Class 11 : Physics (In English) – Chapter 10: Thermal Properties of Matter
EXPLANATION & SUMMARY
🔷 EXPLANATION SECTION (≈1700 words)
🔵 1. Introduction
All materials absorb or release heat energy when their temperature changes. The study of how substances expand, contract, or change state with temperature is called Thermal Properties of Matter.
💡 Concept:
Heat is a form of energy that flows from a body at higher temperature to one at lower temperature until both reach thermal equilibrium.
🟢 2. Temperature and Heat
➡️ Temperature measures how hot or cold a body is and determines the direction of heat flow.
➡️ Heat is energy in transit due to a temperature difference.
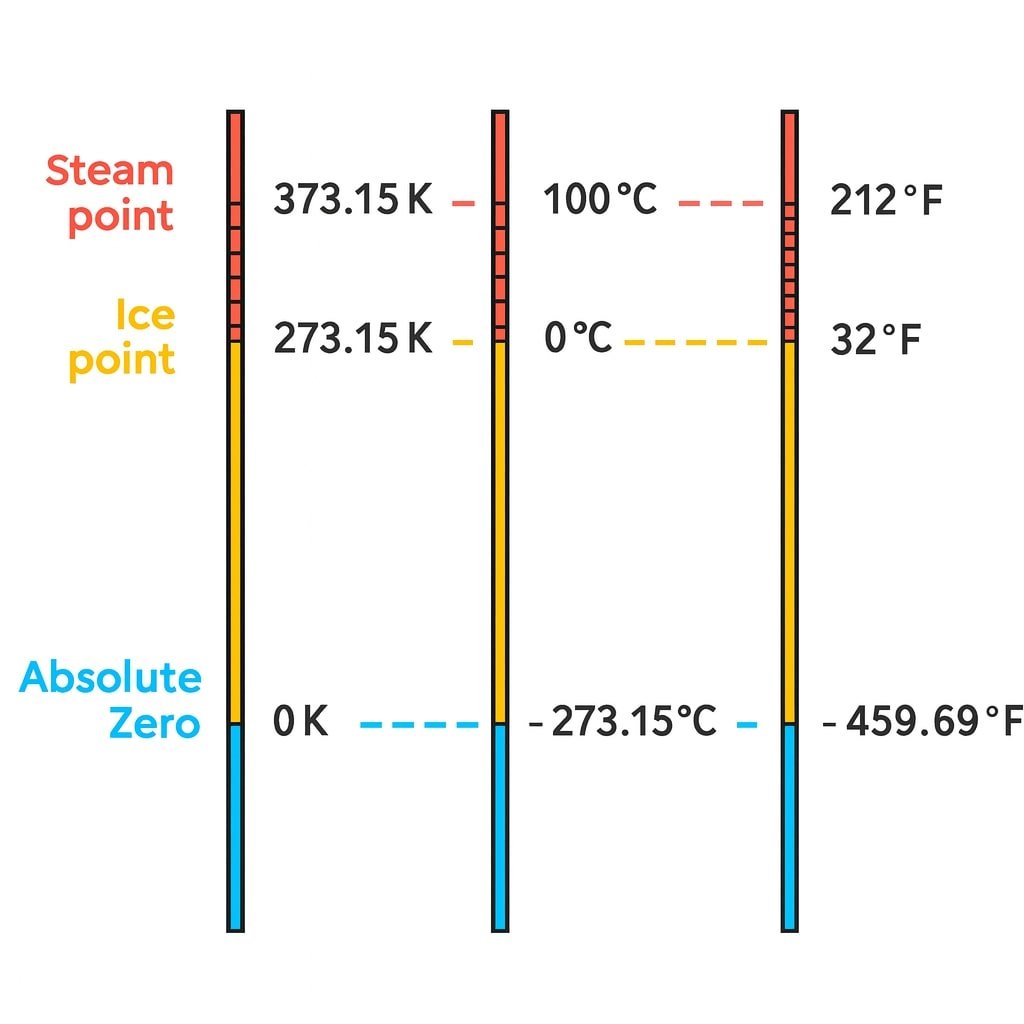
✔️ SI unit of temperature: Kelvin (K)
✔️ SI unit of heat: Joule (J)
💡 Conversion:
°C + 273 = K
Example: 27°C = 27 + 273 = 300 K
🔴 3. Thermal Expansion
When a body is heated, its molecules move faster and further apart. This causes expansion in length, area, or volume.
✴️ (a) Linear Expansion
If a rod of initial length L₀ increases in length by ΔL for a temperature rise ΔT, then
➡️ ΔL = α L₀ ΔT
where α = coefficient of linear expansion (per °C).
💡 Final length:
L = L₀ (1 + αΔT)
✴️ (b) Areal Expansion
For area A₀, the increase in area with rise in temperature is
➡️ ΔA = β A₀ ΔT, where β ≈ 2α
✴️ (c) Cubical Expansion
For volume V₀, the increase in volume is
➡️ ΔV = γ V₀ ΔT, where γ ≈ 3α
✔️ For isotropic solids, γ = 3α
💡 Note: Expansion is proportional to temperature rise only for small ΔT. For large temperature changes, the relationship becomes nonlinear.
🟡 4. Anomalous Expansion of Water
When water is cooled from 4°C to 0°C, it expands instead of contracting.
The volume is minimum and density maximum at 4°C.
💧 This is vital for aquatic life — ice floats on water and insulates the lower layers from freezing.
🔵 5. Thermal Expansion in Everyday Life
✔️ Gaps are left between railway tracks to avoid buckling due to heat.
✔️ Telephone wires sag more in summer due to expansion.
✔️ Glass utensils crack when heated suddenly (thermal stress).
🟢 6. Specific Heat Capacity
It is the heat required to raise the temperature of 1 kg of a substance by 1 K.
➡️ Q = m c ΔT
where
Q = heat supplied,
m = mass,
c = specific heat capacity,
ΔT = temperature change.
💡 Unit: J·kg⁻¹·K⁻¹
✔️ Water has very high specific heat (4186 J·kg⁻¹·K⁻¹), making it ideal for temperature regulation.
🔴 7. Molar Heat Capacity
It is the heat required to raise the temperature of 1 mole of a substance by 1 K.
➡️ C = Q / (n ΔT)
where n = number of moles.
Relation: C = M c,
where M = molar mass.
🟡 8. Principle of Calorimetry
When two bodies at different temperatures are mixed, heat lost by the hotter body equals heat gained by the colder body (if no heat loss to surroundings).
➡️ m₁ c₁ (T₁ − T) = m₂ c₂ (T − T₂)
✔️ Used to determine specific heat, latent heat, etc.
