Class 11 : Chemistry (In English) – Chapter 1: Some Basic Concepts of Chemistry
EXPLANATION & SUMMARY

📌 1. Importance of Chemistry
Question: Why do we study chemistry?
Answer:
Chemistry helps us understand the composition, structure, and transformations of matter.
It plays a crucial role in diverse fields like medicine, agriculture, food production, material science, and environmental studies.
Chemistry contributes to national growth through industrial and technological development.
New materials, fuels, polymers, fertilizers, and life-saving drugs are all products of chemistry.
📌 2. Nature of Matter
Key Points:
Matter is anything that has mass and occupies space.
It can exist in three physical states: solid, liquid, and gas.
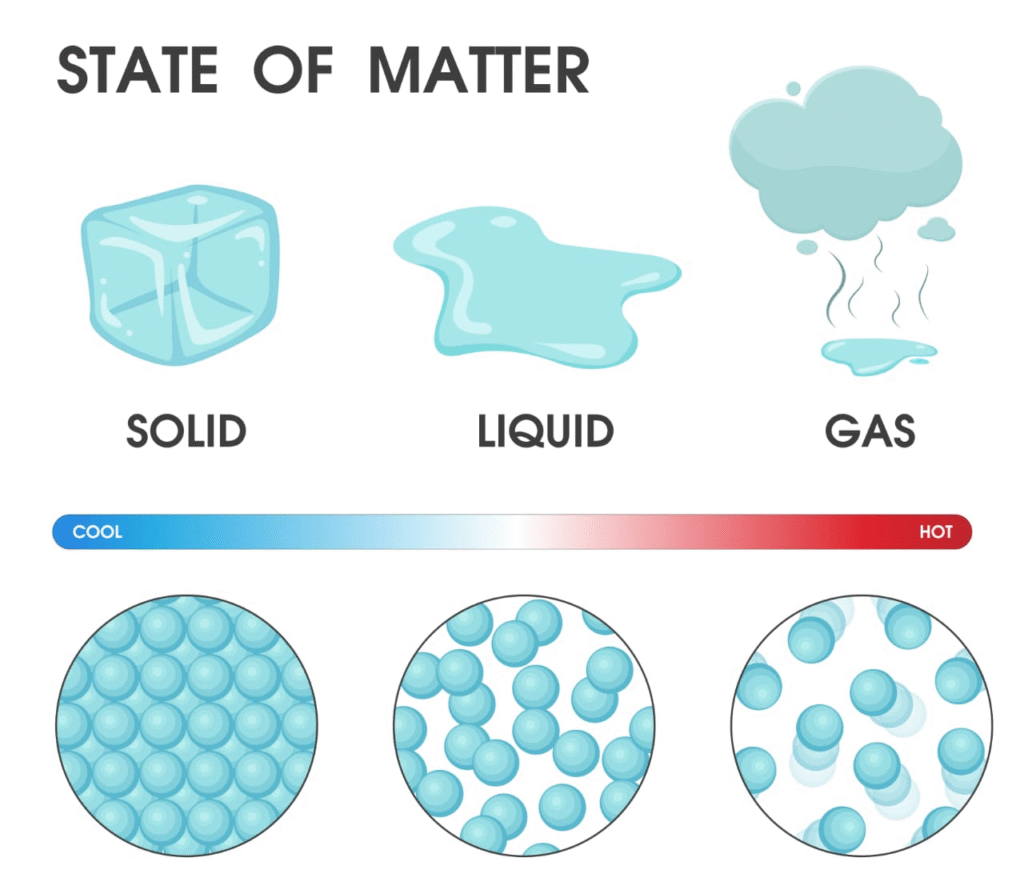
It can be classified chemically into:
Pure substances: Elements and compounds
Mixtures: Homogeneous and heterogeneous mixtures
Examples:
– Gold is an element (pure substance)
– Air is a homogeneous mixture
– Sand and iron filings is a heterogeneous mixture
📌 3. Properties of Matter and Their Measurement
Types of Properties:
Physical properties – Observable without changing composition (e.g., boiling point, density)
Chemical properties – Observed when substance changes into another (e.g., rusting of iron)
Units of Measurement:
Use of SI Units (International System)
Base Units: Mass (kg), Length (m), Time (s), Temperature (K), Amount of substance (mol), etc.
Derived Units:
Volume (m³), Density (kg/m³), etc.
Significant Figures & Scientific Notation:
Significant figures reflect precision in measurement
Scientific notation simplifies writing very large/small numbers
Example: 0.00032 = 3.2 × 10⁻⁴
📌 4. Uncertainty in Measurement
Accuracy refers to closeness to the actual value.
