Class 12 : Chemistry (English) – Chapter 5: Coordination Compounds
EXPLANATION & SUMMARY
✨ Introduction
🔵 Coordination compounds are fascinating chemical species where a central metal atom/ion is surrounded by neutral molecules or anions called ligands, held together via coordinate covalent bonds.
🟢 These compounds are important in biological systems (hemoglobin, chlorophyll), industrial catalysis (Wilkinson’s catalyst, Ziegler–Natta catalyst), analytical chemistry (qualitative analysis with complex ions), and in extraction metallurgy.
🌿 Key Terms
💡 Coordination entity: Central atom/ion + ligands (e.g., [Cu(NH₃)₄]²⁺).
✏ Coordination number (CN): Number of ligand donor atoms directly attached to the central atom.
🟠 Ligands: Species donating electron pairs.
Monodentate: Cl⁻, NH₃.
Bidentate: C₂O₄²⁻, en (ethane-1,2-diamine).
Polydentate: EDTA⁴⁻.
🔴 Chelation: Formation of ring structures by multidentate ligands.
⚡ Oxidation number: Charge of central atom assuming all ligands removed in neutral form.
📌 Coordination polyhedra: Common ones: octahedral, tetrahedral, square planar.
🧪 Nomenclature Rules (IUPAC)
Write cation before anion.
Ligands named alphabetically before metal.
Neutral ligands use molecule name, exceptions: H₂O → aqua, NH₃ → ammine, CO → carbonyl.
Anionic ligands end with “-o” (Cl⁻ → chloro, CN⁻ → cyano).
Oxidation state of metal shown in Roman numerals.
📌 Example: [Co(NH₃)₆]Cl₃ → hexaamminecobalt(III) chloride.
🌟 Bonding Theories
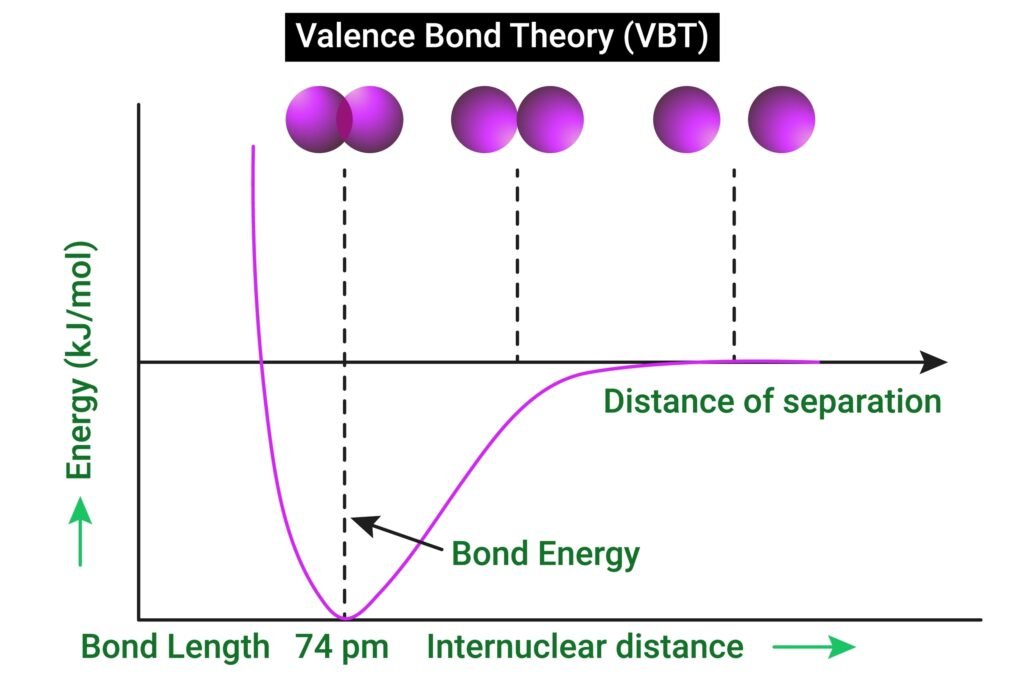
Valence Bond Theory (VBT)
🟦 Explains hybridisation of metal orbitals to accommodate ligands.
Octahedral: d²sp³ or sp³d²
Tetrahedral: sp³
Square planar: dsp²
📌 Example: [Ni(CN)₄]²⁻ → dsp² hybridisation (low-spin, diamagnetic).
Crystal Field Theory (CFT)
💡 Metal–ligand interaction is purely electrostatic.
🟩 In an octahedral field, d-orbitals split into:
Lower t₂g (dxy, dxz, dyz)
Higher e_g (dx²–y², dz²)
🟨 Splitting energy = Δ₀.
📌 If Δ₀ > pairing energy → low-spin; If Δ₀ < pairing energy → high-spin.