🔵 9. Change of State and Latent Heat
When a substance changes its phase (solid ⇄ liquid ⇄ gas), its temperature remains constant even though heat is exchanged.
💡 Latent Heat (L):
Amount of heat required to change the state of 1 kg of substance without temperature change.
➡️ Q = m L
✔️ Latent heat of fusion: Solid → Liquid
✔️ Latent heat of vaporisation: Liquid → Gas
🟢 10. Values for Water
Latent heat of fusion (L_f) = 3.34 × 10⁵ J·kg⁻¹
Latent heat of vaporisation (L_v) = 2.26 × 10⁶ J·kg⁻¹
💧 These high values make water an excellent coolant and temperature stabiliser.
🔴 11. Modes of Heat Transfer
Heat can be transferred by conduction, convection, or radiation.
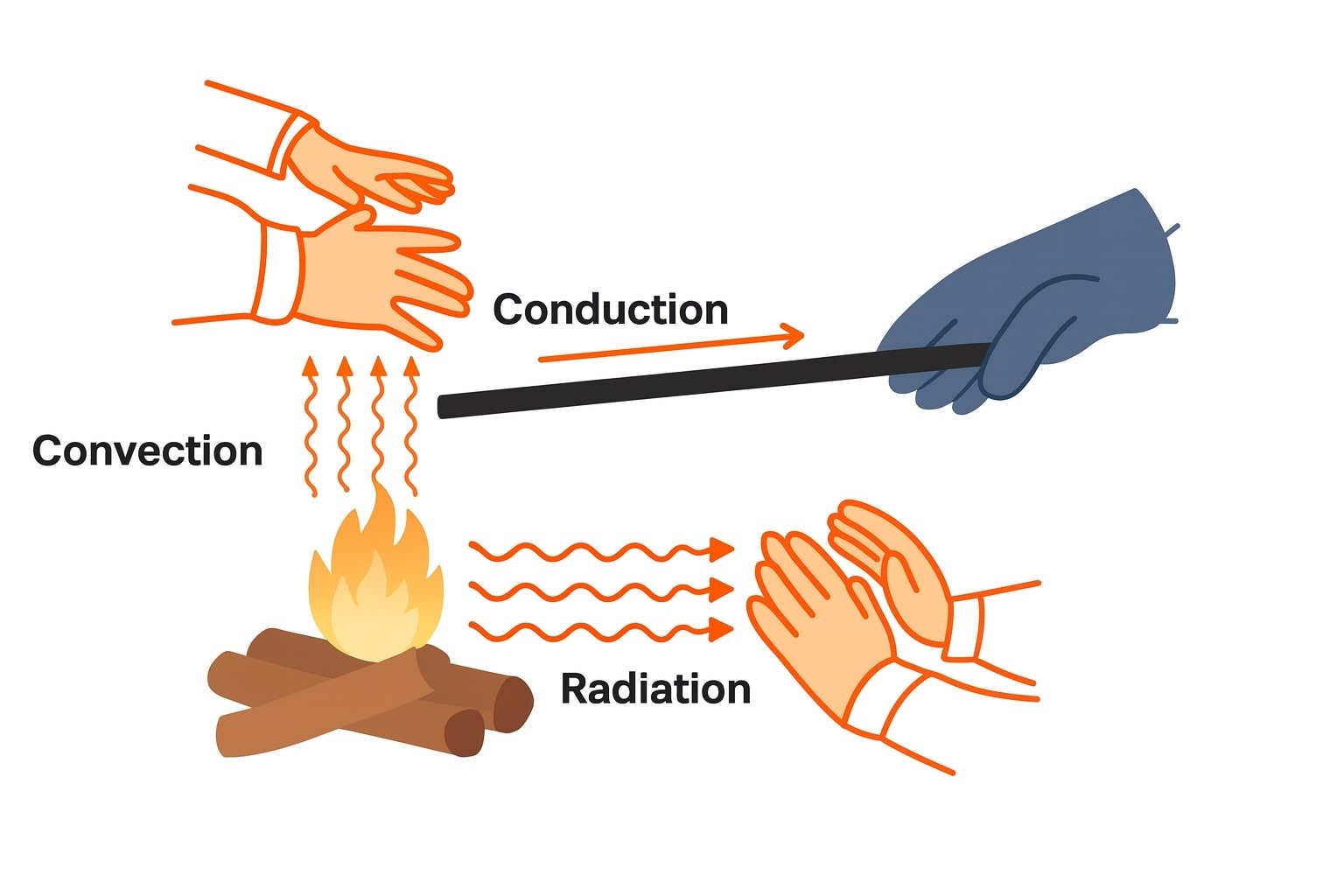
(a) Conduction
Heat transfer within a body without movement of particles.
➡️ Q = (k A ΔT t) / L
where
k = thermal conductivity,
A = cross-sectional area,
L = length,
ΔT = temperature difference,
t = time.
✔️ Metals like copper, silver are good conductors.
(b) Convection
Transfer of heat by movement of fluid particles (liquids/gases).
💡 Example: Sea and land breezes arise due to convection currents.
(c) Radiation
Transfer of heat without any medium, via electromagnetic waves.
✔️ Stefan–Boltzmann law:
➡️ E = σ T⁴,
where σ = 5.67 × 10⁻⁸ W·m⁻²·K⁻⁴
All bodies emit and absorb radiation; good absorbers are also good emitters.
🟡 12. Newton’s Law of Cooling
Rate of cooling ∝ temperature difference between body and surroundings.
➡️ dT/dt ∝ (T − Tₛ)
✔️ Valid for small temperature differences.
💡 Used in designing radiators and measuring emissivity.
🔵 13. Thermal Conductivity (k)
The property that quantifies how well a material conducts heat.
➡️ Rate of heat flow (Q/t) = (k A ΔT) / L
✔️ Unit: W·m⁻¹·K⁻¹
✔️ Metals → high k, good conductors
✔️ Air, wood → low k, insulators
🟢 14. Expansion of Liquids and Gases
Liquids expand more than solids; gases expand even more due to weak molecular forces.
➡️ ΔV = γ V₀ ΔT
For gases (at constant pressure),
➡️ V ∝ T (Charles’ Law)
🔴 15. Thermal Stress and Strain
If expansion is prevented, the body develops thermal stress.
➡️ Stress = Y α ΔT
where
Y = Young’s modulus,
α = coefficient of linear expansion,
ΔT = temperature change.
💡 Example: Sudden cooling of glass causes it to crack.
🟡 16. Relation Between Heat Capacities (for Gases)
For gases,
➡️ C_p − C_v = R
where
R = universal gas constant = 8.314 J·mol⁻¹·K⁻¹
✔️ Ratio γ = C_p / C_v characterises gas behaviour.
