Class 12 : Chemistry (English) – Chapter 1: Solutions
EXPLANATION & SUMMARY
This chapter primarily deals with different types of solutions, the methods to express their concentration, the concept of solubility, the colligative properties of solutions, and Raoult’s law. Here’s a complete explanation of all concepts covered in this lesson in clear and systematic order:
TYPES OF SOLUTIONS
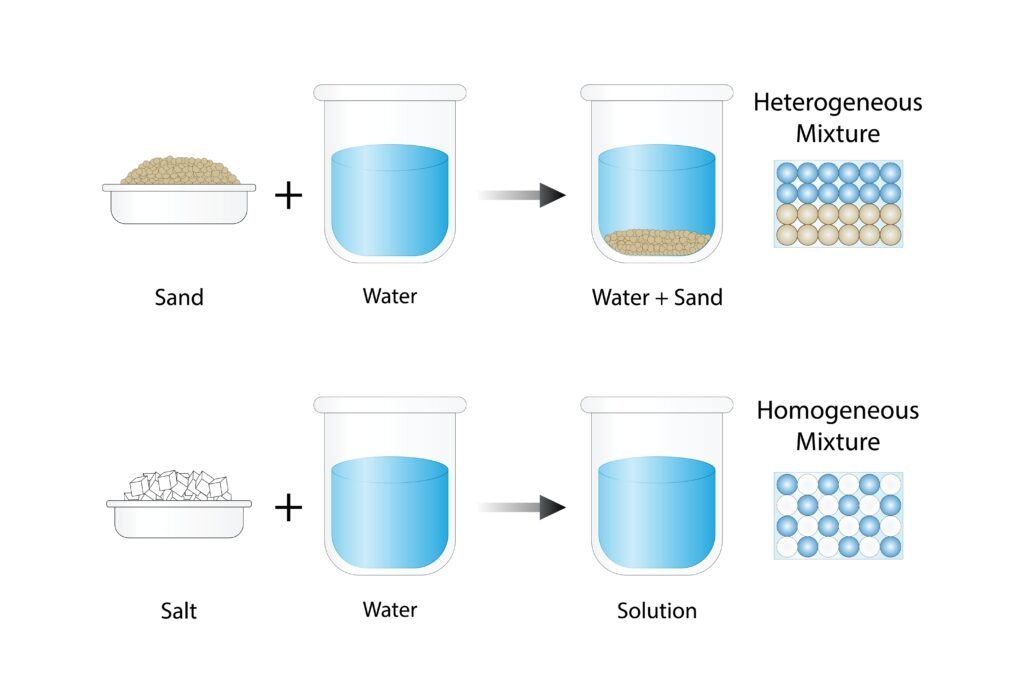
A solution is a homogeneous mixture of two or more substances. The component present in the largest amount is usually called the solvent, and the other is the solute.
Types of Solutions Based on Physical States:
Gaseous Solutions: Gas in gas (air), gas in liquid (oxygen in water)
Liquid Solutions: Liquid in liquid (ethanol in water), solid in liquid (salt in water)
Solid Solutions: Solid in solid (alloys like brass – zinc in copper)
EXPRESSING CONCENTRATION OF SOLUTIONS
The concentration of a solution tells how much solute is present in a given quantity of solvent or solution.
Common ways to express concentration:
a. Mass Percentage (w/w):
Mass of solute / Mass of solution × 100
b. Volume Percentage (v/v):
Volume of solute / Volume of solution × 100