Example: [Fe(CN)₆]³⁻ → low-spin (t₂g⁵ e_g⁰).
Example: [FeF₆]³⁻ → high-spin (t₂g³ e_g²).
Molecular Orbital Theory (MOT)
🟠 Explains bonding more accurately (σ and π interactions).
Predicts colour, magnetism, stability of complexes.
🎨 Colour and Magnetism
🔵 Colour: Due to d–d transitions when light excites an electron between split d-orbitals.
🟢 Paramagnetism: From unpaired d-electrons (μ = √(n(n+2)) BM).
🔴 Diamagnetism: All electrons paired.
⚗ Isomerism in Coordination Compounds
Structural isomerism
Ionisation, Solvate, Linkage, Coordination, Coordination position.
Stereoisomerism
Geometrical: cis–trans (e.g., [Pt(NH₃)₂Cl₂]).
Optical: Non-superimposable mirror images (e.g., [Co(en)₃]³⁺).
🌍 Applications
🟦 Biological: Hemoglobin (Fe²⁺), Chlorophyll (Mg²⁺), Vitamin B₁₂ (Co³⁺).
🟩 Medicinal: Cisplatin (anticancer drug).
🟨 Industrial: Catalysts → Wilkinson’s catalyst, Ziegler–Natta catalyst.
🟧 Analytical: Detection of metal ions in qualitative analysis.
📝 Summary
Coordination compounds consist of a central metal atom/ion surrounded by ligands.
Ligands classified as monodentate, bidentate, polydentate; chelation increases stability.
Nomenclature follows IUPAC rules; oxidation state in Roman numerals.
Bonding explained by VBT (hybridisation), CFT (d-orbital splitting), MOT (σ, π bonding).
Colour arises from d–d transitions; magnetism from unpaired electrons.
Isomerism: structural (ionisation, linkage, solvate, etc.) and stereoisomerism (geometrical, optical).
Applications: biological (hemoglobin, chlorophyll), medicinal (cisplatin), industrial (catalysts), analytical (qualitative analysis).
f-block contraction affects d-block similarities.
🎯 Quick Recap
🟢 Ligands donate electron pairs → form coordinate bonds.
🔵 CFT explains colour (d–d transitions).
🟠 VBT explains hybridisation.
🔴 Isomerism: structural + stereoisomerism.
⚡ Uses: medicines, catalysts, bio-systems.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
Question 5.1
Explain the bonding in coordination compounds in terms of Werner’s postulates.
Answer
🔵 Werner’s Postulates:
🟢 Metals exhibit two types of valencies:
Primary valency → Ionisable (oxidation state).
Secondary valency → Non-ionisable, fixed in space (coordination number).
🟠 Secondary valencies are satisfied by ligands and determine geometry.
🔴 Compounds with same metal but different secondary valency show different properties.
📌 Example: [CoCl₃·6NH₃] → Ionises as [Co(NH₃)₆]Cl₃ → Co³⁺ primary valency satisfied, CN=6 secondary valency.
Question 5.2
FeSO₄ solution mixed with (NH₄)₂SO₄ solution in 1:1 molar ratio gives the test of Fe²⁺ ion but CuSO₄ solution mixed with aqueous ammonia in 1:4 molar ratio does not give the test of Cu²⁺ ion. Explain why.
Answer
🔵 In FeSO₄ + (NH₄)₂SO₄ → Double salt (Mohr’s salt, FeSO₄·(NH₄)₂SO₄·6H₂O) forms.
🟢 Double salts ionise completely in solution → Fe²⁺ ions free → Test positive.
🟠 In CuSO₄ + 4NH₃ → [Cu(NH₃)₄]²⁺ (complex ion) forms.
🔴 Complex ions do not dissociate → Cu²⁺ not available → No test.
Question 5.3
Explain with two examples each of the following: coordination entity, ligand, coordination number, coordination polyhedron, homoleptic and heteroleptic.
Answer
🔵 Coordination entity: Central atom + ligands. Example: [Co(NH₃)₆]³⁺, [CuCl₄]²⁻.
🟢 Ligands: Donors of electron pair. Example: NH₃, Cl⁻.
🟠 Coordination number: Number of donor atoms bonded. Example: [Co(NH₃)₆]³⁺ → CN=6; [Ni(CO)₄] → CN=4.
🔴 Coordination polyhedron: Spatial arrangement. Example: Octahedral in [Fe(CN)₆]³⁻, Square planar in [PtCl₄]²⁻.
⚡ Homoleptic: Same type ligands only, [Co(NH₃)₆]³⁺.
📌 Heteroleptic: Different ligands, [CoCl₂(en)₂]⁺.
Question 5.4
What is meant by unidentate, bidentate and ambidentate ligands? Give two examples for each.
Answer
🔵 Unidentate: One donor atom. Examples: Cl⁻, NH₃.
