical Changes
Appearance changes
No new substance formed
Often reversible
⚗️ Chemical Changes
New substance formed
Properties change completely
Mostly irreversible
⭐ This distinction helps us predict outcomes and control processes.
⚠️ Misconception vs Reality
⚠️ Misconception: All visible changes are chemical
✅ Reality: Many visible changes are purely physical
⚠️ Misconception: Irreversible means chemical always
✅ Reality: Some physical changes are also irreversible (like breaking glass)
🔄 Physical Changes: Same Substance, New Look
Physical changes rearrange particles without changing identity.
🧠 Common examples:
Melting ice
Cutting paper
Stretching rubber
⭐ At the microscopic level, particles remain the same—only distance and arrangement change.
🧠 Physical changes often involve:
Change of state
Change of shape
Change of size
🌡️ Energy and Physical Change
Physical changes are closely linked to energy transfer.
🌡️ Heating:
Increases particle motion
Causes expansion or melting
❄️ Cooling:
Slows particles
Causes contraction or freezing
⭐ Energy moves in and out, but matter keeps its identity.
⚗️ Chemical Changes: Matter Becomes Something New
Chemical changes create new substances with new properties.
🧪 Examples:
Rusting of iron
Burning of wood
Cooking food
🧠 In chemical change:
Old bonds break
New bonds form
⭐ This rearrangement changes behaviour, colour, hardness, smell, and reactivity.
🔥 Signs of a Chemical Change
Scientists look for clues.
🧪 Common indicators:
Colour change
Gas release
Heat or light produced
Formation of a solid
⚠️ Not all signs appear every time—but at least one usually does.
⏳ A Historical Shift in Understanding Change
⏳ Ancient thinkers believed:
Matter could disappear
Fire destroyed substance
🧠 Modern science revealed:
Matter is conserved
Atoms rearrange, not vanish
⭐ This idea transformed chemistry and industry.
🧠 Reversibility: A Helpful Guide, Not a Rule
🔄 Reversible changes
Melting and freezing
Stretching and relaxing
⚗️ Irreversible changes
Burning
Rusting
🧠 But remember: Reversibility is a clue, not a strict law.
🌍 Rusting: A Slow Chemical Drama
Rusting is a classic chemical change.
🧠 Needs:
Iron
Oxygen
Moisture
⚠️ Rust weakens structures.
🧠 Prevention methods:
Painting
Galvanizing
Alloying
⭐ Science protects materials by controlling chemical reactions.
🔥 Burning: Rapid Chemical Change
Burning is fast oxidation.
🧠 Requires:
Fuel
Oxygen
Heat
⭐ Produces:
Heat
Light
New substances (ash, gases)
⚠️ Controlled burning powers engines; uncontrolled burning causes disasters.
🌱 Changes in Living Systems
Life itself is a series of controlled chemical changes.
🧬 Examples:
Digestion
Respiration
Growth
🧠 These changes are carefully regulated—unlike uncontrolled reactions like rusting.
🚀 Modern Science: Designing Changes
🚀 Scientists design changes intentionally.
🧠 Examples:
Making alloys stronger
Creating medicines
Preserving food
⭐ Industry depends on predicting and controlling changes.
⚠️ Environmental Impact of Chemical Changes
Some chemical changes harm nature.
🌍 Problems include:
Pollution
Acid rain
Global warming
🧠 Understanding chemical change helps us:
Reduce harm
Design safer processes
🧪 Physical vs Chemical: Borderline Cases
Some changes confuse learners.
🧠 Examples:
Dissolving salt in water (physical, but tricky)
Cooking food (chemical, even though shape changes)
⭐ Always ask:
👉 Is a new substance formed?
🌍 Change and Time: Fast vs Slow
Changes differ in speed.
⏳ Slow:
Rusting
Soil formation
⚡ Fast:
Explosion
Burning
🧠 Speed depends on:
Temperature
Surface area
Nature of substances
⭐ Amazing Facts
⭐ Glass flows extremely slowly—appearing solid
⭐ Fireflies produce light through chemical reactions
⭐ Our bodies run on thousands of chemical changes every second
⭐ Even rocks change shape over millions of years
🧠 Why Curious Minds Must Understand Change
Understanding change teaches:
Cause and effect
Prediction
Control
🧠 It connects physics, chemistry, biology, and environmental science into one thinking system.
🌟 Final Thought
Nothing changes without a reason.
Nothing stays the same without balance.
🧠 To understand change is to understand how the world keeps moving forward.
——————————————————————————————————————————————————————————————————————————–
