Class 12 : Physics (English) – Chapter 13: Nuclei
EXPLANATION & SUMMARY
🔵 Introduction to the Nucleus
The atom, once thought to be indivisible, is now understood as a system with a tiny but massive nucleus at its centre.
This nucleus contains protons (positively charged) and neutrons (neutral), collectively called nucleons.
➡️ The size of the nucleus is about 10⁻¹⁵ m, whereas the size of the atom is about 10⁻¹⁰ m.
✔️ This means that if an atom were a stadium, the nucleus would be like a marble at the centre, yet carrying nearly all the mass.
🟢 Atomic Mass and Composition
Atomic number (Z): Number of protons.
Mass number (A): Total number of protons + neutrons.
Neutron number (N): N = A – Z.
💡 Concept:
Isotopes → Same Z, different N.
Isobars → Same A, different Z.
Isotones → Same N, different Z.
✏️ Note: Atomic masses are measured in u (atomic mass unit), where 1 u = 1/12th of the mass of a carbon-12 atom.
🔴 Size of the Nucleus
Nuclear radius formula:
R = R₀ A^(1/3), with R₀ ≈ 1.2 × 10⁻¹⁵ m.
➡️ Since radius grows only as cube root of A:
✔️ Larger nuclei are not proportionally much bigger than lighter nuclei.
Volume ∝ A
Mass ∝ A
Hence, nuclear density ≈ constant ≈ 2.3 × 10¹⁷ kg/m³.
🌿 This density is unimaginably high: A sugar cube of nuclear matter would weigh billions of tonnes!
🟡 Mass Defect and Binding Energy
The actual mass of a nucleus is less than the sum of masses of its nucleons.
This difference = Mass defect (Δm).
Formula:
Δm = [Z m_p + N m_n] – m_nucleus
➡️ Binding energy (B) is energy equivalent of Δm:
B = Δm c²
Binding energy per nucleon (B/A): A measure of nuclear stability.
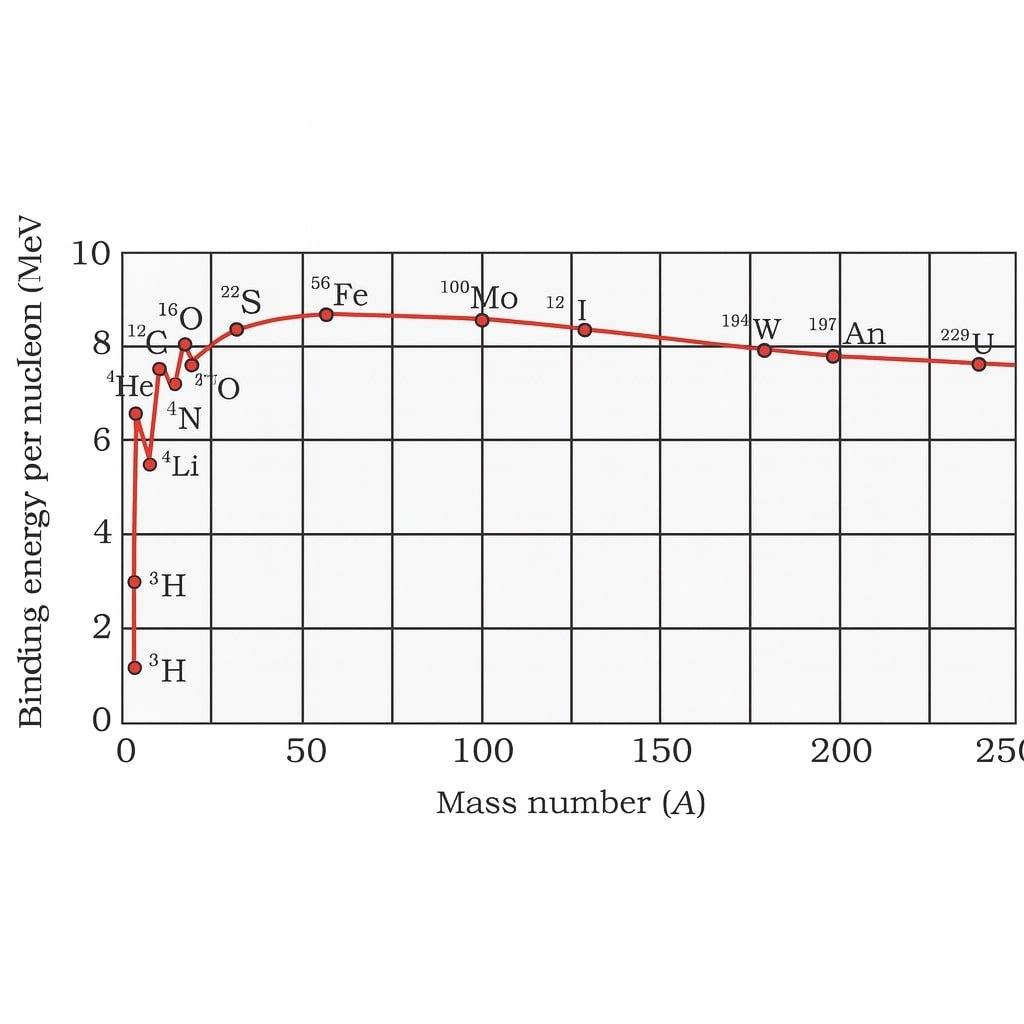
✔️ Curve of B/A vs A shows:
🔵 Light nuclei have small B/A.
🟢 Maximum stability at iron (A ≈ 56).
🔴 Very heavy nuclei have smaller B/A.
💡 This explains:
Fusion of light nuclei → energy release.
Fission of heavy nuclei → energy release.
🔵 Nuclear Force

Nucleons are held together by the nuclear force.
Properties:
✔️ Extremely strong (much stronger than electrostatic force).