🔵 17. Calorimetry Applications
1️⃣ Mixing of Ice and Water:
m₁ c₁ (T₁ − T) = m₂ L_f + m₂ c₂ (T − 0)
2️⃣ Steam Condensation:
m_steam L_v + m_steam c_steam (100 − T) = m_water c_water (T − T₀)
💡 Principle: Heat Lost = Heat Gained
🟢 18. Real-Life Examples of Thermal Effects
✔️ Metal lids loosen on heating (expansion).
✔️ Bimetallic strips in thermostats bend with temperature change.
✔️ Sea water moderates coastal climate (high specific heat).
✔️ Ice floats due to lower density compared to water.
🔴 19. Important Constants
✔️ Specific heat of water = 4.18 × 10³ J·kg⁻¹·K⁻¹
✔️ Specific heat of ice = 2.10 × 10³ J·kg⁻¹·K⁻¹
✔️ Specific heat of steam = 2.01 × 10³ J·kg⁻¹·K⁻¹
✔️ Stefan constant σ = 5.67 × 10⁻⁸ W·m⁻²·K⁻⁴
🌟 SUMMARY (≈300 words)
Heat is energy transferred due to temperature difference.
Temperature measures thermal state; unit = Kelvin.
Substances expand with temperature rise:
ΔL = αL₀ΔT, ΔA = βA₀ΔT, ΔV = γV₀ΔT.
Water shows anomalous expansion: densest at 4°C.
Specific heat capacity (c): Q = m c ΔT.
Latent heat (L): Q = mL for phase change at constant temperature.
Thermal conductivity (k): rate of heat flow = (kAΔT)/L.
Heat transfer modes: conduction, convection, and radiation.
Stefan–Boltzmann law: E = σT⁴.
Newton’s cooling: dT/dt ∝ (T − Tₛ).
Stress due to restricted expansion: Stress = Y α ΔT.
For gases: C_p − C_v = R.
Calorimetry uses conservation of heat to find c or L.
Water’s high specific heat regulates climate and life processes.
🧠 QUICK RECAP
✔️ Q = m c ΔT → Heat–Temperature relation
✔️ Q = mL → Latent Heat
✔️ P = σT⁴ → Radiation law
✔️ Stress = Y α ΔT → Thermal Stress
✔️ C_p − C_v = R → Gas law
✔️ E = σT⁴, dT/dt ∝ (T − Tₛ) → Radiation & Cooling
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
🔵 Question 10.1
The triple points of neon and carbon dioxide are 24.57 K and 216.55 K respectively. Express these temperatures on the Celsius and Fahrenheit scales.
Answer:
🟢 Step 1: Convert Kelvin to Celsius
➡️ Formula: T(°C) = T(K) − 273.15
For Neon:
T₁(°C) = 24.57 − 273.15 = −248.58 °C
For Carbon dioxide:
T₂(°C) = 216.55 − 273.15 = −56.60 °C
🟡 Step 2: Convert Celsius to Fahrenheit
➡️ Formula: T(°F) = (9/5) × T(°C) + 32
For Neon:
T₁(°F) = (9/5) × (−248.58) + 32 = −415.44 °F
For Carbon dioxide:
T₂(°F) = (9/5) × (−56.60) + 32 = −69.88 °F
✔️ Final Answers:
Neon → −248.58 °C = −415.44 °F
Carbon dioxide → −56.60 °C = −69.88 °F
🔵 Question 10.2
Two absolute scales A and B have triple points of water defined to be 200 A and 350 B. What is the relation between T_A and T_B?
Answer:
🟢 Step 1: The triple point of water = 273.16 K
Therefore,
200 A = 273.16 K and 350 B = 273.16 K
🟡 Step 2: Express 1 A and 1 B in Kelvin
1 A = 273.16 / 200 = 1.3658 K
1 B = 273.16 / 350 = 0.7804 K
🟠 Step 3: Relation between T_A and T_B
T_A (in A-units) = T_B (in B-units)
T_A × 1.3658 = T_B × 0.7804
Hence,
T_B = 1.75 T_A
✔️ Final Relation: T_B = 1.75 T_A
🔵 Question 10.3
The electrical resistance in ohms of a certain thermometer varies with temperature according to the approximate law
R = R₀ [1 + α(T − T₀)]
The resistance is 101.6 Ω at the triple point of water 273.16 K, and 165.5 Ω at the normal melting point of lead (600.5 K). What is the temperature when the resistance is 123.4 Ω?
Answer:
🟢 Given:
R₀ = 101.6 Ω, T₀ = 273.16 K
R = 165.5 Ω at T = 600.5 K
Find α and then temperature for R = 123.4 Ω
🟡 Step 1: Using the formula
165.5 = 101.6 [1 + α(600.5 − 273.16)]
Divide both sides by 101.6:
1 + α(327.34) = 165.5 / 101.6 = 1.628
α = (1.628 − 1) / 327.34 = 0.00192 K⁻¹
🟠 Step 2: For R = 123.4 Ω
123.4 = 101.6 [1 + 0.00192 (T − 273.16)]
Divide both sides by 101.6:
1 + 0.00192 (T − 273.16) = 1.215
0.00192 (T − 273.16) = 0.215
T − 273.16 = 112.0
🔴 Step 3:
T = 385.16 K
✔️ Final Answer: Temperature ≈ 385 K
🔵 Question 10.4(a)
The triple point of water is a standard fixed point in modern thermometry. Why? What is wrong in taking the melting point and boiling point of water as standard fixed points?
Answer:
✔️ The triple point of water (273.16 K) is a unique, reproducible, and precise temperature independent of pressure and impurities.
❌ The melting and boiling points of water vary with atmospheric pressure and impurities, so they cannot serve as accurate fixed points.
🔵 Question 10.4(b)
In the Celsius scale, 0 °C and 100 °C were fixed at the melting and boiling points of water. On the absolute scale (Kelvin), one fixed point is 273.16 K. What is the other fixed point?
Answer:
Each 100 °C interval corresponds to 100 K.
Hence, second fixed point = 273.16 + 100 = 373.16 K.
✔️ Other fixed point: 373.16 K
🔵 Question 10.4(c)
The absolute temperature (Kelvin scale) T and Celsius temperature t_c are related as
t_c = T − 273.15
Why do we have 273.15 and not 273.16?
Answer:
273.15 K corresponds exactly to 0 °C by international agreement for simplicity in conversion.
The true triple point (273.16 K) differs by 0.01 K, which is negligible and ignored in practical use.