🟢 Bidentate: Two donor atoms. Examples: Ethylenediamine (en), C₂O₄²⁻.
🟠 Ambidentate: Can donate through two atoms but one at a time. Examples: NO₂⁻ (N or O donor), SCN⁻ (S or N donor).
Question 5.5
Specify the oxidation numbers of the metals in the following coordination entities:
(i) [Co(H₂O)(CN)(en)₂]²⁺
(ii) [PtCl₂]²⁻
(iii) [Cr(NH₃)₆]Cl₃
(iv) [CoBr₂(en)₂]⁺
(v) K₃[Fe(CN)₆]
Answer
✏ Step-by-step:
(i) [Co(H₂O)(CN)(en)₂]²⁺ → Let Co = x
x + 0 + (–1) + 0 = +2 → x = +3 → Oxidation = +3
(ii) [PtCl₂]²⁻ → x + 2(–1) = –2 → x = 0 → Oxidation = 0
(iii) [Cr(NH₃)₆]Cl₃ → Complex cation charge = +3 → Cr oxidation = +3
(iv) [CoBr₂(en)₂]⁺ → x + 2(–1) = +1 → x = +3
(v) K₃[Fe(CN)₆] → [Fe(CN)₆]³⁻ → x + 6(–1) = –3 → x = +3
✅ Final: Co(+3), Pt(0), Cr(+3), Co(+3), Fe(+3).
Question 5.6
Using IUPAC norms write the formulas for the following:
(i) Tetrahydroxidozincate(II)
(ii) Potassium tetrachloridopalladate(II)
(iii) Diamminedichloridoplatinum(II)
(iv) Potassium tetracyanidoniccolate(II)
(v) Pentaamminenitrito-O-cobalt(III)
(vi) Hexaamminecobalt(III) sulphate
(vii) Potassium trioxalatochromate(III)
(viii) Hexaammineplatinum(IV)
(ix) Potassium hexacyanidoferrate(III)
(x) Tetrabromidocuprate(II)
Answer
(i) [Zn(OH)₄]²⁻
(ii) K₂[PdCl₄]
(iii) [Pt(NH₃)₂Cl₂]
(iv) K₂[Ni(CN)₄]
(v) [Co(NH₃)₅(NO₂)]²⁺
(vi) [Co(NH₃)₆]₂(SO₄)₃
(vii) K₃[Cr(C₂O₄)₃]
(viii) [Pt(NH₃)₆]⁴⁺
(ix) K₃[Fe(CN)₆]
(x) [CuBr₄]²⁻
Question 5.7
Using IUPAC norms write the systematic names of the following:
(i) [Co(NH₃)₆]Cl₃
(ii) [Pt(NH₃)₂Cl(NO₂)]Cl
(iii) [Ti(H₂O)₆]³⁺
(iv) [Co(NH₃)₄Cl(NO₂)]Cl
(v) [Mn(H₂O)₆]³⁺
(vi) [Co(en)₃]³⁺
(vii) [NiCl₄]²⁻
(viii) [Ni(CO)₄]
(ix) [Cr(NH₃)₆]Cl₃
Answer
(i) Hexaamminecobalt(III) chloride
(ii) Diamminemonochloridonitrito-N-platinum(II) chloride
(iii) Hexaaquatitanium(III) ion
(iv) Tetraamminechloridonitrito-N-cobalt(III) chloride
(v) Hexaaquamanganese(III) ion
(vi) Tris(ethane-1,2-diamine)cobalt(III) ion
(vii) Tetrachloridonickelate(II)
(viii) Tetracarbonylnickel(0)
(ix) Hexaamminechromium(III) chloride
Question 5.8
List various types of isomerism possible for coordination compounds, giving an example of each.
Answer
🟦 Structural isomerism
Ionisation: [Co(NH₃)₅Br]SO₄ vs [Co(NH₃)₅SO₄]Br
Solvate: [Cr(H₂O)₆]Cl₃ vs [Cr(H₂O)₅Cl]Cl₂·H₂O
Linkage: [Co(NH₃)₅(NO₂)]Cl₂ vs [Co(NH₃)₅(ONO)]Cl₂
Coordination: [Cr(NH₃)₆][Co(CN)₆] vs [Co(NH₃)₆][Cr(CN)₆]
🟩 Stereoisomerism
Geometrical: cis-[Pt(NH₃)₂Cl₂] vs trans-[Pt(NH₃)₂Cl₂]
Optical: cis-[Co(en)₂Cl₂]⁺ enantiomers
Question 5.9
How many geometrical isomers are possible in the following coordination entities?
(i) [Cr(C₂O₄)₃]³⁻
(ii) [Co(NH₃)₃Cl₃]
Answer
(i) [Cr(C₂O₄)₃]³⁻ → Chelating bidentate ligands only → No geometrical isomerism.
(ii) [Co(NH₃)₃Cl₃] → Facial (fac) and meridional (mer) → 2 isomers possible.