✔️ Short range (works within ~2–3 femtometres).
✔️ Charge independent (acts same between p–p, n–n, p–n).
✔️ Saturation → each nucleon interacts only with nearest neighbours.
✏️ Note: Despite protons repelling by Coulomb’s law, nuclear force keeps them bound.
🟢 Radioactivity
Unstable nuclei undergo radioactive decay spontaneously.
Types:
Alpha decay (α): Nucleus emits a helium nucleus (2p + 2n).
Z decreases by 2, A decreases by 4.
Beta minus decay (β⁻): Neutron → Proton + electron + antineutrino.
Z increases by 1, A unchanged.
Beta plus decay (β⁺): Proton → Neutron + positron + neutrino.
Z decreases by 1, A unchanged.
Gamma decay (γ): Excited nucleus emits a photon; A and Z unchanged.
🔴 Law of Radioactive Decay
If N(t) is the number of undecayed nuclei at time t:
N(t) = N₀ e^(−λt)
where λ = decay constant.
Activity A(t) = λN(t).
Half-life T₁/₂ = (ln 2)/λ.
Mean life τ = 1/λ = 1.44 T₁/₂.
✔️ The law is exponential and universal.
💡 Example: If activity drops to 1/16th, 4 half-lives have passed.
🟡 Nuclear Energy
(a) Fission
➡️ A heavy nucleus like U-235 splits into two smaller nuclei when struck by a neutron.
Releases ~200 MeV per fission.
Used in nuclear reactors and atomic bombs.
✔️ In reactors, control rods regulate neutron supply to keep reaction steady.
(b) Fusion
➡️ Two light nuclei combine, e.g.:
²H + ³H → ⁴He + n + 17.6 MeV
Source of energy in stars.
Requires very high temperature (~10⁷ K).
Promising for future energy (hydrogen bomb, fusion reactors).
🔵 Applications of Radioactivity
Carbon dating: C-14 (half-life 5730 years) helps determine age of fossils.
Medical use: I-131 for thyroid, Co-60 for cancer therapy.
Industrial use: Tracers in pipelines, thickness gauges.
🟢 Environmental Concerns
Fission reactors produce radioactive waste.
Accidents (Chernobyl, Fukushima) highlight risks.
Fusion is cleaner but technologically challenging.
✨ Summary Section (~300 words)
🔵 Overview
The nucleus is a tiny, dense core with protons and neutrons.
Mass number (A) = Z + N.
Nuclear radius ∝ A^(1/3); density nearly constant.