🔵 Question 10.4(d)
What is the temperature of the triple point of water on an absolute scale whose unit interval equals that of the Fahrenheit scale?
Answer:
💡 The Fahrenheit scale has 180 divisions for 100 °C interval.
Therefore, 1 K = 1.8 °F.
Triple point of water = 273.16 K = 273.16 × 1.8 = 491.7 on this scale.
✔️ Final Answer: 491.7 absolute Fahrenheit units
🔵 Question 10.5
Two ideal gas thermometers A and B use oxygen and hydrogen respectively. The following observations are made:
Triple point of water:
Pressure for thermometer A = 1.250 × 10⁵ Pa
Pressure for thermometer B = 0.200 × 10⁵ Pa
Normal melting point of sulphur:
Pressure for thermometer A = 1.797 × 10⁵ Pa
Pressure for thermometer B = 0.287 × 10⁵ Pa
(a) What is the absolute temperature of normal melting point of sulphur as read by thermometers A and B?
Answer:
🟢 For thermometer A:
T₂ / T₁ = P₂ / P₁
T₁ = 273.16 K
P₂ / P₁ = 1.797 / 1.250 = 1.4376
T₂ = 273.16 × 1.4376 = 392.6 K
🟡 For thermometer B:
P₂ / P₁ = 0.287 / 0.200 = 1.435
T₂ = 273.16 × 1.435 = 392.2 K
✔️ Final Answer: 392 K (approximately for both A and B)
(b) What is the reason behind the slight difference in readings of thermometers A and B? What further step is needed?
Answer:
🔵 The small difference arises because real gases slightly deviate from ideal gas behaviour, and different gases have different compressibilities.
🟢 To minimize this difference, readings should be extrapolated to zero pressure, where gases behave ideally and all thermometers give identical results.
✔️ After extrapolation, both thermometers will agree exactly.
🔵 Question 10.6
A steel tape 1 m long is correctly calibrated for a temperature of 27.0 °C. The length of a steel rod measured by this tape is found to be 63.0 cm on a hot day when the temperature is 45.0 °C. What is the actual length of the steel rod on that day? What is the length of the same steel rod on a day when the temperature is 27.0 °C? Coefficient of linear expansion of steel = 1.20 × 10⁻⁵ K⁻¹.
Answer
🟢 Step 1: Expansion factor of the tape at 45 °C
➡️ ΔT = 45 − 27 = 18 K
➡️ Scale factor = 1 + αΔT = 1 + (1.20×10⁻⁵)(18) = 1.000216
🟡 Step 2: Convert the reading (63.0 cm) to actual length at 45 °C
➡️ L₄₅ = (0.630 m) × 1.000216 = 0.630136 m = 63.014 cm
🔴 Step 3: Find length at 27 °C (rod is also steel)
➡️ L₂₇ = L₄₅ / (1 + αΔT) = 0.630136 / 1.000216 = 0.630 m = 63.0 cm
✔️ Final: Actual length at 45 °C ≈ 63.014 cm; length at 27 °C = 63.0 cm.
🔵 Question 10.7
A large steel wheel is to be fitted on to a shaft of the same material. At 27 °C, the outer diameter of the shaft is 8.70 cm and the diameter of the central hole in the wheel is 8.69 cm. The shaft is cooled using ‘dry ice’. At what temperature of the shaft does the wheel slip on the shaft? (α_steel = 1.20 × 10⁻⁵ K⁻¹.)
Answer
🟢 Step 1: Let shaft temperature be T (°C). For slipping, D_shaft(T) = D_hole(27 °C).
➡️ 8.70[1 + α(T − 27)] = 8.69
🟡 Step 2: Solve for T
➡️ 1 + α(T − 27) = 8.69/8.70 = 0.99885
➡️ α(T − 27) = −0.00115
➡️ T − 27 = −0.00115 / (1.20×10⁻⁵) ≈ −95.8 K
✔️ Final: T ≈ −68.8 °C (≈ −69 °C).
🔵 Question 10.8
A hole is drilled in a copper sheet. The diameter of the hole is 4.24 cm at 27.0 °C. What is the change in the diameter of the hole when the sheet is heated to 227 °C? (α_cu = 1.70 × 10⁻⁵ K⁻¹.)
Answer
🟢 Step 1: ΔT = 227 − 27 = 200 K
🟡 Step 2: Holes expand like the material: ΔD = D α ΔT
➡️ ΔD = 4.24 × (1.70×10⁻⁵) × 200 = 0.0144 cm = 0.144 mm
✔️ Final: Increase in diameter = 0.0144 cm (new diameter ≈ 4.254 cm).
🔵 Question 10.9
A brass wire 1.8 m long at 27 °C is held taut with little tension between two rigid supports. If the wire is cooled to a temperature of −39 °C, what is the tension developed in the wire, if its diameter is 2.0 mm? (α_brass = 2.0 × 10⁻⁵ K⁻¹; Y_brass = 0.91 × 10¹¹ Pa.)
Answer
🟢 Step 1: Temperature fall
➡️ ΔT = (−39 − 27) = −66 K (magnitude 66 K)
🟡 Step 2: Thermal strain prevented ⇒ stress = Y α ΔT
➡️ σ = (0.91×10¹¹)(2.0×10⁻⁵)(66) = 1.20×10⁸ Pa
🟠 Step 3: Cross-sectional area (d = 2.0 mm = 2.0×10⁻³ m)
➡️ A = (π/4)d² = π×10⁻⁶ m²
🔴 Step 4: Tension
➡️ F = σA = (1.20×10⁸)(π×10⁻⁶) ≈ 3.77×10² N
✔️ Final: Tension ≈ 3.8 × 10² N (≈ 377 N).
🔵 Question 10.10
A brass rod of length 50 cm and diameter 3.0 mm is joined to a steel rod of the same length and diameter. What is the change in length of the combined rod at 250 °C, if the original lengths are at 40.0 °C? Is there a ‘thermal stress’ developed at the junction? (α_brass = 2.0 × 10⁻⁵ K⁻¹, α_steel = 1.2 × 10⁻⁵ K⁻¹.) Ends are free to expand.
Answer
🟢 Step 1: ΔT = 250 − 40 = 210 K; L₀ (each) = 0.50 m
🟡 Step 2: Individual expansions
➡️ Brass: ΔL_b = L₀ α_b ΔT = 0.50×(2.0×10⁻⁵)×210 = 0.00210 m = 2.10 mm
➡️ Steel: ΔL_s = L₀ α_s ΔT = 0.50×(1.2×10⁻⁵)×210 = 0.00126 m = 1.26 mm
🟠 Step 3: Net expansion of combination
➡️ ΔL_total = ΔL_b + ΔL_s = 3.36 mm
🔴 Stress check (free ends): No external constraint ⇒ no thermal stress at the junction.