Precision refers to how close repeated values are to each other.
Errors in Measurement:
Systematic Errors – Have definite causes and follow a pattern
Random Errors – Occur due to chance, unpredictable
Calculation Rules for Significant Figures:
In addition/subtraction: Least decimal places rule
In multiplication/division: Least significant figures rule
📌 5. Laws of Chemical Combinations
These laws describe how substances react:
🧪 1. Law of Conservation of Mass
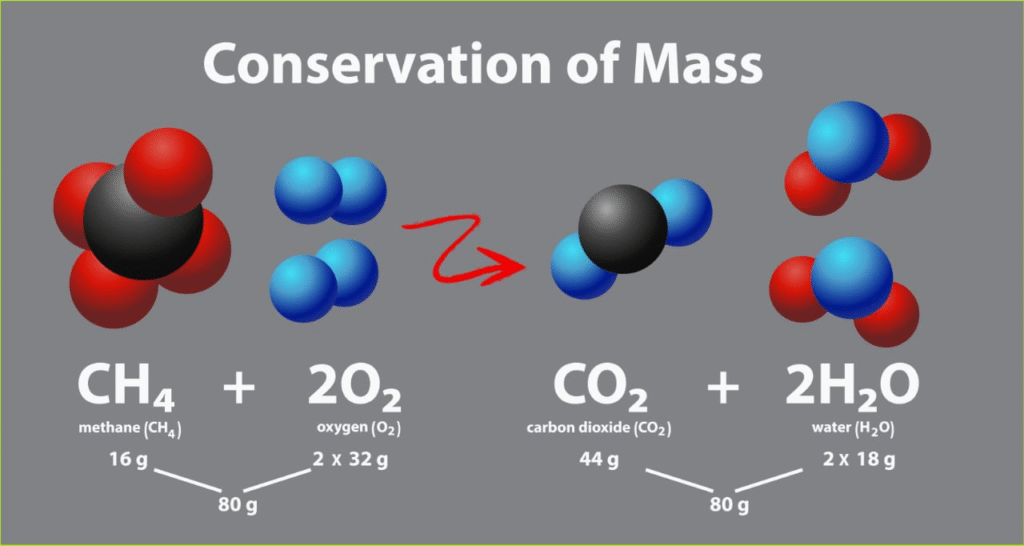
“Mass can neither be created nor destroyed.”
Total mass before reaction = Total mass after reaction
🧪 2. Law of Definite Proportions
A chemical compound always contains its elements in a fixed ratio by mass.
🧪 3. Law of Multiple Proportions
If two elements combine to form more than one compound, the ratio of masses of one element that combine with a fixed mass of the other is a simple whole number.
🧪 4. Gay Lussac’s Law of Gaseous Volumes
Gases react in simple whole number ratios by volume at constant temperature and pressure.
🧪 5. Avogadro’s Law
Equal volumes of gases at the same temperature and pressure contain equal number of molecules.
📌 6. Dalton’s Atomic Theory
Postulates:
All matter is made up of atoms
Atoms are indivisible and indestructible
Atoms of same element are identical in mass and properties
Atoms combine in fixed ratios to form compounds
In chemical reactions, atoms are rearranged, not created/destroyed
📌 7. Atomic and Molecular Masses
🧮 Atomic Mass
Relative atomic mass = Mass of one atom compared to 1/12 of a carbon-12 atom
Example: Atomic mass of hydrogen = 1.008 u
🧮 Molecular Mass
Sum of atomic masses in a molecule
Example: H₂O → 2×1.008 + 16 = 18.016 u
📌 8. Mole Concept and Molar Mass
🧪 1 mole = 6.022 × 10²³ particles (Avogadro Number)
Can be atoms, ions, molecules, etc.