Question 5.10
Draw the structures of optical isomers of:
(i) [Cr(C₂O₄)₃]³⁻
(ii) [PtCl₂(en)₂]²⁺
(iii) [Cr(NH₃)₂Cl₂(en)]⁺
Answer
🔵 Optical isomers (non-superimposable mirror images):
(i) [Cr(C₂O₄)₃]³⁻ → Δ and Λ forms.
(ii) [PtCl₂(en)₂]²⁺ → cis form only gives optical isomers.
(iii) [Cr(NH₃)₂Cl₂(en)]⁺ → cis form gives optical isomers.
Question 5.11
Draw all the isomers (geometrical and optical) of:
(i) [CoCl₂(en)₂]⁺
(ii) [Co(NH₃)₄Cl₂]⁺
(iii) [Co(NH₃)₂Cl₂(en)]⁺
Answer
🔵 (i) [CoCl₂(en)₂]⁺
Geometrical: cis, trans.
Optical: Only cis form gives optical isomers (Δ and Λ).
🟢 (ii) [Co(NH₃)₄Cl₂]⁺
Geometrical: cis and trans.
Optical: None.
🟠 (iii) [Co(NH₃)₂Cl₂(en)]⁺
Geometrical: cis and trans.
Optical: cis form shows optical isomerism.
Question 5.12
Write all the geometrical isomers of [Pt(NH₃)₂BrCl(NO₂)] and state how many of them exhibit optical isomerism.
Answer
🔵 Square planar [Pt(NH₃)₂BrCl(NO₂)] can have three geometrical isomers:
cis–BrCl
cis–BrNO₂
cis–ClNO₂
🟢 Optical isomerism: None, because square planar complexes generally do not show chirality unless all four ligands are different.
Question 5.13
Aqueous copper sulphate solution (blue in colour) gives:
(i) A green precipitate with aqueous potassium fluoride.
(ii) A bright green solution with aqueous potassium chloride.
Explain these experimental results.
Answer
💡 Stepwise:
In CuSO₄ solution → [Cu(H₂O)₆]²⁺ (blue).
With KF → Formation of [CuF₆]⁴⁻ (green precipitate due to partial substitution of H₂O).
With KCl → Formation of [CuCl₄]²⁻ (bright green solution).
🎯 Colour change arises from ligand substitution altering d–d transitions.
Question 5.14
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H₂S(g) is passed through this solution?
Answer
🔵 Excess KCN + CuSO₄ → [Cu(CN)₄]³⁻ forms (stable complex).
🟢 In this complex, free Cu²⁺ ions are absent.
🟠 Hence H₂S cannot precipitate CuS because Cu²⁺ required for precipitation is not available.
Question 5.15
Discuss the nature of bonding in the following coordination entities on the basis of valence bond theory:
(i) [Fe(CN)₆]⁴⁻
(ii) [FeF₆]³⁻
(iii) [Co(C₂O₄)₃]³⁻
Answer
🔵 (i) [Fe(CN)₆]⁴⁻
Fe²⁺ → d⁶.
CN⁻ = strong field ligand → pairing occurs.
Hybridisation: d²sp³ (inner orbital complex).
Geometry: Octahedral, low spin, diamagnetic.
🟢 (ii) [FeF₆]³⁻
Fe³⁺ → d⁵.
F⁻ = weak field ligand → no pairing.
Hybridisation: sp³d² (outer orbital complex).
Geometry: Octahedral, high spin, paramagnetic.
🟠 (iii) [Co(C₂O₄)₃]³⁻
Co³⁺ → d⁶.
Oxalate (C₂O₄²⁻) = strong field bidentate ligand.
Pairing occurs → d²sp³ hybridisation.
Octahedral, low spin, paramagnetic (some unpaired electrons remain).
Question 5.16
Draw a figure to show the splitting of d orbitals in an octahedral crystal field.
Answer
🟦 In an octahedral field:
d orbitals split into:
t₂g set (lower energy): dxy, dxz, dyz.
e_g set (higher energy): dx²–y², dz².
Energy gap = Δ₀ (octahedral splitting energy).
📌 Diagram (to be drawn by student in exam):
t₂g (lower, 3 orbitals) → gap Δ₀ → e_g (higher, 2 orbitals).
Question 5.17
What is spectrochemical series? Explain the difference between a weak field ligand and a strong field ligand.
Answer
🔵 Spectrochemical series: Arrangement of ligands in order of increasing field strength (Δ splitting power).
💡 Order: I⁻ < Br⁻ < Cl⁻ < F⁻ < OH⁻ < H₂O < NH₃ < en < CN⁻ < CO.
🟢 Weak field ligands:
Small Δ, cause high-spin complexes.
Example: F⁻, Cl⁻.
🟠 Strong field ligands:
Large Δ, cause low-spin complexes.