🟢 Key Concepts
Mass defect arises because the nucleus is lighter than its nucleons’ sum.
This lost mass = binding energy, showing stability.
Binding energy per nucleon peaks at iron (A ≈ 56).
🔴 Forces
Nuclear force: short-ranged, very strong, charge-independent, saturating.
🟡 Radioactivity
Decay follows exponential law.
Half-life T₁/₂ = ln 2 / λ.
Mean life τ = 1.44 T₁/₂.
α decay → Z–2, A–4.
β⁻ decay → Z+1.
β⁺ decay → Z–1.
γ decay → Z, A unchanged.
🔵 Nuclear Energy
Fission: Heavy nucleus splits → 200 MeV energy.
Fusion: Light nuclei fuse → immense energy (sun, hydrogen bomb).
🟢 Applications
Archaeology (carbon dating).
Medicine (diagnosis, therapy).
Industry (tracers, thickness control).
🔴 Concerns
Fission → radioactive waste.
Fusion → promising but challenging.
📝 Quick Recap
✔️ Nucleus = protons + neutrons; tiny but massive.
✔️ Nuclear radius ∝ A^(1/3); density constant.
✔️ Mass defect → binding energy; stability peaks at Fe.
✔️ Radioactive decay follows N(t) = N₀ e^(−λt).
✔️ Fission (heavy nuclei split) and fusion (light nuclei join) release huge energy.
✔️ Uses: dating, medicine, industry; concerns: waste, safety, environment.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
Question 13.1
Obtain the binding energy (in MeV) of a nitrogen nucleus (₇¹⁴N).
Given: m(₇¹⁴N) = 14.00307 u, mₚ = 1.007825 u, mₙ = 1.008665 u.
Answer 13.1
🔵 Step 1: Identify Z and N
Z = 7, A = 14 ⇒ N = 14 − 7 = 7
🟢 Step 2: Mass defect
Δm = [Z mₚ + N mₙ] − m(nucleus)
= [7(1.007825) + 7(1.008665)] − 14.00307
= 14.11543 − 14.00307 = 0.11236 u
🟠 Step 3: Binding energy
B = Δm × 931.5 = 0.11236 × 931.5 ≈ 104.66 MeV
🔴 Step 4: Binding energy per nucleon
B/A = 104.66 / 14 ≈ 7.48 MeV
✔️ Final Answer: Binding energy ≈ 104.66 MeV
Question 13.2
Obtain the binding energy of the nuclei ₂₆⁵⁶Fe and ₈₃²⁰⁸Bi.
Given: m(₂₆⁵⁶Fe) = 55.934939 u, m(₈₃²⁰⁸Bi) = 208.980388 u.
Answer 13.2
🔵 For ₂₆⁵⁶Fe: Z = 26, N = 30
Δm = [26(1.007825) + 30(1.008665)] − 55.934939
= 56.46340 − 55.934939 = 0.528461 u
B = 0.528461 × 931.5 ≈ 492.26 MeV
B/A = 492.26 / 56 ≈ 8.79 MeV
🟢 For ₈₃²⁰⁸Bi: Z = 83, N = 125
Δm = [83(1.007825) + 125(1.008665)] − 208.980388
= 209.732600 − 208.980388 = 0.752212 u
B = 0.752212 × 931.5 ≈ 700.69 MeV
B/A = 700.69 / 208 ≈ 3.37 MeV
✔️ Final Answer: Fe-56 = 492.26 MeV (8.79 MeV/nucleon); Bi-208 = 700.69 MeV (3.37 MeV/nucleon)
Question 13.3
A coin of mass 3.0 g is entirely made of ₂₉⁶³Cu (m = 62.92960 u). Calculate the nuclear energy required to separate all the neutrons and protons.