c. Mass by Volume Percentage (w/v):
Mass of solute / Volume of solution × 100
d. Parts per million (ppm):
Used for very dilute solutions.
Mass of solute / Mass of solution × 10⁶
e. Mole Fraction (χ):
Moles of component / Total moles of all components
If there are only two components A and B:
χA + χB = 1
f. Molarity (M):
Moles of solute / Volume of solution in litres
g. Molality (m):
Moles of solute / Mass of solvent in kg
Molality is temperature-independent.
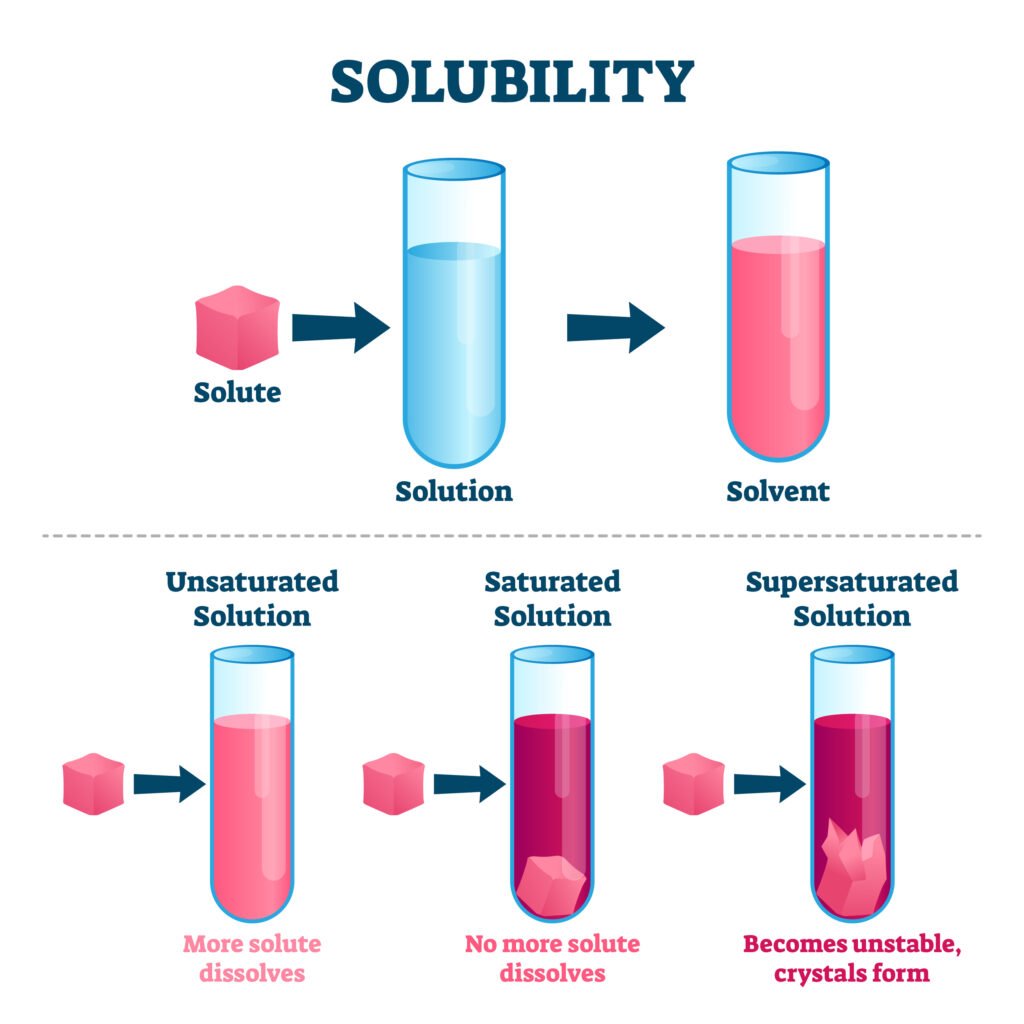
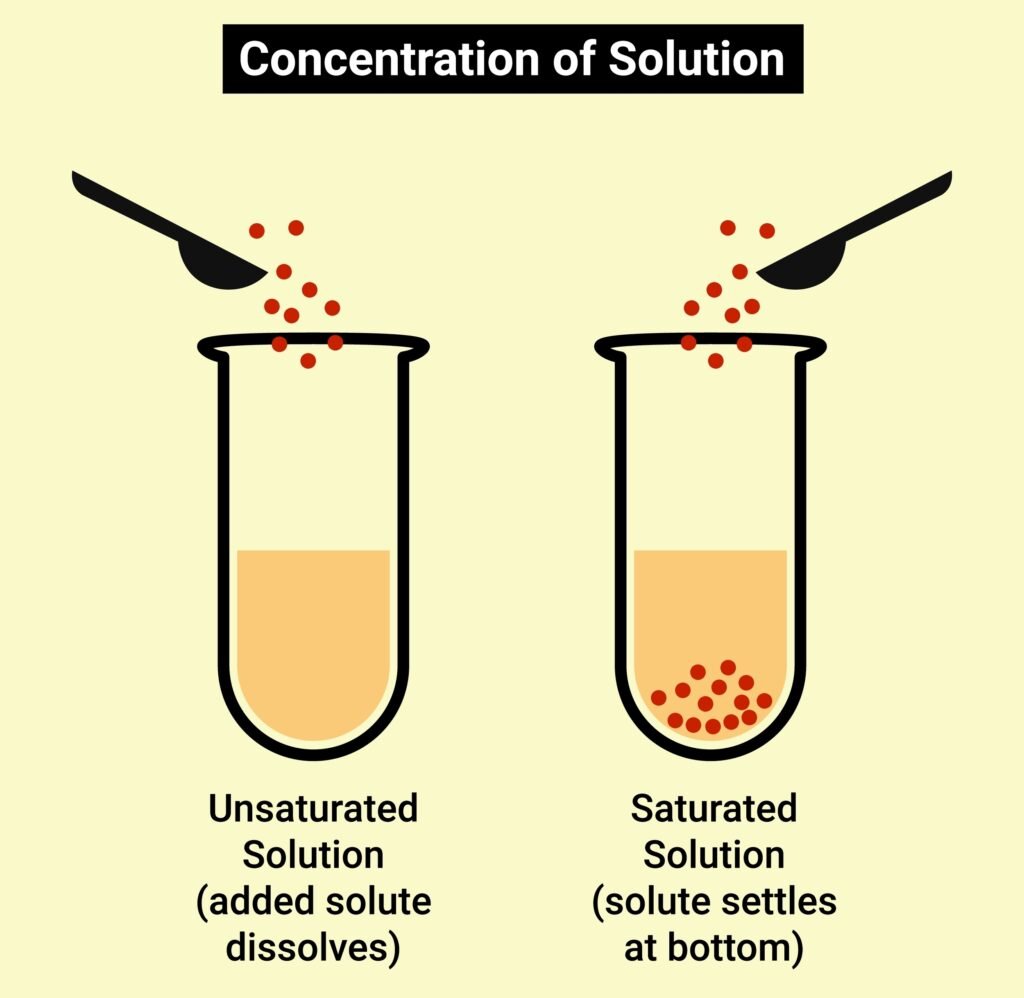
SOLUBILITY
Solubility refers to the maximum amount of a solute that can dissolve in a specific amount of solvent at a specific temperature.
Factors Affecting Solubility:
a. Nature of solute and solvent:
“Like dissolves like” – polar solutes dissolve in polar solvents.
b. Temperature:
For solids in liquids: solubility generally increases with temperature.
For gases in liquids: solubility decreases with temperature.
c. Pressure (Henry’s Law):
The solubility of a gas in a liquid is directly proportional to the pressure of the gas.
Henry’s Law: p = KH × x
Where p = partial pressure of gas, x = mole fraction in solution, KH = Henry’s constant.