Example: CN⁻, CO.
Question 5.18
What is crystal field splitting energy? How does the magnitude of Δ₀ decide the actual configuration of d orbitals in a coordination entity?
Answer
🔵 Crystal Field Splitting Energy (Δ₀): Difference in energy between t₂g and e_g orbitals in an octahedral field.
🟢 If Δ₀ < pairing energy → electrons remain unpaired → high-spin complex. 🟠 If Δ₀ > pairing energy → electrons pair up in t₂g before occupying e_g → low-spin complex.
Question 5.19
[Cr(NH₃)₆]³⁺ is paramagnetic while [Ni(CN)₄]²⁻ is diamagnetic. Explain why.
Answer
🔵 [Cr(NH₃)₆]³⁺ → Cr³⁺ (d³), NH₃ (weak field ligand) → No pairing → 3 unpaired electrons → Paramagnetic.
🟢 [Ni(CN)₄]²⁻ → Ni²⁺ (d⁸), CN⁻ (strong field ligand) → Pairing occurs → dsp² hybridisation → 0 unpaired electrons → Diamagnetic.
Question 5.20
A solution of [Ni(H₂O)₆]²⁺ is green but a solution of [Ni(CN)₄]²⁻ is colourless. Explain.
Answer
🔵 [Ni(H₂O)₆]²⁺ → Weak field ligand → Δ small → d–d transition possible → Colour observed (green).
🟢 [Ni(CN)₄]²⁻ → Strong field ligand → Large Δ → Electronic transitions not in visible region → Appears colourless.
Question 5.21
[Fe(CN)₆]⁴⁻ and [Fe(H₂O)₆]²⁺ are of different colours in dilute solutions. Why?
Answer
🔵 Colour depends on d–d transitions.
🟢 In [Fe(CN)₆]⁴⁻ → CN⁻ (strong field ligand) → Large Δ → Transition corresponds to one wavelength.
🟠 In [Fe(H₂O)₆]²⁺ → H₂O (weaker ligand) → Smaller Δ → Different transition wavelength.
🎯 Hence colours differ due to ligand field strength altering d-orbital splitting.
Question 5.22
Discuss the nature of bonding in metal carbonyls.
Answer
🔵 Bonding explained by synergic effect:
🟢 CO donates lone pair of electrons from C atom → σ-bond to metal.
🟠 Back-donation: Metal d-orbitals → π* antibonding orbital of CO → π-backbond.
⚡ This dual interaction strengthens M–C bond but weakens C–O bond.
Question 5.23
Give the oxidation state, d orbital occupation and coordination number of the central metal ion in the following complexes:
(i) K₃[Co(C₂O₄)₃]
(ii) [Ni(NH₃)₆]Cl₂
(iii) [CoF₆]³⁻
(iv) [Mn(H₂O)₆]SO₄
(v) cis-[CrCl₂(en)₂]Cl
Answer
(i) K₃[Co(C₂O₄)₃] → Co³⁺, d⁶, CN=6.
(ii) [Ni(NH₃)₆]Cl₂ → Ni²⁺, d⁸, CN=6.
(iii) [CoF₆]³⁻ → Co³⁺, d⁶, CN=6.
(iv) [Mn(H₂O)₆]SO₄ → Mn²⁺, d⁵, CN=6.
(v) cis-[CrCl₂(en)₂]Cl → Cr³⁺, d³, CN=6.
Question 5.24
Write down the IUPAC name, oxidation state, electronic configuration, and coordination number of the following complexes. Also give stereochemistry and magnetic moment:
(i) K₃[Cr(C₂O₄)₃]·3H₂O
(ii) [CrCl₃(py)₃]
(iii) K₄[Mn(CN)₆]
(iv) [Co(NH₃)₃Cl₃]
(v) Cs[FeCl₄]
Answer
(i) Potassium trioxalatochromate(III) trihydrate → Cr³⁺, d³, CN=6, Octahedral, paramagnetic (3 unpaired).
(ii) Tris(pyridine)trichloridochromium(III) → Cr³⁺, d³, CN=6, Octahedral, paramagnetic.
(iii) Potassium hexacyanomanganate(II) → Mn²⁺, d⁵, CN=6, Octahedral, paramagnetic.
(iv) Triamminetrichloridocobalt(III) → Co³⁺, d⁶, CN=6, Octahedral, paramagnetic/diamagnetic depending on spin.
(v) Caesium tetrachloridoferrate(III) → Fe³⁺, d⁵, CN=4, Tetrahedral, paramagnetic.
Question 5.25
Explain the violet colour of [Ti(H₂O)₆]³⁺ on the basis of crystal field theory.
Answer
🔵 Ti³⁺ → d¹ system.
🟢 In octahedral field, electron present in one t₂g orbital.
🟠 Absorption of visible light promotes it to e_g orbital → d–d transition.