✔️ Final: Total expansion = 3.36 mm; no thermal stress develops.
🔵 Question 10.11
The coefficient of volume expansion of glycerine is 49 × 10⁻⁵ K⁻¹. What is the fractional change in its density for a 30 °C rise in temperature?
Answer
🟢 Step 1: For small changes, ρ ∝ 1/V ⇒ (Δρ/ρ) ≈ −βΔT
➡️ β = 49×10⁻⁵ = 4.9×10⁻⁴ K⁻¹
🟡 Step 2: Compute
➡️ Δρ/ρ = −(4.9×10⁻⁴)(30) = −1.47×10⁻²
✔️ Final: Fractional change in density = −0.0147 (i.e., density decreases by 1.47%).
🔵 Question 10.12
A 10 kW drilling machine is used to drill a bore in a small aluminium block of mass 8.0 kg. How much is the rise in temperature of the block in 2.5 minutes, assuming 50% of power is used in heating the machine itself or lost to the surroundings? (Specific heat of aluminium = 0.91 J g⁻¹ K⁻¹.)
Answer
🟢 Step 1: Effective power to block
➡️ P_eff = 0.5 × 10 kW = 5 kW = 5000 J s⁻¹
🟡 Step 2: Heat supplied in t = 2.5 min = 150 s
➡️ Q = P_eff × t = 5000 × 150 = 7.5×10⁵ J
🟠 Step 3: Use Q = m c ΔT with c_Al = 0.91 J g⁻¹ K⁻¹ = 910 J kg⁻¹ K⁻¹
➡️ ΔT = Q/(m c) = (7.5×10⁵)/(8.0×910) ≈ 103 K
✔️ Final: Rise in temperature ≈ 1.0×10² K (about 103 °C).
🔵 Question 10.13
A copper block of mass 2.5 kg is heated in a furnace to a temperature of 500 °C and then placed on a large ice block. What is the maximum amount of ice that can melt? (Specific heat of copper = 0.39 J g⁻¹ K⁻¹; latent heat of fusion of water = 335 J g⁻¹.)
Answer
🟢 Step 1: Heat released by copper on cooling from 500 °C to 0 °C
➡️ c_Cu = 0.39 J g⁻¹ K⁻¹ = 390 J kg⁻¹ K⁻¹
➡️ Q = m c ΔT = 2.5 × 390 × 500 = 4.875×10⁵ J
🟡 Step 2: Ice melted (L = 335 J g⁻¹ = 3.35×10⁵ J kg⁻¹)
➡️ m_ice = Q/L = (4.875×10⁵)/(3.35×10⁵) ≈ 1.46 kg
✔️ Final: Maximum ice melted ≈ 1.46 kg.
🔵 Question 10.14
In an experiment on the specific heat of a metal, a 0.20 kg block of the metal at 150 °C is dropped into a copper calorimeter of mass 0.025 kg containing 150 cm³ of water at 27 °C. The final temperature is 40 °C. Compute the specific heat of the metal. (Neglect heat loss. Take c_water = 4.2×10³ J kg⁻¹ K⁻¹, c_copper = 0.39 J g⁻¹ K⁻¹.)
Answer
🟢 Step 1: Known data
➡️ m_m = 0.20 kg, T_m: 150 → 40 °C (ΔT_m = 110 K)
➡️ m_w = 150 cm³ = 0.150 kg, ΔT_w = 13 K
➡️ m_cu = 0.025 kg, ΔT_cu = 13 K
➡️ c_w = 4200 J kg⁻¹ K⁻¹, c_cu = 390 J kg⁻¹ K⁻¹
🟡 Step 2: Heat lost by metal = Heat gained by water + calorimeter
➡️ m_m c_m ΔT_m = m_w c_w ΔT_w + m_cu c_cu ΔT_cu
➡️ 0.20 c_m (110) = 0.150(4200)(13) + 0.025(390)(13)
🟠 Step 3: Evaluate RHS
➡️ RHS = 8190 + 126.75 = 8316.75 J
🔴 Step 4: Solve for c_m
➡️ 22 c_m = 8316.75 ⇒ c_m ≈ 378.9 J kg⁻¹ K⁻¹
✔️ Final: Specific heat of the metal ≈ 3.79 × 10² J kg⁻¹ K⁻¹ (≈ 0.38 J g⁻¹ K⁻¹).
🔵 Question 10.15
Given below are observations on molar specific heats at room temperature of some common gases:
Hydrogen → 4.87 cal mol⁻¹ K⁻¹
Nitrogen → 4.97 cal mol⁻¹ K⁻¹
Oxygen → 5.02 cal mol⁻¹ K⁻¹
Nitric oxide → 4.99 cal mol⁻¹ K⁻¹
Carbon monoxide → 5.01 cal mol⁻¹ K⁻¹
Chlorine → 6.17 cal mol⁻¹ K⁻¹
The measured molar specific heats of these gases are markedly different from those for monatomic gases. Typically, molar specific heat of a monatomic gas is 2.92 cal mol⁻¹ K⁻¹. Explain this difference. What can you infer from the somewhat larger value for chlorine?
Answer:
🟢 Step 1: Recall molar heat for monatomic gases
➡️ For a monatomic gas, only translational motion contributes to energy.
➡️ Hence, Cᵥ = (3/2)R = 2.98 cal mol⁻¹ K⁻¹ ≈ 2.92 cal mol⁻¹ K⁻¹.
🟡 Step 2: Explanation for diatomic and polyatomic gases
➡️ Diatomic gases such as H₂, N₂, O₂, NO, CO have additional rotational degrees of freedom, increasing internal energy.
➡️ Thus, Cᵥ = (5/2)R ≈ 4.97 cal mol⁻¹ K⁻¹.
✅ This matches well with the given measured values (around 4.9–5.0).
🟠 Step 3: Reason for the higher value of chlorine (6.17 cal mol⁻¹ K⁻¹)
➡️ Heavier molecules like Cl₂ have vibrational modes that begin to get excited even at room temperature.
➡️ These extra degrees of freedom absorb additional energy, slightly increasing specific heat capacity.
🔴 Step 4: Final inference
✔️ Monatomic gases → translational energy only → ~2.92 cal mol⁻¹ K⁻¹.
✔️ Diatomic gases → translational + rotational → ~5 cal mol⁻¹ K⁻¹.
✔️ Heavier diatomic gases (e.g., Cl₂) → translational + rotational + partial vibrational → ~6 cal mol⁻¹ K⁻¹.