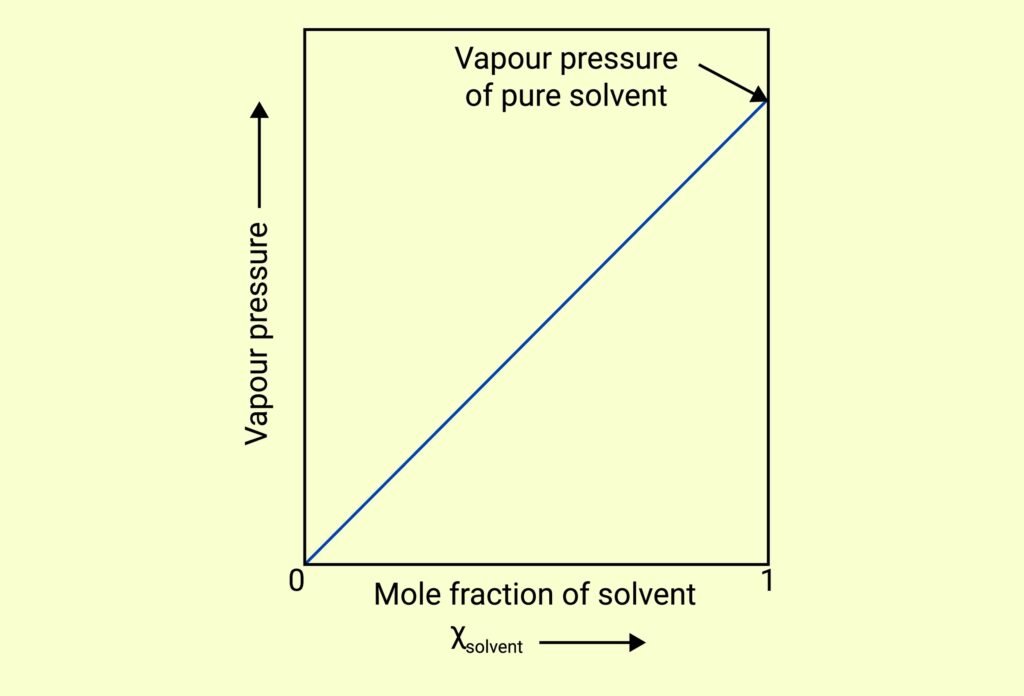
VAPOUR PRESSURE OF LIQUID SOLUTIONS
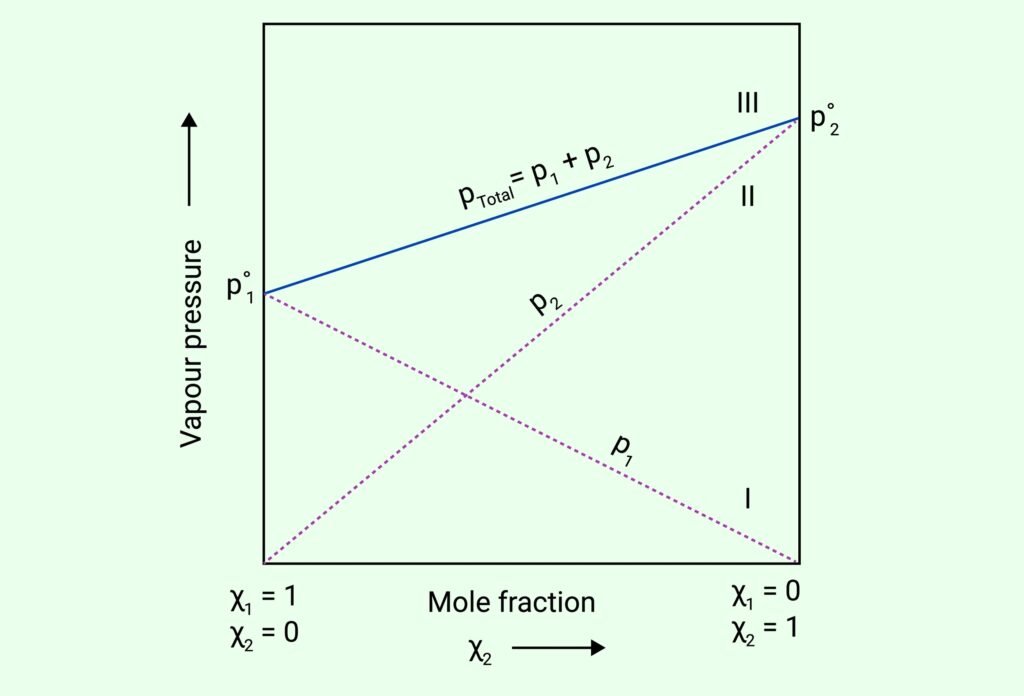
Raoult’s Law (for volatile solutes):
For a solution of two volatile liquids A and B:
pA = pA° × xA, pB = pB° × xB
Total vapour pressure:
pTotal = pA + pB = pA°xA + pB°xB
Where:
pA° and pB° are vapour pressures of pure components,
xA and xB are mole fractions.
If solute is non-volatile, only the solvent contributes to vapour pressure:
pSolution = xSolvent × p°Solvent
Ideal Solutions: Follow Raoult’s Law at all compositions; no enthalpy change or volume change.