🎯 The complementary colour observed is violet.
Question 5.26
What is meant by the chelate effect? Give an example.
Answer
💡 Chelate effect = Extra stability of complexes with multidentate ligands due to ring formation.
🟢 Example: [Cu(en)₂]²⁺ is more stable than [Cu(NH₃)₄]²⁺ because chelation reduces entropy loss and increases bond strength.
Question 5.27
Discuss briefly, giving one example in each case, the role of coordination compounds in:
(i) Biological systems
(ii) Medicinal chemistry
(iii) Analytical chemistry
(iv) Extraction/metallurgy of metals
Answer
🔵 Biological: Hemoglobin (Fe²⁺ complex with porphyrin).
🟢 Medicinal: Cisplatin [Pt(NH₃)₂Cl₂] → anticancer drug.
🟠 Analytical: Complexometric titrations using EDTA.
🔴 Metallurgy: Cyanide complexes [Ag(CN)₂]⁻ in silver extraction.
Question 5.28
How many ions are produced from the complex Co(NH₃)₆Cl₂ in solution?
(i) 6, (ii) 4, (iii) 3, (iv) 2
Answer
🟦 Complex: [Co(NH₃)₆]Cl₂ → gives [Co(NH₃)₆]²⁺ + 2Cl⁻.
🎯 Number of ions = 3.
✅ Correct answer: (iii) 3.
Question 5.29
Amongst the following ions which one has the highest magnetic moment value?
(i) [Cr(H₂O)₆]³⁺
(ii) [Fe(H₂O)₆]²⁺
(iii) [Zn(H₂O)₆]²⁺
Answer
🟦 [Cr(H₂O)₆]³⁺ → d³ → 3 unpaired → μ = √(3(3+2)) = 3.87 BM.
🟩 [Fe(H₂O)₆]²⁺ → d⁶ → 4 unpaired → μ = √(4(4+2)) = 4.90 BM.
🟧 [Zn(H₂O)₆]²⁺ → d¹⁰ → 0 unpaired → μ = 0.
🎯 Highest magnetic moment = [Fe(H₂O)₆]²⁺.
Question 5.30
Amongst the following, the most stable complex is:
(i) [Fe(H₂O)₆]³⁺
(ii) [Fe(NH₃)₆]³⁺
(iii) [Fe(C₂O₄)₃]³⁻
(iv) [FeCl₆]³⁻
Answer
🔵 Stability order: chelating ligands > neutral ligands > anions.
🟢 Most stable: [Fe(C₂O₄)₃]³⁻ (oxalate is bidentate, chelate effect).
Question 5.31
What will be the correct order for the wavelengths of absorption in the visible region for the following:
[Ni(NO₂)₆]⁴⁻, [Ni(NH₃)₆]²⁺, [Ni(H₂O)₆]²⁺ ?
Answer
💡 Stronger ligand → higher Δ → shorter wavelength absorbed.
🔵 NO₂⁻ > NH₃ > H₂O (ligand strength).
🟠 Wavelength order (longest → shortest):
[Ni(H₂O)₆]²⁺ > [Ni(NH₃)₆]²⁺ > [Ni(NO₂)₆]⁴⁻.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
📘 CODE 3 — Chemistry Class 12
Section A (Q1–Q16) — MCQs (16 × 1 = 16 marks)
Options for Assertion–Reason Questions:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Question 1. The IUPAC name of [Co(NH₃)₆]Cl₃ is:
Hexaamminecobalt(III) chloride
Hexaamminecobalt(II) chloride
Hexaamminecobalt trichloride
Cobalt(III) hexammine chloride
Answer: 1
Question 2. Which of the following is a chelating ligand?
NH₃
CN⁻
en (ethane-1,2-diamine)
H₂O
Answer: 3
Question 3. Which of the following shows optical isomerism?
[Pt(NH₃)₂Cl₂]
[Co(en)₃]³⁺
[NiCl₄]²⁻
[Fe(CN)₆]³⁻
Answer: 2
Question 4. In [Cu(NH₃)₄]²⁺, the coordination number of Cu is:
2
3
4
6
Answer: 3
Question 5. The oxidation state of Fe in K₃[Fe(CN)₆] is:
+2
+3
+4
0
Answer: 2
Question 6. Which of the following is a strong field ligand?
H₂O
OH⁻
NH₃
CN⁻
Answer: 4
Question 7. Assertion (A): [Fe(CN)₆]⁴⁻ is diamagnetic.
Reason (R): CN⁻ causes pairing of 3d electrons.
Answer: 1
Question 8. Assertion (A): [Ni(CN)₄]²⁻ is square planar.
Reason (R): CN⁻ is a strong field ligand.
Answer: 1
Question 9. Which geometry is shown by [Ni(CO)₄]?
Tetrahedral
Square planar
Octahedral
Trigonal bipyramidal
Answer: 1
Question 10. Which one is coloured?