✅ Final Answer:
The higher specific heats of diatomic gases arise from rotational energy. Chlorine’s somewhat larger value indicates the beginning of vibrational motion participation at room temperature.
🔵 Question 10.16
A child running a temperature of 101 °F is given an antipyretic medicine that causes an increase in the rate of evaporation of sweat. If the fever is brought down to 98 °F in 20 minutes, what is the average rate of extra evaporation?
Mass of child = 30 kg, specific heat of human body = that of water, latent heat of vaporization = 580 cal g⁻¹.
Answer:
🟢 Step 1: Convert to Celsius
➡️ 101 °F = (5/9)(101 − 32) = 38.3 °C
➡️ 98 °F = (5/9)(98 − 32) = 36.7 °C
➡️ ΔT = 1.6 °C
🟡 Step 2: Heat lost by body
➡️ Q = m c ΔT
➡️ m = 30 kg = 3.0×10⁴ g, c = 1 cal g⁻¹ °C⁻¹
➡️ Q = 3.0×10⁴ × 1 × 1.6 = 4.8×10⁴ cal
🟠 Step 3: Evaporation of sweat (latent heat = 580 cal g⁻¹)
➡️ m_evap = Q / L = (4.8×10⁴) / 580 ≈ 82.8 g
🔴 Step 4: Time = 20 min = 1200 s
➡️ Rate = 82.8 g / 1200 s = 0.069 g s⁻¹
✔️ Final Answer: Average rate of extra evaporation = 0.069 g s⁻¹ (≈ 70 mg/s).
🔵 Question 10.17
A ‘thermocol’ icebox (30 cm cube, wall thickness 5 cm, k = 0.01 J s⁻¹ m⁻¹ K⁻¹) is used for storing cooked food. If 4.0 kg of ice is put in the box, estimate the amount of ice remaining after 6 h when the outside temperature is 45 °C.
Latent heat of fusion of water = 3.35×10⁵ J kg⁻¹.
Answer:
🟢 Step 1: Dimensions
Outer side = 30 cm = 0.30 m → inner side = 0.20 m (subtract twice the thickness).
Area for heat transfer ≈ 6 × (0.20)² = 0.24 m²
🟡 Step 2: Heat flow rate
➡️ Q̇ = kAΔT / x
➡️ Q̇ = (0.01)(0.24)(45 − 0)/0.05 = 2.16 J s⁻¹
🟠 Step 3: Total heat in 6 h
➡️ t = 6×3600 = 21600 s
➡️ Q = 2.16 × 21600 = 4.67×10⁴ J
🔴 Step 4: Ice melted
➡️ m = Q / L = (4.67×10⁴)/(3.35×10⁵) = 0.139 kg
✔️ Final Answer: Ice melted = 0.14 kg,
Ice remaining = 4.00 − 0.14 = 3.86 kg.
🔵 Question 10.18
A brass boiler has a base area 0.15 m² and thickness 1.0 cm. It boils water at the rate of 6.0 kg/min when placed on a gas stove. Estimate the temperature of the part of the flame in contact with the boiler.
Thermal conductivity of brass = 109 J s⁻¹ m⁻¹ K⁻¹, heat of vaporization of water = 2.26×10⁶ J kg⁻¹.
Answer:
🟢 Step 1: Heat conducted through brass = heat used for vaporization
➡️ Q/t = kAΔT / x = mL / t
🟡 Step 2: Substitute
m = 6 kg/min = 0.1 kg/s, L = 2.26×10⁶ J/kg, A = 0.15 m², x = 0.01 m, k = 109
➡️ (0.1)(2.26×10⁶) = 109×0.15×(ΔT)/0.01
➡️ 2.26×10⁵ = 1635 ΔT
➡️ ΔT = 138 K
🟠 Step 3: Water boiling temperature = 100 °C → flame side T = 100 + 138 = 238 °C
✔️ Final Answer: Temperature at contact with flame = 238 °C.
🔵 Question 10.19
Explain why:
(a) A body with large reflectivity is a poor emitter.
➡️ High reflectivity means less absorption; by Kirchhoff’s law, good absorbers are good emitters. Hence, a highly reflective surface emits poorly.
(b) A brass tumbler feels much colder than a wooden tray on a chilly day.
➡️ Brass is a better conductor; it draws heat rapidly from your hand, making it feel colder.
(c) An optical pyrometer calibrated for an ideal black body gives low readings for real objects.
➡️ Real bodies emit less radiation than a black body; hence apparent temperature is lower.
(d) The earth would be inhospitably cold without its atmosphere.
➡️ Greenhouse gases absorb and re-radiate infrared heat, maintaining warmth. Without this, heat would escape rapidly into space.
(e) Houses with whitewashed walls are cooler in summer.
➡️ White surfaces reflect sunlight, absorbing less heat, keeping interiors cooler.
✔️ Concept: Reflectivity and emissivity are complementary; conduction and radiation govern thermal sensations.
🔵 Question 10.20
A body cools from 80 °C to 50 °C in 5 minutes. Calculate the time it takes to cool from 60 °C to 30 °C. The temperature of surroundings is 20 °C.
Answer:
🟢 Step 1: Use Newton’s law of cooling
➡️ Rate ∝ (T − T_s), ⇒ (T − T_s)/(T₀ − T_s) = e^(−kt)
🟡 Step 2: From first data
For 80 → 50 °C in 5 min (300 s), T_s = 20 °C
(50 − 20)/(80 − 20) = e^(−k×300)
➡️ 30/60 = 0.5 = e^(−300k)
➡️ k = (ln 2)/300 = 0.00231 s⁻¹
🟠 Step 3: For 60 → 30 °C,
(30 − 20)/(60 − 20) = e^(−k t)
➡️ 10/40 = 0.25 = e^(−0.00231t)
➡️ ln(0.25) = −0.00231t
➡️ t = (1.3863)/0.00231 = 600 s
✔️ Final Answer: Time to cool = 10 minutes.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
🔷 Lesson: Thermal Properties of Matter – Section A (Q1–Q18)
Question 1:
The SI unit of specific heat capacity is:
🔵 (A) J·kg⁻¹·K⁻¹
🟢 (B) cal·g⁻¹·°C⁻¹
🟠 (C) erg·g⁻¹·K⁻¹
🔴 (D) J·mol⁻¹·K⁻¹
Answer: (A) J·kg⁻¹·K⁻¹
Question 2:
The relation between Celsius and Kelvin scales of temperature is:
🔵 (A) T(K) = t(°C) − 273
🟢 (B) T(K) = t(°C) + 273
🟠 (C) T(K) = t(°C) × 273
🔴 (D) T(K) = t(°C) / 273
Answer: (B) T(K) = t(°C) + 273
Question 3:
For a rod of length L₀, the increase in length ΔL due to temperature rise ΔT is given by:
🔵 (A) ΔL = βL₀ΔT
🟢 (B) ΔL = γL₀ΔT
🟠 (C) ΔL = αL₀ΔT
🔴 (D) ΔL = αL₀/ΔT
Answer: (C) ΔL = αL₀ΔT
Question 4:
Coefficient of linear expansion (α) has the unit:
🔵 (A) K
🟢 (B) K⁻¹
🟠 (C) m/K
🔴 (D) m²/K
Answer: (B) K⁻¹
Question 5:
Which of the following expands most for equal rise in temperature?