Answer 13.3
🔵 Step 1: Number of atoms
Moles = 3 / 63 = 0.04762 mol
Atoms = 0.04762 × 6.023×10²³ ≈ 2.87×10²² nuclei
🟢 Step 2: Mass defect for ₂₉⁶³Cu
Z = 29, N = 34
Δm = [29(1.007825) + 34(1.008665)] − 62.92960
= 63.521535 − 62.92960 = 0.591935 u
🟠 Step 3: Binding energy per nucleus
B = 0.591935 × 931.5 ≈ 551.39 MeV
🔴 Step 4: Total energy
E(MeV) = 551.39 × 2.87×10²² ≈ 1.58×10²⁵ MeV
E(J) = 1.58×10²⁵ × 1.6×10⁻¹³ ≈ 2.53×10¹² J
✔️ Final Answer: ≈ 2.53×10¹² J
Question 13.4
Obtain approximately the ratio of the nuclear radii of ₇₉¹⁹⁷Au and ₄₇¹⁰⁷Ag.
Answer 13.4
🔵 Formula: R ∝ A^(1/3)
🟢 Ratio = (197/107)^(1/3) ≈ 1.226
✔️ Final Answer: R(Au)/R(Ag) ≈ 1.23
Question 13.5
Calculate Q-value of the reactions:
(i) ²H + ²H → ³He + ¹n
(ii) ¹²C + ²H → ²⁰Ne + ⁴He
Given: m(²H) = 2.014102 u, m(³He) = 3.016049 u, m(¹²C) = 12.000000 u, m(²⁰Ne) = 19.992439 u, m(⁴He) = 4.002603 u, mₙ = 1.008665 u.
Answer 13.5
🔹 For (i):
Δm = [2×2.014102] − [3.016049 + 1.008665] = 0.003490 u
Q = 0.003490 × 931.5 ≈ 3.25 MeV (positive)
✔️ Exothermic
🔹 For (ii):
Δm = [12.000000 + 2.014102] − [19.992439 + 4.002603] = −9.98094 u
Q = −9.98094 × 931.5 ≈ −9.30×10³ MeV (negative)
✔️ Endothermic
Question 13.6
Check whether fission of ₂₆⁵⁶Fe → 2(₁₃²⁸Al) is energetically possible.
Given: m(₂₆⁵⁶Fe) = 55.93494 u, m(₁₃²⁸Al) = 27.98191 u.
Answer 13.6
🔵 Mass defect: Δm = 55.93494 − 2(27.98191) = −0.02888 u
🟢 Q = −0.02888 × 931.5 ≈ −26.90 MeV
✔️ Final Answer: Q < 0 ⇒ Fission is not energetically possible
Question 13.7
The fission properties of ₉₄²³⁹Pu are very similar to those of ₉₂²³⁵U. The average energy released per fission is 180 MeV. How much energy, in MeV, is released if all the atoms in 1 kg of ₉₄²³⁹Pu undergo fission?
Answer 13.7
🔵 Step 1: Moles of ₂³⁹Pu
molar mass ≈ 239 g/mol → moles = 1000 g / 239 g/mol ≈ 4.184 mol
🟢 Step 2: Number of atoms
atoms = 4.184 × 6.023×10²³ ≈ 2.52×10²⁴
🟠 Step 3: Total fission energy
E = (number of fissions) × (energy per fission)
E ≈ 2.52×10²⁴ × 180 MeV ≈ 4.54×10²⁶ MeV
✔️ Final Answer: ≈ 4.54×10²⁶ MeV
Question 13.8
How long can an electric lamp of 100 W be kept glowing by fusion of 2.0 kg of deuterium?
Take the fusion reaction as: ²H + ²H → ³He + n + 3.27 MeV.
Answer 13.8
🔵 Step 1: Amount of deuterium atoms
molar mass of D ≈ 2 g/mol → moles = 2000 g / 2 g/mol = 1000 mol
atoms = 1000 × 6.023×10²³ = 6.023×10²⁶
🟢 Step 2: Number of fusion reactions (2 D per reaction)
reactions = 6.023×10²⁶ / 2 = 3.012×10²⁶
🟠 Step 3: Total energy released
E(MeV) = 3.012×10²⁶ × 3.27 ≈ 9.85×10²⁶ MeV
convert to joules: E(J) = 9.85×10²⁶ × 1.6×10⁻¹³ ≈ 1.58×10¹⁴ J
🔴 Step 4: Time for a 100 W lamp (P = 100 J s⁻¹)
t = E / P = 1.58×10¹⁴ / 100 = 1.58×10¹² s
in years: t ≈ 1.58×10¹² / 3.154×10⁷ ≈ 5.0×10⁴ years
✔️ Final Answer: ≈ 5.0×10⁴ years
Question 13.9
Calculate the height of the potential barrier for a head-on collision of two deuterons. (Hint: Treat deuterons as hard spheres of radius 2.0 fm that just touch each other.)