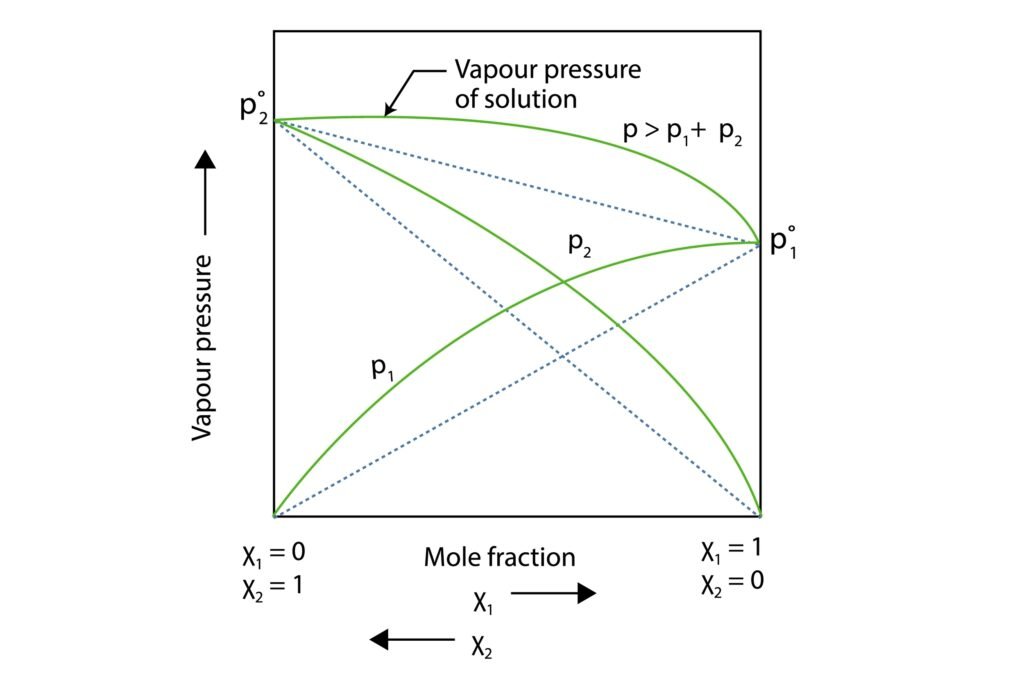
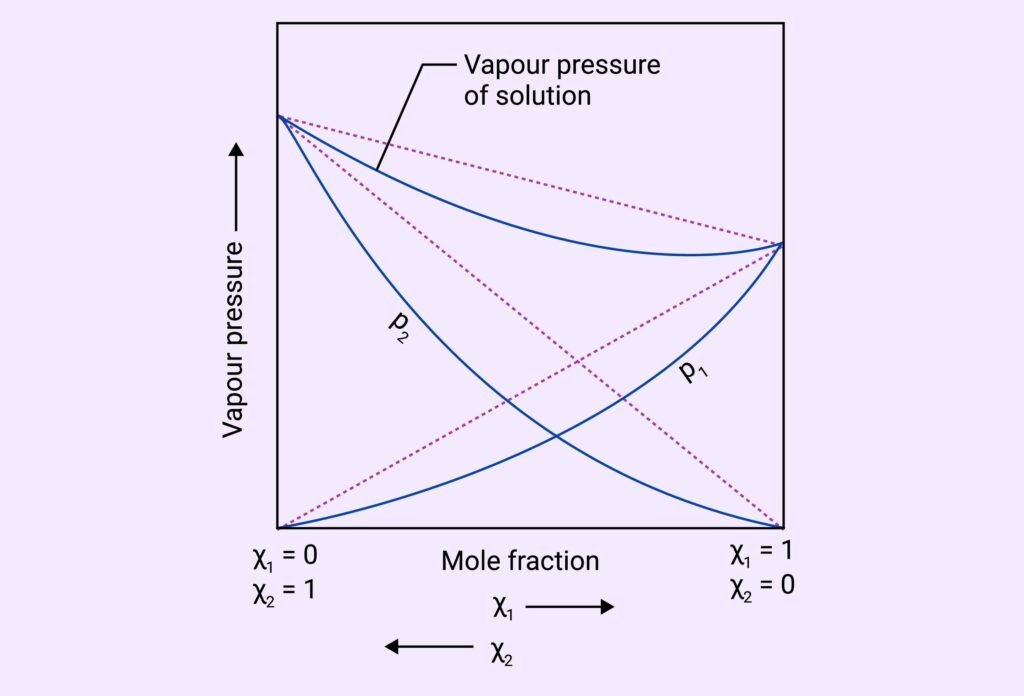
Non-Ideal Solutions: Deviate from Raoult’s Law.
Positive Deviation: vapour pressure more than predicted (weak A-B interactions)
Negative Deviation: vapour pressure less than predicted (strong A-B interactions)
COLLIGATIVE PROPERTIES
Colligative properties depend only on the number of solute particles and not their nature.
There are four types:
(a) Relative Lowering of Vapour Pressure (RLVP)
For a solution with non-volatile solute:
(p⁰ – p) / p⁰ = xSolute = n₂ / (n₁ + n₂)
If n₂ ≪ n₁, then
(p⁰ – p) / p⁰ ≈ n₂ / n₁
(b) Elevation of Boiling Point (ΔTb)
Boiling point of solution > pure solvent
ΔTb = Kb × m
Where:
Kb = ebullioscopic constant
m = molality
(c) Depression of Freezing Point (ΔTf)
Freezing point of solution < pure solvent
ΔTf = Kf × m
Where:
Kf = cryoscopic constant
m = molality
(d) Osmotic Pressure (π)
π = CRT
Where:
π = osmotic pressure
C = molar concentration
R = gas constant (0.0821 L·atm/K·mol)
T = absolute temperature
Osmosis: flow of solvent through a semipermeable membrane from dilute to concentrated solution.
Reverse Osmosis: Used in water purification (RO systems)
VAN’T HOFF FACTOR (i)
Used when the solute undergoes association or dissociation.
i = Actual number of particles in solution / Expected number of particles
i affects all colligative properties:
ΔTb, ΔTf, π, RLVP ∝ i
Examples:
NaCl → 2 ions (i ≈ 2)
BaCl₂ → 3 ions (i ≈ 3)
Glucose → no dissociation (i = 1)
Corrected formulae:
ΔTb = i × Kb × m
ΔTf = i × Kf × m
π = i × C × R × T
(p⁰ – p) / p⁰ = i × xSolute
ABNORMAL MOLECULAR MASSES
Colligative property-based molecular mass may differ due to:
Dissociation: apparent molar mass < actual
Association: apparent molar mass > actual
Corrected Molecular Mass:
Mobserved = Mtheoretical / i
PRACTICAL APPLICATIONS
Antifreeze in car radiators: Ethylene glycol lowers freezing point.