🔵 (A) Solid
🟢 (B) Liquid
🟠 (C) Gas
🔴 (D) All expand equally
Answer: (C) Gas
Question 6:
At which temperature does water have maximum density?
🔵 (A) 0°C
🟢 (B) 2°C
🟠 (C) 4°C
🔴 (D) 10°C
Answer: (C) 4°C
Question 7:
Heat required to raise temperature of unit mass by 1°C is called:
🔵 (A) Specific heat
🟢 (B) Latent heat
🟠 (C) Molar heat
🔴 (D) Thermal conductivity
Answer: (A) Specific heat
Question 8:
Latent heat is the:
🔵 (A) Heat absorbed without temperature change
🟢 (B) Heat absorbed with temperature rise
🟠 (C) Energy per mole per K
🔴 (D) Specific heat per unit mass
Answer: (A) Heat absorbed without temperature change
Question 9:
For a solid, the ratio γ/α is approximately equal to:
🔵 (A) 1
🟢 (B) 2
🟠 (C) 3
🔴 (D) ½
Answer: (C) 3
Question 10:
Which law is used in calorimetry?
🔵 (A) Newton’s law
🟢 (B) Law of conservation of heat
🟠 (C) Law of conservation of energy
🔴 (D) Law of conduction
Answer: (C) Law of conservation of energy
Question 11:
In conduction, heat flows due to:
🔵 (A) Movement of particles
🟢 (B) Vibration of particles
🟠 (C) Radiation
🔴 (D) None
Answer: (B) Vibration of particles
Question 12:
The relation for heat conduction is:
🔵 (A) Q = (A k ΔT t)/L
🟢 (B) Q = (k L t)/A
🟠 (C) Q = (A ΔT L)/k
🔴 (D) Q = (A t ΔT)/k
Answer: (A) Q = (A k ΔT t)/L
Question 13:
Stefan–Boltzmann constant σ has units:
🔵 (A) W·m⁻²·K⁻²
🟢 (B) W·m⁻²·K⁻⁴
🟠 (C) J·m⁻²·K⁻³
🔴 (D) W·m·K⁻⁴
Answer: (B) W·m⁻²·K⁻⁴
Question 14:
Radiant energy emitted per second per unit area of a black body is proportional to:
🔵 (A) T²
🟢 (B) T³
🟠 (C) T⁴
🔴 (D) 1/T
Answer: (C) T⁴
Question 15:
The rate of cooling of a hot body is proportional to:
🔵 (A) (T − Tₛ)
🟢 (B) (T + Tₛ)
🟠 (C) T/Tₛ
🔴 (D) T² − Tₛ²
Answer: (A) (T − Tₛ)
Question 16:
If expansion is prevented, the stress developed is given by:
🔵 (A) σ = Y ΔT
🟢 (B) σ = Y α ΔT
🟠 (C) σ = α ΔT / Y
🔴 (D) σ = Y / αΔT
Answer: (B) σ = Y α ΔT
Question 17:
For a gas, C_p − C_v = R. The value of R is:
🔵 (A) 8.314 J·mol⁻¹·K⁻¹
🟢 (B) 1.38 × 10⁻²³ J·K⁻¹
🟠 (C) 6.02 × 10²³ J·mol⁻¹·K⁻¹
🔴 (D) 9.8 J·mol⁻¹·K⁻¹
Answer: (A) 8.314 J·mol⁻¹·K⁻¹
Question 18:
Which of the following materials has the highest thermal conductivity?
🔵 (A) Copper
🟢 (B) Glass
🟠 (C) Wood
🔴 (D) Air
Answer: (A) Copper
🔶 SECTION B — Very Short & Short Answer Questions (Q19–Q23)
Question 19:
Define coefficient of linear expansion.
Answer:
💡 The coefficient of linear expansion (α) of a solid is the increase in length per unit original length per degree rise in temperature.
➡️ Formula: α = (ΔL) / (L₀ ΔT)
✔️ Unit: K⁻¹
✔️ It represents how much a material expands with temperature.
Question 20:
What is anomalous expansion of water?
Answer:
💧 When water is cooled from 4°C to 0°C, it expands instead of contracting.
➡️ It has maximum density at 4°C.
💡 This is called anomalous expansion and helps aquatic life survive in winter as water below ice stays at 4°C.
Question 21:
State and explain Newton’s law of cooling.
Answer:
🧠 Statement: The rate of cooling of a body is directly proportional to the temperature difference between the body and its surroundings, provided this difference is small.
➡️ Mathematically: dT/dt ∝ (T − Tₛ)
where T = temperature of body, Tₛ = temperature of surroundings.
💡 Application: Used in designing radiators and determining emissivity of materials.
Question 22:
What is thermal stress? Give its expression.
Answer:
✏️ Definition: When a material is heated but its expansion is prevented, it develops thermal stress.
➡️ Formula: σ = Y α ΔT
where
σ = stress, Y = Young’s modulus, α = coefficient of expansion, ΔT = temperature change.
✔️ This stress may cause materials like glass to crack during sudden cooling.
Question 23:
State Stefan–Boltzmann law.
Answer:
💡 The total radiant energy emitted per unit area per second by a black body is directly proportional to the fourth power of its absolute temperature.
➡️ E = σ T⁴
where σ = 5.67 × 10⁻⁸ W·m⁻²·K⁻⁴.
✔️ This law explains how radiating power increases rapidly with temperature.
🔷 SECTION C — Mid-Length Numericals / Theory (Q24–Q27)
Question 24:
A brass rod 1 m long expands by 1.8 × 10⁻³ m when its temperature rises by 100°C. Find its coefficient of linear expansion.
Answer:
✏️ Given:
L₀ = 1 m, ΔL = 1.8 × 10⁻³ m, ΔT = 100°C
➡️ Formula: α = ΔL / (L₀ ΔT)
➡️ α = (1.8 × 10⁻³) / (1 × 100) = 1.8 × 10⁻⁵ K⁻¹
✔️ Coefficient of linear expansion α = 1.8 × 10⁻⁵ K⁻¹
Question 25:
Explain how heat is transferred by conduction.