🧮 Molar Mass = Mass of 1 mole of a substance
Expressed in g/mol
Example: Molar mass of H₂O = 18.016 g/mol
Conversions using mole:
Moles = Given mass / Molar mass
Moles = Number of particles / Avogadro’s number
Moles = Volume of gas at STP / 22.4 L
📌 9. Percentage Composition
Formula:
% of element = (Mass of element / Molar mass of compound) × 100
Example:
In H₂O:
% H = (2 × 1) / 18 × 100 = 11.1%
% O = (16) / 18 × 100 = 88.9%
📌 10. Empirical and Molecular Formula
🧪 Empirical Formula – Simplest whole-number ratio of atoms
🧪 Molecular Formula – Actual number of atoms of each element in one molecule
Steps to Find Empirical Formula:
Convert % to grams
Convert grams to moles (divide by atomic mass)
Divide all moles by the smallest mole value
Round to nearest whole number
Molecular Formula = Empirical Formula × n, where
n = Molar mass / Empirical formula mass
📌 11. Stoichiometry and Calculations Based on Chemical Equations
Stoichiometry = Quantitative study of reactants and products
Balanced Equation Example:
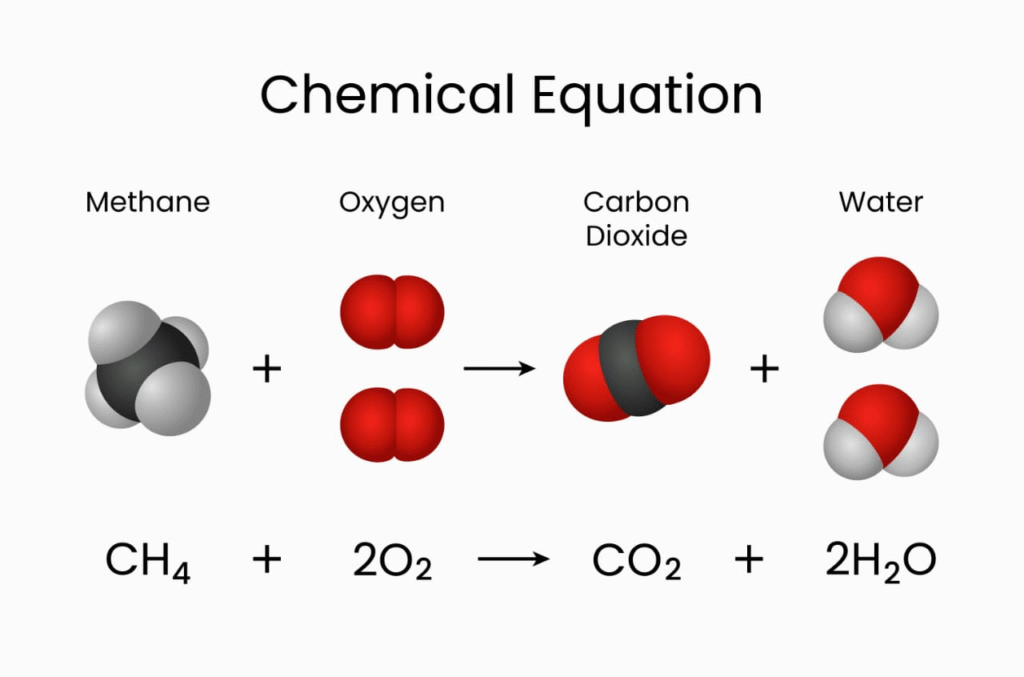
CH₄ + 2O₂ → CO₂ + 2H₂O
From the above:
1 mole CH₄ reacts with 2 moles O₂
Produces 1 mole CO₂ and 2 moles H₂O
Use mole ratios to solve:
Mass → Moles → Mole ratio → Moles → Mass
📌 12. Limiting Reagent
In a chemical reaction, the limiting reagent is the one that gets used up first, thus limiting the amount of product formed.
Steps:
Calculate moles of all reactants
Determine which reactant produces the least product → that’s the limiting reagent
📌 13. Reactions in Solutions: Molarity and Concentration
Molarity (M) = Moles of solute / Volume of solution (in L)
Other terms:
Molality = Moles of solute / Mass of solvent (kg)
Normality = Equivalents of solute / Volume of solution (L)
Mole fraction = Moles of component / Total moles
📌 14. Percent Yield
In reactions:
Theoretical Yield = Maximum calculated yield
Actual Yield = What you get practically
% Yield = (Actual Yield / Theoretical Yield) × 100
✅ Summary (≈300 words)
This chapter lays the foundation for chemistry by introducing essential concepts such as the classification of matter, measurement of properties, and the laws governing chemical reactions. It starts by explaining the significance of chemistry in everyday life and its wide-ranging applications.
Matter is classified physically into solids, liquids, and gases, and chemically into pure substances and mixtures. Properties are measured using SI units, and the concepts of accuracy, precision, and error in measurements are explored using significant figures and scientific notation.
The chapter covers important chemical laws such as the Law of Conservation of Mass, Definite and Multiple Proportions, Gay Lussac’s Law, and Avogadro’s Law. Dalton’s Atomic Theory is presented as the historical basis for understanding atoms.
Atomic and molecular masses are defined and calculated, followed by an explanation of the mole concept and molar mass. Students learn how to convert between mass, number of particles, and volume using the mole.