Salting roads: Salt lowers freezing point of water.
IV Fluids: Must be isotonic to blood plasma to avoid osmotic effects.
Reverse Osmosis (RO): Uses osmotic pressure to purify water.
Food preservation: Salt/sugar creates high osmotic pressure preventing microbial growth.
CONCEPTUAL CLARIFICATIONS
Difference between Molality and Molarity:
Molarity changes with temperature (volume changes), molality does not.
Ideal vs Non-Ideal Solutions:
Ideal: No volume or heat change on mixing
Non-Ideal: Show enthalpy or volume change
Why colligative properties are useful:
They help determine molecular mass of solute, detect abnormal behaviour like dissociation, and are used in biological and industrial applications.

————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
Question 1.1:
Define the term solution. How many types of solutions are formed? Write briefly about each type with an example.
Answer:
A solution is a homogeneous mixture of two or more components uniformly dispersed throughout a single phase.
Types of solutions based on physical states:
Solid in solid – Example: Brass (copper + zinc alloy).
Gas in liquid – Example: Carbonated water (CO₂ dissolved in H₂O).
Liquid in liquid – Example: Ethanol in water.
Gas in gas – Example: Air (mixture of N₂ and O₂).
Question 1.2:
Give an example of a solid solution in which the solute is a gas.
Answer:
Hydrogen gas dissolved in palladium metal (Pd–H system).
Question 1.3:
Define the following terms:
(i) Mole fraction
(ii) Molality
(iii) Molarity
(iv) Mass percentage
Answer:
(i) Mole fraction (xᵢ) = nᵢ / Σnⱼ
(ii) Molality (m) = moles of solute / mass of solvent in kg
(iii) Molarity (M) = moles of solute / volume of solution in litres
(iv) Mass % = (mass of solute / mass of solution) × 100
Question 1.4:
Concentrated nitric acid is 68% HNO₃ by mass, density 1.504 g/mL. What is its molarity?
Answer:
In 100 g solution:
HNO₃ = 68 g; Molar mass = 63 g/mol ⇒ Moles = 68/63 = 1.079 mol
Volume = 100/1.504 = 66.49 mL = 0.06649 L
Molarity = 1.079 / 0.06649 ≈ 16.23 M
Question 1.5:
A 10% w/w glucose solution has density 1.2 g/mL. Find its molality, mole fraction, and molarity.
Answer:
In 100 g solution: Glucose = 10 g ⇒ Moles = 10/180 = 0.0556 mol
Water = 90 g = 0.090 kg ⇒ Molality = 0.0556 / 0.090 = 0.618 m
Moles of water = 90/18 = 5 mol
Mole fraction of glucose = 0.0556 / (0.0556 + 5) = 0.0111
Molarity = 0.0556 / (100/1.2 ÷ 1000) = 0.0556 / 0.08333 = 0.667 M
Question 1.6:
How many mL of 0.1 M HCl are needed to react with 1 g mixture of equimolar Na₂CO₃ and NaHCO₃?
Answer:
Average molar mass = (106 + 84)/2 = 95 g/mol ⇒ Moles = 1/95 ≈ 0.0105 mol
Each salt = 0.00525 mol
Na₂CO₃ requires 2 mol HCl/mol ⇒ 0.00525×2 = 0.0105 mol
NaHCO₃ requires 1 mol HCl/mol ⇒ 0.00525×1 = 0.00525 mol
Total HCl = 0.01575 mol ⇒ Volume = 0.01575 / 0.1 = 157.5 mL
Question 1.7:
300 g of 25% solution is mixed with 400 g of 40% solution. Find mass % of final solution.
Answer:
Solute: 300×0.25 = 75 g, 400×0.40 = 160 g ⇒ Total = 235 g
Total mass = 700 g ⇒ Mass % = (235/700)×100 = 33.57%
Question 1.8:
Antifreeze: 222.6 g C₂H₆O₂ + 200 g H₂O; density = 1.072 g/mL. Find molality and molarity.
Answer:
Molar mass of C₂H₆O₂ = 62 g/mol ⇒ Moles = 222.6/62 = 3.593 mol
Molality = 3.593 / 0.200 = 17.97 m
Total volume = 422.6 / 1.072 = 394.2 mL = 0.3942 L
Molarity = 3.593 / 0.3942 ≈ 9.12 M
Question 1.9:
Water contains 15 ppm CHCl₃. Find its mass % and molality.
Answer:
15 ppm = 15 mg/kg = 1.5×10⁻³%
Molality = (15×10⁻³ g) / 119.38 g/mol = 1.256×10⁻⁴ mol/kg = 1.256×10⁻⁴ m
Question 1.10:
Discuss the role of molecular interactions in alcohol–water solution.
Answer:
Alcohol forms hydrogen bonds with water. These interactions are different from pure components and cause deviations from ideality, affecting properties like vapor pressure and boiling point.
Question 1.11:
Why do gases become less soluble in liquids at higher temperatures?
Answer:
Solubility of gases decreases with temperature because added heat allows gas molecules to escape from the solvent (based on Le Châtelier’s principle).
Question 1.12:
State Henry’s law and mention its applications.
Answer:
Henry’s law: p = k·x (pressure is proportional to mole fraction of gas in liquid).
Applications:
Carbonated drinks
Scuba diving (avoiding bends)
Industrial gas absorption
Question 1.13:
Mass of ethane = 6.56×10⁻³ g exerts 1 bar pressure. What is the pressure for 5×10⁻² g?
Answer:
Pressure ∝ mass ⇒
p = 1 × (5×10⁻² / 6.56×10⁻³) = 7.62 bar
Question 1.14:
What are positive and negative deviations from Raoult’s law? How are they related to ΔHₘᵢₓ?