Answer:
💡 In solids, atoms vibrate around mean positions. When one end is heated, energy is passed from hot to cold region due to atomic collisions.
➡️ Rate of heat transfer:
Q/t = (k A ΔT) / L
where k = thermal conductivity.
✔️ Metals like copper and silver are good conductors.
✔️ Insulators such as wood and air have very low conductivity.
Question 26:
What is the relation between Cₚ and Cᵥ for gases?
Answer:
💡 For one mole of an ideal gas:
➡️ Cₚ − Cᵥ = R, where R = 8.314 J·mol⁻¹·K⁻¹
✔️ Cₚ is molar specific heat at constant pressure.
✔️ Cᵥ is molar specific heat at constant volume.
✔️ The difference arises because gases do external work at constant pressure.
Question 27:
A metal ball of mass 0.5 kg is heated from 30°C to 80°C, requiring 1050 J of heat. Find its specific heat.
Answer:
✏️ Given:
m = 0.5 kg, Q = 1050 J, ΔT = 50°C
➡️ Formula: Q = m c ΔT
c = Q / (m ΔT)
➡️ c = 1050 / (0.5 × 50) = 1050 / 25 = 42 J·kg⁻¹·K⁻¹
✔️ Specific heat = 42 J·kg⁻¹·K⁻¹
🔶 SECTION D — Long Answer Questions (Q28–Q31)
Question 28:
Explain the three modes of heat transfer with examples.
Answer:
💡 Heat transfer occurs in three distinct ways — Conduction, Convection, and Radiation.
➡️ (a) Conduction:
✏️ Heat flows through a substance from a region of higher temperature to lower temperature without movement of particles.
Formula: Q = (kAΔTt) / L
where,
k = thermal conductivity, A = area, L = length, ΔT = temperature difference, t = time.
✔️ Example: Handle of a metal pan becomes hot even when only one end is heated.
➡️ (b) Convection:
✏️ Heat is transferred by actual movement of fluid particles from hot region to cold region.
✔️ Example: Formation of sea and land breezes, heating of water in a pot.
💡 Warm fluid rises and cooler fluid descends, forming convection currents.
➡️ (c) Radiation:
✏️ Transfer of heat without any medium, by electromagnetic waves (infrared).
Stefan–Boltzmann Law: E = σT⁴
✔️ Example: Heat from the Sun reaches the Earth through space.
🧠 Summary:
Conduction → solids
Convection → liquids & gases
Radiation → vacuum
Question 29:
Derive the expression for rate of heat flow by conduction in a uniform rod.
Answer:
✏️ Step 1: Concept
Let one end of a rod be at temperature T₁ and the other at T₂ (T₁ > T₂).
💡 Heat flows from hot to cold end.
✏️ Step 2: Relation
Rate of heat flow (Q/t) ∝
temperature difference (T₁ − T₂)
area of cross-section (A)
and inversely ∝
length (L)
Thus, Q/t ∝ A(T₁ − T₂)/L
✏️ Step 3: Introducing constant of proportionality
➡️ Q/t = k A (T₁ − T₂)/L,
where k = thermal conductivity.
✔️ Unit: W·m⁻¹·K⁻¹
💡 Greater value of k means better conductor (e.g., copper).
Question 30:
Explain Newton’s law of cooling and derive its expression.
Answer:
💡 Statement:
The rate of cooling of a body is directly proportional to the difference in temperature between the body and its surroundings, for small temperature differences.
✏️ Step 1: Let T = temperature of body, Tₛ = surroundings temperature.
Rate of loss of heat = −dQ/dt = hA(T − Tₛ),
where h = heat transfer coefficient.
✏️ Step 2: Since Q = m c ΔT,
−m c (dT/dt) = hA(T − Tₛ)
➡️ dT/dt = − (hA / m c) (T − Tₛ)
✔️ Thus, rate of fall of temperature is proportional to (T − Tₛ).
💡 Application:
Used in cooling of hot liquids, measurement of emissivity, and in industrial cooling design.
Question 31:
What is thermal stress? Derive its expression.
Answer:
✏️ Definition:
If a material is heated and its expansion is prevented, thermal stress develops within it.
✏️ Step 1: Without restriction:
Free expansion = ΔL = α L₀ ΔT
✏️ Step 2: When expansion is prevented:
No change in length ⇒ strain prevented.
Hence, strain = α ΔT (tendency only).
✏️ Step 3: Stress–strain relation:
Stress = Y × strain
➡️ σ = Y α ΔT
where,
σ = thermal stress,
Y = Young’s modulus,
α = coefficient of linear expansion,
ΔT = temperature rise.
✔️ Unit: N·m⁻² (Pascal)
💡 If expansion is not allowed, internal stress may cause deformation or fracture (e.g., glass cracking on rapid cooling).
🔷 SECTION E — Case / Application Based Questions (Q32–Q33)
Question 32:
A copper rod (k = 400 W·m⁻¹·K⁻¹) of length 0.2 m and area 2 × 10⁻⁴ m² has its ends kept at 80°C and 20°C. Find the rate of heat flow through it.
Answer:
✏️ Given:
k = 400 W·m⁻¹·K⁻¹
A = 2 × 10⁻⁴ m²
ΔT = (80 − 20) = 60 K
L = 0.2 m
➡️ Formula: Q/t = (k A ΔT) / L
Substitute →
Q/t = (400 × 2 × 10⁻⁴ × 60) / 0.2
= (400 × 0.0002 × 60) / 0.2
= (4.8) / 0.2 = 24 W
✔️ Rate of heat flow = 24 joules per second (24 W)
Question 33:
A steel wire (α = 1.2 × 10⁻⁵ K⁻¹, Y = 2 × 10¹¹ N·m⁻²) is heated through 50°C while its ends are fixed. Find the thermal stress developed.
Answer:
✏️ Given:
α = 1.2 × 10⁻⁵ K⁻¹
Y = 2 × 10¹¹ N·m⁻²
ΔT = 50°C
➡️ Formula: σ = Y α ΔT
σ = 2 × 10¹¹ × 1.2 × 10⁻⁵ × 50
σ = 2 × 10¹¹ × 6 × 10⁻⁴
σ = 1.2 × 10⁸ N·m⁻²
✔️ Thermal stress = 1.2 × 10⁸ Pa (compressive)
💡 Such large stresses can damage rigid joints if expansion is prevented.
————————————————————————————————————————————————————————————————————————————