Answer 13.9
🔵 Step 1: Separation at contact
each radius = 2.0 fm → centre-to-centre r = 4.0 fm = 4.0×10⁻¹⁵ m
🟢 Step 2: Coulomb potential energy
U = (1 / 4πϵ₀) (e² / r)
≈ (9.0×10⁹) × (1.6×10⁻¹⁹)² / (4.0×10⁻¹⁵) J
🟠 Step 3: Compute
(1.6×10⁻¹⁹)² = 2.56×10⁻³⁸
U ≈ 9.0×10⁹ × (2.56×10⁻³⁸ / 4.0×10⁻¹⁵)
= 9.0×10⁹ × 6.4×10⁻²⁴ = 5.76×10⁻¹⁴ J
🔴 Step 4: Convert to MeV (1 MeV = 1.6×10⁻¹³ J)
U ≈ 5.76×10⁻¹⁴ / 1.6×10⁻¹³ ≈ 0.36 MeV
✔️ Final Answer: ≈ 0.36 MeV
Question 13.10
From R = R₀ A^(1/3), where R₀ is a constant and A is the mass number of a nucleus, show that the nuclear matter density is nearly constant (independent of A).
Answer 13.10
🔵 Step 1: Nuclear mass
M ≈ A m_N, where m_N is average nucleon mass (≈ constant)
🟢 Step 2: Nuclear volume
V = (4/3)πR³ = (4/3)π (R₀ A^(1/3))³ = (4/3)π R₀³ A
🟠 Step 3: Density
ρ = M / V = (A m_N) / ((4/3)π R₀³ A) = m_N / ((4/3)π R₀³)
🔴 Step 4: Conclusion
A cancels out; ρ depends only on constants m_N and R₀ → independent of A
✔️ Final Answer: Nuclear density is nearly constant for all nuclei
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
Section A (Q1–Q18: MCQs)
Question 1
Which of the following correctly represents the relation between A, Z, and N?
🔵 (A) A = Z – N
🟢 (B) A = Z + N
🟠 (C) A = Z × N
🔴 (D) A = Z/N
Answer: (B) A = Z + N
✔️ Final Answer: A = Z + N
Question 2
The radius of a nucleus varies with mass number as:
🔵 (A) R ∝ A
🟢 (B) R ∝ A^(1/2)
🟠 (C) R ∝ A^(1/3)
🔴 (D) R ∝ A^(2/3)
Answer: (C) R ∝ A^(1/3)
✔️ Final Answer: R ∝ A^(1/3)
Question 3
The approximate nuclear density is:
🔵 (A) 10^3 kg/m^3
🟢 (B) 10^6 kg/m^3
🟠 (C) 10^12 kg/m^3
🔴 (D) 10^17 kg/m^3
Answer: (D) 10^17 kg/m^3
✔️ Final Answer: 10^17 kg/m^3
Question 4
Which force binds protons and neutrons inside the nucleus?
🔵 (A) Gravitational force
🟢 (B) Coulomb force
🟠 (C) Strong nuclear force
🔴 (D) Weak nuclear force
Answer: (C) Strong nuclear force
✔️ Final Answer: Strong nuclear force
Question 5
In beta-minus (β⁻) decay, the atomic number:
🔵 (A) decreases by 1
🟢 (B) increases by 1
🟠 (C) remains unchanged
🔴 (D) decreases by 2
Answer: (B) increases by 1
✔️ Final Answer: Increases by 1
Question 6
The half-life of a radioactive sample is 10 days. In 30 days, its activity will reduce to:
🔵 (A) 1/2
🟢 (B) 1/4
🟠 (C) 1/8
🔴 (D) 1/16
Answer: (C) 1/8
✔️ Final Answer: 1/8
Question 7
The maximum binding energy per nucleon is observed for nuclei around:
🔵 (A) Hydrogen
🟢 (B) Helium
🟠 (C) Iron
🔴 (D) Uranium
Answer: (C) Iron
✔️ Final Answer: Iron (A ≈ 56)
Question 8
If Q-value of a reaction is positive, the reaction is:
🔵 (A) Endothermic
🟢 (B) Exothermic
🟠 (C) Forbidden
🔴 (D) Impossible
Answer: (B) Exothermic
✔️ Final Answer: Exothermic
Question 9
Mean life τ and half-life T1/2 are related as:
🔵 (A) τ = T1/2
🟢 (B) τ = T1/2 / ln2
🟠 (C) τ = T1/2 × ln2
🔴 (D) τ = (T1/2)^2
Answer: (B) τ = T1/2 / ln2
✔️ Final Answer: τ = T1/2 / ln2
Question 10
Carbon dating is based on isotope:
🔵 (A) C-12
🟢 (B) C-13
🟠 (C) C-14
🔴 (D) C-11
Answer: (C) C-14
✔️ Final Answer: C-14
Question 11
Which of the following statements is correct for gamma (γ) emission?