Percentage composition and empirical/molecular formulas are explained with step-by-step methods. Stoichiometry is introduced to connect chemical equations to quantitative calculations. The concept of limiting reagent is used to predict product formation in reactions.
The chapter ends with discussions on solution concentrations such as molarity, molality, and mole fraction, and the calculation of percent yield based on theoretical and actual outcomes.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
🔵 Question 1.1
Calculate the molar mass of the following:
(i) H₂O (ii) CO₂ (iii) CH₄
🟢 Answer 1.1
(i) H₂O
→ Atomic mass of H = 1.008 u, O = 16.00 u
→ Molar mass = 2 × 1.008 + 16.00 = 18.016 g/mol
(ii) CO₂
→ C = 12.01 u, O = 16.00 u
→ Molar mass = 12.01 + 2 × 16.00 = 44.01 g/mol
(iii) CH₄
→ C = 12.01 u, H = 1.008 u
→ Molar mass = 12.01 + 4 × 1.008 = 16.042 g/mol
🔵 Question 1.2
Calculate the mass percent of different elements present in sodium sulphate (Na₂SO₄).
🟢 Answer 1.2
Molar mass of Na₂SO₄ = 2×23 + 32 + 4×16 = 142 g/mol
% Na = (46 / 142) × 100 = 32.39%
% S = (32 / 142) × 100 = 22.54%
% O = (64 / 142) × 100 = 45.07%
🔵 Question 1.3
Determine the empirical formula of an oxide of iron, which has 69.9% iron and 30.1% dioxygen by mass.
🟢 Answer 1.3
Step 1: Assume 100 g sample: Fe = 69.9 g, O = 30.1 g
Step 2: Moles of Fe = 69.9 / 55.85 ≈ 1.251
Moles of O = 30.1 / 16.00 ≈ 1.881
Step 3: Ratio: Fe:O = 1.251:1.881 ≈ 2:3
Empirical formula = Fe₂O₃
🔵 Question 1.4
Calculate the amount of carbon dioxide that could be produced when
(i) 1 mole of carbon is burnt in air
(ii) 1 mole of carbon is burnt in 16 g of dioxygen
(iii) 2 moles of carbon are burnt in 16 g of dioxygen
🟢 Answer 1.4
Balanced reaction: C + O₂ → CO₂
(i) 1 mol C + 1 mol O₂ → 1 mol CO₂
→ 1 mol CO₂ = 44 g produced
(ii) 16 g O₂ = 1 mol
→ With 1 mol C → 44 g CO₂
(iii) 16 g O₂ = 1 mol, but 2 mol C
→ O₂ is limiting
→ 1 mol C reacts → 44 g CO₂
🔵 Question 1.5
Calculate the mass of sodium acetate (CH₃COONa) required to make 500 mL of 0.375 molar aqueous solution.
(Molar mass of CH₃COONa = 82.0245 g mol⁻¹)
🟢 Answer 1.5
Moles = Molarity × Volume (L) = 0.375 × 0.5 = 0.1875 mol
Mass = Moles × Molar mass = 0.1875 × 82.0245 = 15.38 g
🔵 Question 1.6
Calculate the concentration of nitric acid in moles per litre in a sample which has a density 1.41 g mL⁻¹ and mass % of HNO₃ = 69%.
🟢 Answer 1.6
In 1000 mL: mass = 1.41 × 1000 = 1410 g
Mass of HNO₃ = 69% of 1410 = 972.9 g
Molar mass of HNO₃ = 63 g/mol
Moles = 972.9 / 63 ≈ 15.44 mol
Molarity = 15.44 M
🔵 Question 1.7
How much copper can be obtained from 100 g of copper sulphate (CuSO₄)?
🟢 Answer 1.7
Molar mass CuSO₄ = 63.5 + 32 + 4×16 = 159.5 g/mol
% Cu = (63.5 / 159.5) × 100 ≈ 39.81%
Mass of Cu = 39.81% of 100 = 39.81 g
🔵 Question 1.8
Determine the molecular formula of an oxide of iron, in which the mass % of iron and oxygen are 69.9 and 30.1 respectively.