Answer:
Positive deviation: weaker A–B interactions, higher vapor pressure, ΔHₘᵢₓ > 0
Negative deviation: stronger A–B interactions, lower vapor pressure, ΔHₘᵢₓ < 0
Question 1.15:
A 2% non-volatile solute solution exerts 1.004 bar vapor pressure at boiling point. Find molar mass.
Answer:
ΔT_b = 0.004 K; K_b = 0.52 ⇒ m = ΔT_b/K_b = 0.00769 mol/kg
Molality = (2/M)/0.098 ⇒ 0.00769 = (2/M)/0.098 ⇒ M ≈ 260–264 g/mol
Question 1.16:
Ideal solution: 26 g heptane (M=100) and 35 g octane (M=114); p⁰₁ = 105.2 kPa, p⁰₂ = 46.8 kPa. Find vapor pressure.
Answer:
n₁ = 0.26 mol, n₂ = 0.307 mol
x₁ = 0.458, x₂ = 0.542
p = 0.458×105.2 + 0.542×46.8 = 76.7 kPa
Question 1.17:
Vapor pressure of water = 12.3 kPa at 300 K. What is vapor pressure of 1 molal solute solution?
Answer:
x_solute = 1 / (1 + 55.5) = 0.0177
Δp = 12.3 × 0.0177 = 0.218 kPa
New vapor pressure = 12.3 – 0.218 = 12.08 kPa
Question 1.18:
Find mass of solute (M = 40 g/mol) in 114 g octane to reduce vapor pressure to 80%.
Answer:
p/p₀ = x_solvent = 0.80 ⇒ n_solute = (1/0.80 – 1) = 0.25 mol
mass = 0.25 × 40 = 10 g
Question 1.19:
Solution: 30 g solute in 90 g water gives p = 2.8 kPa; after adding 18 g water, p = 2.9 kPa.
(i) Find molar mass of solute
(ii) Find vapor pressure of pure water
Answer:
Let M = molar mass, p₀ = vapor pressure of water
First case: n_sol = 30/M, n_H₂O = 90/18 = 5 mol
p₁ = p₀ × (5 / (5 + 30/M)) = 2.8
Second case: n_H₂O = (90+18)/18 = 6 mol
p₂ = p₀ × (6 / (6 + 30/M)) = 2.9
Divide equations and solve: M ≈ 60 g/mol, p₀ ≈ 3.2 kPa
Question 1.20:
Freezing point of 5% sugar (M = 342 g/mol) is 271 K. Find freezing point of 5% glucose (M = 180 g/mol).
Answer:
m_glucose = (5/180)/0.095 = 0.293 m
ΔT_f = 1.86 × 0.293 = 0.545 K
Freezing point = 273.15 – 0.545 = 272.61 K
Question 1.21:
Two elements A and B form compounds AB₂ and AB₄. When 1 g of AB₂ and AB₄ are each dissolved in 20 g benzene, AB₂ lowers the freezing point by 2.3 K and AB₄ by 1.3 K. Kf for benzene = 5.1 K kg mol⁻¹. Calculate atomic masses of A and B.
Answer:
For AB₂:
M₁ = (5.1 × 1000 × 1) / (2.3 × 20) = 110.87 g/mol
For AB₄:
M₂ = (5.1 × 1000 × 1) / (1.3 × 20) = 196.15 g/mol
Let A = x, B = y
Then,
x + 2y = 110.87
x + 4y = 196.15
Subtracting: 2y = 85.28 → y = 42.64 g/mol
x = 110.87 − 85.28 = 25.59 g/mol
Question 1.22:
A 0.5% (w/w) aqueous KCl solution freezes at −0.24°C. Find the van’t Hoff factor (i) and degree of dissociation (α). (Kf = 1.86 K kg mol⁻¹)
Answer:
Mass of solvent = 99.5 g = 0.0995 kg
Molality = (0.5 / 74.5) / 0.0995 = 0.0675 mol/kg
i = ΔTf / (Kf × molality) = 0.24 / (1.86 × 0.0675) = 1.91
Since KCl dissociates into K⁺ and Cl⁻:
i = 1 + α ⇒ α = i − 1 = 0.91 or 91%
Question 1.23:
Solubility of H₂S in water at STP is 0.195 mol/kg. Calculate Henry’s law constant.
Answer:
Mole fraction of H₂S ≈ 0.195 / (0.195 + 55.5) ≈ 0.00351
KH = P / x = 1 / 0.00351 = 285 bar
Question 1.24:
Henry’s law constant for CO₂ in water at 298 K is 1.67 × 10⁸ Pa. How much CO₂ is dissolved in 500 mL soda water under 2.5 atm CO₂ pressure?
Answer:
P = 2.5 atm = 2.5 × 101325 = 253312.5 Pa
x = 253312.5 / 1.67 × 10⁸ = 1.517 × 10⁻³
Moles of water = 500 / 18 = 27.78 mol
Moles of CO₂ = 1.517 × 10⁻³ × 27.78 = 0.0421 mol
Mass = 0.0421 × 44 = 1.85 g
Question 1.25:
P°A = 450 mmHg, P°B = 700 mmHg, total pressure = 600 mmHg. Find liquid and vapour phase compositions.
Answer:
600 = xA × 450 + (1 − xA) × 700
Solving: xA = 0.4, xB = 0.6
yA = (xA × P°A) / Ptotal = (0.4 × 450) / 600 = 0.3
yB = 1 − yA = 0.7
Question 1.26:
Vapour pressure of water at 298 K is 23.8 mmHg. 50 g urea in 850 g water. Find vapour pressure of solution and relative lowering.