[Zn(H₂O)₆]²⁺
[Ti(H₂O)₆]³⁺
[Cu(NH₃)₄]²⁺
Both 2 and 3
Answer: 4
Question 11. Which is not an ambidentate ligand?
SCN⁻
NO₂⁻
CN⁻
en
Answer: 4
Question 12. Which is the correct order of ligand strength?
I⁻ < Br⁻ < Cl⁻ < CN⁻
CN⁻ < NH₃ < H₂O < Br⁻
Br⁻ < Cl⁻ < OH⁻ < CO
NH₃ < CN⁻ < CO < I⁻
Answer: 1
Question 13. Which isomerism is not shown by [Co(NH₃)₆]³⁺?
Ionisation
Linkage
Optical
Geometrical
Answer: 2
Question 14. In [FeF₆]³⁻, the number of unpaired electrons is:
2
3
4
5
Answer: 4
Question 15. The hybridisation of Ni in [Ni(CN)₄]²⁻ is:
sp³
dsp²
d²sp³
sp³d²
Answer: 2
Question 16. The colour of [Ti(H₂O)₆]³⁺ is due to:
Charge transfer
d–d transition
Crystal defect
Ionisation
Answer: 2
Section B (Q17–Q21) — Very Short Answer (2 marks each)
Question 17. What is meant by the coordination number of a central atom?
Answer
🟦 Definition: Number of ligand donor atoms directly attached to the central metal ion.
🟢 Example: [Fe(CN)₆]³⁻ → CN=6.
Question 18. Differentiate between double salts and coordination compounds.
Answer
🟦 Double salts: Ionise completely; lose identity in solution. Eg. Mohr’s salt.
🟩 Coordination compounds: Retain identity as complex ion; do not dissociate completely. Eg. [Cu(NH₃)₄]SO₄.
Question 19. What is a chelate ligand? Give one example.
Answer
🟦 Chelating ligand = Multidentate ligand that forms ring with central ion.
🟩 Example: C₂O₄²⁻ (oxalate).
Question 20. Write the IUPAC name of [Cr(NH₃)₄Cl₂]Cl.
Answer
🟦 Complex: [Cr(NH₃)₄Cl₂]Cl
🟩 IUPAC Name = Tetraamminedichloridochromium(III) chloride.
Question 21. Name one biologically important coordination compound of Fe and one of Mg.
Answer
🟦 Hemoglobin → Fe²⁺ complex.
🟩 Chlorophyll → Mg²⁺ complex.
Section C (Q22–Q28) — Short Answer (3 marks each)
Question 22. What are ambidentate ligands? Give two examples.
Answer
🟦 Ligands with two possible donor atoms, but bind through only one at a time.
🟩 Examples: NO₂⁻ (N or O donor), SCN⁻ (S or N donor).
Question 23. Write the hybridisation, magnetic behaviour and geometry of [Fe(CN)₆]³⁻.
Answer
🟦 Fe³⁺ = d⁵.
🟩 CN⁻ = strong field ligand → pairing occurs.
🟧 Hybridisation = d²sp³.
🔴 Geometry = Octahedral.
⚡ Magnetic behaviour = 1 unpaired electron → paramagnetic.
Question 24. Differentiate between homoleptic and heteroleptic complexes with examples.
Answer
🟦 Homoleptic: Same type of ligands. Eg. [Co(NH₃)₆]³⁺.
🟩 Heteroleptic: Different ligands. Eg. [Co(NH₃)₄Cl₂]⁺.
Question 25. Give the formula and IUPAC name of potassium trioxalatochromate(III).
Answer
🟦 Formula = K₃[Cr(C₂O₄)₃].
🟩 Name = Potassium trioxalatochromate(III).
Question 26. What is linkage isomerism? Give one example.
Answer
🟦 Isomerism due to ambidentate ligands binding through different atoms.
🟩 Example: [Co(NH₃)₅(NO₂)]²⁺ (nitro) vs [Co(NH₃)₅(ONO)]²⁺ (nitrito).
Question 27. Explain why [Ni(CO)₄] is diamagnetic but [NiCl₄]²⁻ is paramagnetic.
Answer
🟦 [Ni(CO)₄]: Ni²⁺ (d⁸), CO strong field → pairing → dsp² → diamagnetic.
🟩 [NiCl₄]²⁻: Ni²⁺ (d⁸), Cl⁻ weak field → no pairing → sp³ → paramagnetic.
Question 28. Give two differences between crystal field theory (CFT) and valence bond theory (VBT).
Answer
🟦 VBT: Based on hybridisation concept, qualitative, does not explain colour.
🟩 CFT: Based on d-orbital splitting, quantitative, explains colour and magnetism.
Section D (Q29–Q30) — Case-Based Questions (2 × 4 = 8 marks)
Question 29.