🔵 (A) A decreases, Z decreases
🟢 (B) A unchanged, Z unchanged
🟠 (C) A increases, Z decreases
🔴 (D) A unchanged, Z increases
Answer: (B) A unchanged, Z unchanged
✔️ Final Answer: A and Z unchanged
Question 12
The SI unit of radioactivity is:
🔵 (A) Curie (Ci)
🟢 (B) Becquerel (Bq)
🟠 (C) Tesla (T)
🔴 (D) Weber (Wb)
Answer: (B) Becquerel (Bq)
✔️ Final Answer: Becquerel (Bq)
Question 13
Which of the following has the least penetration power?
🔵 (A) α-particle
🟢 (B) β-particle
🟠 (C) γ-ray
🔴 (D) Neutrino
Answer: (A) α-particle
✔️ Final Answer: α-particle
Question 14
The equation of radioactive decay is:
🔵 (A) N = N0 e^(λt)
🟢 (B) N = N0 e^(−λt)
🟠 (C) N = N0 (1 − λt)
🔴 (D) N = N0 λt
Answer: (B) N = N0 e^(−λt)
✔️ Final Answer: N = N0 e^(−λt)
Question 15
Which conservation law explains neutrino emission in β-decay?
🔵 (A) Conservation of momentum
🟢 (B) Conservation of lepton number
🟠 (C) Conservation of charge
🔴 (D) All of these
Answer: (D) All of these
✔️ Final Answer: All of these
Question 16
Binding energy per nucleon decreases for very heavy nuclei because:
🔵 (A) Coulomb repulsion increases
🟢 (B) Nuclear force becomes weak
🟠 (C) Electrons dominate
🔴 (D) Protons escape
Answer: (A) Coulomb repulsion increases
✔️ Final Answer: Coulomb repulsion increases
Question 17
If activity becomes 1/16 of initial, number of elapsed half-lives is:
🔵 (A) 2
🟢 (B) 3
🟠 (C) 4
🔴 (D) 5
Answer: (C) 4
✔️ Final Answer: 4 half-lives
Question 18
The mass defect Δm of a nucleus leads to binding energy B according to:
🔵 (A) B = Δm × g
🟢 (B) B = Δm × c^2
🟠 (C) B = Δm × h
🔴 (D) B = Δm / c^2
Answer: (B) B = Δm × c^2
✔️ Final Answer: B = Δm × c^2
Section B (Q19–Q23: Very Short Answer)
Question 19
Define binding energy per nucleon. Why is it a measure of stability?
Answer:
🔵 B/A = total binding energy ÷ A.
🟢 Higher B/A ⇒ nucleons more tightly bound.
✔️ Final Answer: Binding energy per nucleon is stability index
Question 20
State two properties of nuclear force.
Answer:
🔵 Very strong but short-ranged (~2–3 fm).
🟢 Saturating nature – each nucleon interacts with nearby nucleons only.
✔️ Final Answer: Strong, short-ranged, saturating
Question 21
Write the relation between half-life and decay constant.
Answer:
🔵 N = N0 e^(−λt).
🟢 At t = T1/2, N = N0/2.
🟠 ⇒ e^(−λT1/2) = 1/2 → λT1/2 = ln2.