Answer:
Moles urea = 50 / 60 = 0.833
Moles water = 850 / 18 = 47.22
xH₂O = 47.22 / (47.22 + 0.833) = 0.9827
Vapour pressure = 0.9827 × 23.8 = 23.39 mmHg
Relative lowering = (23.8 − 23.39) / 23.8 = 0.0173
Question 1.27:
Water boils at 99.63°C at 750 mmHg. Find mass of sucrose to be added to 500 g water to raise b.p. to 100°C. (Kb = 0.52 K kg mol⁻¹)
Answer:
ΔTb = 0.37 K
Molality = 0.37 / 0.52 = 0.712 mol/kg
Moles = 0.712 × 0.5 = 0.356
Mass = 0.356 × 342 = 121.75 g
Question 1.28:
Find mass of ascorbic acid (M = 176 g/mol) to dissolve in 75 g acetic acid to lower melting point by 1.5°C. (Kf = 3.9)
Answer:
Molality = 1.5 / 3.9 = 0.385
Moles = 0.385 × 0.075 = 0.0289
Mass = 0.0289 × 176 = 5.08 g
Question 1.29:
4 g MgSO₄ (M = 120) in 100 g water. Calculate boiling point. Assume complete ionisation. (Kb = 0.52)
Answer:
Molality = (4 / 120) / 0.1 = 0.333
i = 2 ⇒ ΔTb = 2 × 0.52 × 0.333 = 0.346
Boiling point = 100 + 0.346 = 100.35°C
Question 1.30:
1.75 g Na₂SO₄ in 50 g water. Calculate freezing point. (Kf = 1.86)
Answer:
Molality = (1.75 / 142) / 0.05 = 0.246
i = 3 ⇒ ΔTf = 3 × 1.86 × 0.246 = 1.374
Freezing point = 0 − 1.374 = −1.374°C
Question 1.31:
How much CaCl₂ (i = 2.47) in 2.5 L water to get π = 0.75 atm at 27°C?
Answer:
T = 300 K, R = 0.0821
0.75 = 2.47 × M × 0.0821 × 300
M = 0.0123 mol/L
Moles = 0.0123 × 2.5 = 0.0308
Mass = 0.0308 × 111 = 3.42 g
Question 1.32:
25 mg K₂SO₄ in 2 L water at 25°C. Find osmotic pressure.
Answer:
Moles = 0.025 / 174 = 1.437 × 10⁻⁴
Molarity = 7.18 × 10⁻⁵
i = 3
π = 3 × 7.18 × 10⁻⁵ × 0.0821 × 298 = 5.27 × 10⁻³ atm
Question 1.33:
Find molarity of sugar solution with same osmotic pressure as 0.1 M NaCl at 300 K.
Answer:
i(NaCl) = 2 ⇒ effective concentration = 0.2 M
Sugar (i = 1): molarity = 0.2 M
Question 1.34:
1 g polymer (M = 185000) in 450 mL water at 37°C. Find osmotic pressure in pascals.
Answer:
Moles = 1 / 185000 = 5.41 × 10⁻⁶
Molarity = 5.41 × 10⁻⁶ / 0.45 = 1.20 × 10⁻⁵
π = M × R × T = 1.20 × 10⁻⁵ × 8.314 × 310 = 0.031 Pa
Question 1.35:
30 g solute in 90 g water: p = 2.8 kPa. After 18 g water added: p = 2.9 kPa. Find (i) molar mass (ii) vapour pressure of water.
Answer:
Initial: n_sol = 30/M, n_water = 90/18 = 5 mol
Final: water = 108 g → 6 mol
Using Raoult’s law and ratio of vapour pressures:
M ≈ 60 g/mol, P₀ ≈ 3.4 kPa
Question 1.36:
5% cane sugar lowers freezing point to 271 K. What is freezing point of 5% glucose solution?
Answer:
ΔTf = 2.15 K, m (sugar) = 0.154
Kf = 2.15 / 0.154 ≈ 14
Glucose: m = (5 / 180) / 0.095 = 0.292
ΔTf = 14 × 0.292 = 4.09
Freezing point = 273.15 − 4.09 = 269.06 K
Question 1.37:
Mix 480 mL of 1.5 M and 520 mL of 1.2 M solution. Find final molarity.
Answer:
Moles = (1.5 × 0.48) + (1.2 × 0.52) = 1.344
Volume = 1.0 L ⇒ Molarity = 1.344 M
Question 1.38:
A 2% solute solution exerts 1.004 bar at boiling point. Find molar mass.
Answer:
Relative lowering = (1.013 − 1.004)/1.013 = 0.00888
n_water = 98 / 18 = 5.44
(2 / M) / 5.44 = 0.00888 ⇒ M = 41.4 g/mol
Question 1.39:
Heptane: 26 g (M = 100), Octane: 35 g (M = 114), P₀ = 105.2 and 46.8 kPa. Find vapour pressure.
Answer:
n_heptane = 0.26, n_octane = 0.307
x_heptane = 0.458, x_octane = 0.542
P_total = (0.458 × 105.2) + (0.542 × 46.8) = 76.7 kPa
Question 1.40:
7 g protein in 100 mL has π = 25 mmHg at 37°C. Find molar mass.
Answer:
π = 25 / 760 = 0.0329 atm
T = 310 K
Molarity = π / (R × T) = 1.29 × 10⁻³ mol/L
Molar mass = 7 / (1.29 × 10⁻³ × 0.1) = 54264 g/mol
Question 1.41:
25 mg K₂SO₄ in 2 L at 25°C. Find osmotic pressure.
Answer:
Moles = 0.025 / 174 = 1.437 × 10⁻⁴
Molarity = 7.18 × 10⁻⁵
π = 3 × 7.18 × 10⁻⁵ × 0.0821 × 298 = 5.27 × 10⁻³ atm
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
SECTION A (1 MARK EACH)
Answer the following questions briefly.
Q.1 Define molality.
Answer: Molality is the number of moles of solute dissolved per kilogram of solvent.
Q.2 What is the van’t Hoff factor for NaCl in aqueous solution?
Answer: 2
Q.3 What type of deviation from Raoult’s law is shown by a solution formed by mixing acetone and chloroform?
Answer: Negative deviation
Q.4 State Henry’s law.