Read the passage and answer:
When aqueous copper sulphate is treated with excess ammonia, a deep blue solution forms. Similarly, Fe³⁺ salts give blood red colour with thiocyanate ions.
(i) Write the formula of the complex formed with Cu²⁺ and ammonia.
(ii) Explain the colour of the solution formed.
(iii) Write the formula of the complex formed with Fe³⁺ and thiocyanate.
Answer
🟦 (i) Complex formed = [Cu(NH₃)₄(H₂O)₂]²⁺.
🟩 (ii) Colour arises due to d–d transitions in the octahedral crystal field.
🟧 (iii) Complex formed = [Fe(SCN)(H₂O)₅]²⁺ → gives blood-red colour.
Question 30.
Study the following passage and answer:
Potassium dichromate is a widely used oxidising agent. In acidic solution, it oxidises iodide ions to iodine and Fe²⁺ to Fe³⁺.
(i) Write the balanced ionic equation for oxidation of I⁻ by dichromate.
(ii) Write the balanced ionic equation for oxidation of Fe²⁺ by dichromate.
(iii) Why is K₂Cr₂O₇ orange in colour?
Answer
🟦 (i) Cr₂O₇²⁻ + 14H⁺ + 6I⁻ → 2Cr³⁺ + 3I₂ + 7H₂O
🟩 (ii) Cr₂O₇²⁻ + 14H⁺ + 6Fe²⁺ → 2Cr³⁺ + 6Fe³⁺ + 7H₂O
🟧 (iii) Orange colour due to charge transfer transition in Cr₂O₇²⁻.
Section E (Q31–Q33) — Long Answer (3 × 5 = 15 marks)
Question 31.
Explain the bonding in metal carbonyls.
OR
Explain the crystal field splitting in octahedral complexes.
Answer
🟦 Bonding in metal carbonyls
CO acts as a ligand through C atom → donates lone pair into vacant orbital of metal (σ donation).
Metal back-donates electrons from filled d-orbitals into π* antibonding orbital of CO (π-back bonding).
This synergic bonding stabilises the complex, weakens C–O bond, strengthens M–C bond.
Evidence: IR stretching frequency of C–O bond decreases compared to free CO.
✅ OR
🟩 Octahedral field splitting
d-orbitals split into lower energy t₂g (dxy, dxz, dyz) and higher energy e_g (dx²–y², dz²).
Energy difference = Δ₀ (CFSE).
Weak field ligands → small Δ₀ → high-spin complexes.
Strong field ligands → large Δ₀ → low-spin complexes.
Question 32.
Discuss isomerism in coordination compounds with examples.
OR
Explain the applications of coordination compounds in biological systems, medicine, industry and analysis.
Answer
🟦 Isomerism in coordination compounds
Structural isomerism
Ionisation: [Co(NH₃)₅SO₄]Br vs [Co(NH₃)₅Br]SO₄
Linkage: [Co(NH₃)₅(NO₂)]Cl₂ vs [Co(NH₃)₅(ONO)]Cl₂
Solvate: [Cr(H₂O)₆]Cl₃ vs [Cr(H₂O)₅Cl]Cl₂·H₂O
Stereoisomerism
Geometrical: cis-/trans- [Pt(NH₃)₂Cl₂]
Optical: [Co(en)₃]³⁺ shows enantiomers.
✅ OR
🟩 Applications
Biological: Hemoglobin (Fe²⁺ complex), Chlorophyll (Mg²⁺ complex).
Medicine: Cisplatin [Pt(NH₃)₂Cl₂] → anticancer.
Industry: Catalysts like Wilkinson’s catalyst, Ziegler–Natta.
Analysis: EDTA in complexometric titrations; [Fe(SCN)(H₂O)₅]²⁺ in detection of Fe³⁺.
Question 33.
Explain why [NiCl₄]²⁻ is paramagnetic and [Ni(CN)₄]²⁻ is diamagnetic.
OR
Describe the postulates of Werner’s coordination theory.
Answer
🟦 [NiCl₄]²⁻
Ni²⁺ → d⁸.
Cl⁻ = weak field ligand → sp³ hybridisation (tetrahedral).
No pairing → 2 unpaired electrons → paramagnetic.
🟩 [Ni(CN)₄]²⁻
Ni²⁺ → d⁸.
CN⁻ = strong field ligand → pairing occurs → dsp² hybridisation (square planar).
All electrons paired → diamagnetic.
✅ OR
🟧 Werner’s postulates
Metals show two types of valencies: primary (ionisable) and secondary (non-ionisable, fixed in space).
Secondary valency determines coordination number and geometry.
Complexes with same metal but different secondary valencies differ in properties.
Examples: [CoCl₃·6NH₃] ionises as [Co(NH₃)₆]Cl₃ → CN=6 (octahedral).
————————————————————————————————————————————————————————————————————————————