✔️ Final Answer: T1/2 = ln2 / λ
Question 22
What is Q-value of a nuclear reaction? When is it exothermic?
Answer:
🔵 Q = [m(initial) − m(final)] c^2.
🟢 If Q > 0, exothermic.
✔️ Final Answer: Q > 0 → exothermic
Question 23
Why is U-235 used as fuel in reactors instead of U-238?
Answer:
🔵 U-235 undergoes fission with thermal neutrons.
🟢 U-238 requires fast neutrons.
✔️ Final Answer: U-235 is fissionable with slow neutrons
Section C (Q24–Q28: Mid-length Numericals/Theory)
Question 24
A nucleus has A = 64. Calculate its radius using R0 = 1.2×10^−15 m.
Answer:
🔵 Formula: R = R0 A^(1/3).
🟢 64^(1/3) = 4.
🟠 R = 1.2×10^−15 × 4 = 4.8×10^−15 m.
✔️ Final Answer: 4.8×10^−15 m
Question 25
The activity of a sample drops from 8000/s to 1000/s. How many half-lives have elapsed?
Answer:
🔵 Ratio = 1000/8000 = 1/8 = (1/2)^3.
🟢 Elapsed half-lives = 3.
✔️ Final Answer: 3 half-lives
Question 26
A radionuclide has mean life 10 h. Calculate half-life.
Answer:
🔵 Relation: τ = T1/2 / ln2.
🟢 T1/2 = τ ln2 = 10 × 0.693 = 6.93 h.
✔️ Final Answer: 6.93 h
Question 27
Explain why α-decay occurs in heavy nuclei.
Answer:
🔵 Coulomb repulsion is very high in heavy nuclei.
🟢 α-emission reduces Z and A, lowering Coulomb energy.
✔️ Final Answer: α-decay reduces repulsion, giving stability
Question 28
Show that nuclear density is independent of A.
Answer:
🔵 R = R0 A^(1/3).
🟢 V ∝ A, M ∝ A.
🟠 ρ = M/V = constant.
✔️ Final Answer: Nuclear density constant, independent of A
Section D (Q29–Q31: Long Answer)
Question 29
Derive the law of radioactive decay.
Answer:
🔵 dN/dt = −λN.
🟢 ∫ dN/N = −λ ∫ dt.
🟠 lnN = −λt + C.
🔴 N = N0 e^(−λt).
🟣 T1/2 = ln2 / λ.
✔️ Final Answer: N = N0 e^(−λt), T1/2 = ln2 / λ
Question 30
Explain binding energy and its relation with mass defect.
Answer:
🔵 Δm = Zmp + Nmn − Mnucleus.
🟢 Binding energy B = Δm c^2.
🟠 B/A measures stability.
✔️ Final Answer: B = Δm c^2, B/A = stability index
Question 31
Discuss α, β, and γ decays in terms of A and Z.
Answer:
🔵 α: ΔA = −4, ΔZ = −2.
🟢 β⁻: ΔA = 0, ΔZ = +1.
🟠 β⁺: ΔA = 0, ΔZ = −1.
🔴 γ: ΔA = 0, ΔZ = 0.
✔️ Final Answer: α changes A,Z; β changes Z; γ leaves A,Z same
Section E (Q32–Q33: Case/Application)
Question 32
A radioactive sample has N0 nuclei. Write the expression for total decays recorded by a detector of efficiency η in time T.
Answer:
🔵 N(t) = N0 e^(−λt).
🟢 A(t) = λN0 e^(−λt).
🟠 R(t) = ηA(t).
🔴 C = ηN0 (1 − e^(−λT)).
✔️ Final Answer: C = ηN0 (1 − e^(−λT))
Question 33
An α-decay has Q = 5.3 MeV. If daughter mass ≈ 56 times α, find K of α-particle.
Answer:
🔵 Kd/Kα = mα/md = 1/56.
🟢 Q = Kα (1 + 1/56).
🟠 Kα = 5.3 × 56/57 ≈ 5.21 MeV.
🔴 Kd ≈ 0.09 MeV.
✔️ Final Answer: Kα ≈ 5.21 MeV, Kd ≈ 0.09 MeV
————————————————————————————————————————————————————————————————————————————