Answer: Henry’s law states that the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas.
Q.5 Which will have a higher boiling point: 1 molal NaCl or 1 molal glucose solution? Why?
Answer: 1 molal NaCl, because it ionizes to give more particles, causing higher elevation in boiling point.
Q.6 What happens to the solubility of gases in liquids with increase in temperature?
Answer: It decreases.
Q.7 What is an ideal solution?
Answer: A solution that obeys Raoult’s law at all concentrations and temperatures.
Q.8 Which colligative property is best suited to determine the molar mass of a solute?
Answer: Depression in freezing point
Q.9 Give an example of a solution showing positive deviation from Raoult’s law.
Answer: Ethanol and acetone
Q.10 Write the relationship between mole fraction of a component and partial pressure in a binary solution.
Answer: pA = xA × pA⁰
SECTION B (2 MARKS EACH)
Answer the following in about 30–40 words.
Q.11 Explain why elevation of boiling point is considered a colligative property.
Answer: It depends only on the number of solute particles, not their nature. More particles lower vapor pressure and raise boiling point.
QWhy do aquatic species face difficulty in breathing in warm water?
Answer: The solubility of oxygen gas decreases with rise in temperature, so less oxygen is available in warm water.
Q.12 Define osmotic pressure. Write the formula.
Answer: Osmotic pressure is the minimum pressure required to stop osmosis.
π = C × R × T, where C = molarity, R = gas constant, T = temperature.
Q.13 How does adding a non-volatile solute to a solvent affect vapor pressure?
Answer: It lowers the vapor pressure due to reduced number of solvent molecules at the surface.
Q.14 What is meant by abnormal molar mass? How is it caused?
Answer: When observed molar mass deviates from theoretical value due to association or dissociation of solute in solution.
SECTION C (3 MARKS EACH)
Answer the following in about 50–60 words.
Q.15 A solution contains 5 g of urea (molar mass = 60 g/mol) in 100 g of water. Calculate the lowering of vapor pressure if the vapor pressure of pure water is 32 mmHg.
Answer:
Moles of urea = 5/60 = 0.083 mol
Moles of water = 100/18 = 5.56 mol
Mole fraction of solute = 0.083 / (0.083 + 5.56) ≈ 0.0147
Lowering of vapor pressure = 32 × 0.0147 ≈ 0.47 mmHg
Q.16 What is Raoult’s law for a binary liquid solution?
Answer:
Raoult’s law states that the partial vapor pressure of each component is directly proportional to its mole fraction:
pA = xA × pA⁰,
pB = xB × pB⁰,
Total vapor pressure = pA + pB
Q.17 State two differences between ideal and non-ideal solutions with examples.
Answer:
Ideal: Obey Raoult’s law (e.g., benzene + toluene)
Non-ideal: Deviate from Raoult’s law (e.g., acetone + chloroform)
Ideal solutions show no enthalpy change; non-ideal ones do.
Q.18 Explain the effect of pressure and temperature on solubility of gases in liquids.
Answer:
Pressure: Solubility increases with pressure (Henry’s law).
Temperature: Solubility decreases with temperature due to increased kinetic energy.
Q.19 Define colligative properties. Name four such properties.
Answer:
Colligative properties depend on the number of solute particles, not their type.
Examples:
(i) Relative lowering of vapor pressure
(ii) Elevation of boiling point
(iii) Depression of freezing point
(iv) Osmotic pressure
SECTION D (5 MARKS EACH)
Answer the following in about 80–100 words.
Q.20 Define Henry’s law. Derive the expression for calculating solubility of a gas using this law. Mention its applications.
Answer:
Henry’s law states that the partial pressure of a gas in a liquid is directly proportional to its mole fraction:
p = KH × x
Where:
p = partial pressure of gas,
x = mole fraction of gas,
KH = Henry’s law constant
Applications:
Bottling of soft drinks and soda
Scuba diving: to prevent nitrogen narcosis
Preservation of packaged food using nitrogen
Q.21 Derive the relation between elevation in boiling point and molar mass of solute.
Answer:
ΔTb = Kb × m
m = (w2 × 1000) / (M2 × w1)
Substitute in the first equation:
ΔTb = (Kb × w2 × 1000) / (M2 × w1)
Rearranged:
M2 = (Kb × 1000 × w2) / (ΔTb × w1)
Where:
w2 = mass of solute, w1 = mass of solvent,
M2 = molar mass of solute, Kb = molal elevation constant
Q.22 Explain with a diagram the reason for abnormal molar mass in case of electrolytes.
Answer:
Electrolytes dissociate into ions, increasing the number of solute particles.
This alters colligative properties, giving abnormal molar mass.
van’t Hoff factor, i = observed colligative property / expected
Corrected formula:
π = iCRT,
ΔTf = iKf × m
This explains why observed molar mass is lower than calculated.
SECTION E (CASE STUDY: 5 MARKS)
Read the paragraph and answer the questions.
Q.23 A solution is prepared by dissolving 6.0 g of urea (molar mass = 60 g/mol) in 100 g of water. The vapor pressure of pure water is 23.8 mmHg. Calculate the relative lowering of vapor pressure and hence the vapor pressure of the solution.
(2 MARKS) What is the mole fraction of urea?
Answer:
Moles of urea = 6/60 = 0.1 mol
Moles of water = 100/18 = 5.56 mol
Mole fraction of urea = 0.1 / (0.1 + 5.56) ≈ 0.0177
(1 MARK)Q.24 What is the relative lowering of vapor pressure?
Answer:
Relative lowering = mole fraction of solute = 0.0177
(1 MARK)Q.25 Calculate vapor pressure of solution.
Answer:
Psolution = 23.8 × (1 – 0.0177) = 23.38 mmHg
(1 MARK)Q.26 Name the colligative property used here.
Answer:
Relative lowering of vapor pressure
————————————————————————————————————————————————————————————————————————————