🟢 Answer 1.8
Same as Q1.3
Fe = 69.9 g → 1.251 mol
O = 30.1 g → 1.881 mol
Ratio ≈ 2:3
Empirical formula = Fe₂O₃,
Empirical mass = 2×55.85 + 3×16 = 159.7 g/mol
Molecular formula = Fe₂O₃
🔵 Question 1.9
Calculate the atomic mass (average) of chlorine using the following data:
35Cl (75.77%, 34.9689 u),
37Cl (24.23%, 36.9659 u)
🟢 Answer 1.9
Average atomic mass =
(75.77×34.9689 + 24.23×36.9659)/100
= (2650.18 + 895.87)/100 = 35.544 u
🔵 Question 1.10
In three moles of ethane (C₂H₆), calculate the following:
(i) Number of moles of carbon atoms
(ii) Number of moles of hydrogen atoms
(iii) Number of molecules of ethane
🟢 Answer 1.10
(i) 1 mol C₂H₆ → 2 mol C
→ 3 mol C₂H₆ → 3×2 = 6 mol C
(ii) 1 mol C₂H₆ → 6 mol H
→ 3 mol C₂H₆ → 3×6 = 18 mol H
(iii) 1 mol = 6.022×10²³ molecules
→ 3 mol = 1.807 × 10²⁴ molecules
🔵 Question 1.11
What is the concentration of sugar (C₁₂H₂₂O₁₁) in mol L⁻¹ if its 20 g are dissolved in enough water to make a final volume up to 2 L?
🟢 Answer 1.11
Molar mass of C₁₂H₂₂O₁₁ = (12×12.01 + 22×1.008 + 11×16) = 342.3 g/mol
Moles of sugar = 20 / 342.3 = 0.05844 mol
Molarity = Moles / Volume = 0.05844 / 2 = 0.0292 mol/L
🔵 Question 1.12
If the density of methanol is 0.793 kg L⁻¹, what is its volume needed for making 2.5 L of its 0.25 M solution?
🟢 Answer 1.12
Molar mass of CH₃OH = 32.04 g/mol
Moles required = 0.25 × 2.5 = 0.625 mol
Mass = 0.625 × 32.04 = 20.025 g
Density = 0.793 g/mL = 0.793 kg/L
Volume = Mass / Density = 20.025 / 0.793 = 25.25 mL or 0.02525 L
🔵 Question 1.13
Pressure is determined as force per unit area of the surface. The SI unit of pressure, pascal is shown below:
1 Pa = 1 N m⁻²
If mass of air at sea level is 1034 g cm⁻², calculate the pressure in pascal.
🟢 Answer 1.13
Force = mass × g = 1034 g = 1.034 kg × 9.8 = 10.1332 N
Area = 1 cm² = 1 × 10⁻⁴ m²
Pressure = Force / Area = 10.1332 / 1×10⁻⁴ = 1.013 × 10⁵ Pa
🔵 Question 1.14
What is the SI unit of mass? How is it defined?
🟢 Answer 1.14
The SI unit of mass is kilogram (kg).
It is defined by fixing the value of Planck’s constant h = 6.62607015 × 10⁻³⁴ Js, and using a defined meter and second.
🔵 Question 1.15
Match the following prefixes with their multiples:
(i) micro 10⁻⁶
(ii) deca 10¹
(iii) mega 10⁶
(iv) giga 10⁹
(v) femto 10⁻¹⁵
🟢 Answer 1.15
(i) micro → 10⁻⁶
(ii) deca → 10¹
(iii) mega → 10⁶
(iv) giga → 10⁹
(v) femto → 10⁻¹⁵
✅ Correctly matched
🔵 Question 1.16
What do you mean by significant figures?
🟢 Answer 1.16
Significant figures are the digits in a number that carry meaningful information about its precision.
They include:
✔️ All non-zero digits
✔️ Zeros between significant digits
✔️ Trailing zeros in decimal numbers
They indicate the certainty in measurement.
🔵 Question 1.17
A sample of drinking water was found to be severely contaminated with chloroform, CHCl₃, supposed to be carcinogenic in nature. The level of contamination was 15 ppm (by mass).
(i) Express this in per cent by mass.
(ii) Determine the molality of chloroform in the water sample.
🟢 Answer 1.17
(i) 1 ppm = 1 part in 10⁶
15 ppm = 15 / 10⁶ = 1.5 × 10⁻³ %
(ii) Assume 1 kg water → mass of CHCl₃ = 15 mg = 0.015 g
Molar mass CHCl₃ = 12 + 1 + 3×35.5 = 119.5 g/mol
Moles = 0.015 / 119.5 = 1.26 × 10⁻⁴ mol
Molality = moles / mass of solvent (kg) = 1.26 × 10⁻⁴ mol/kg
🔵 Question 1.18
Express the following in the scientific notation:
(i) 0.0048 (ii) 234,000 (iii) 8008 (iv) 500.0 (v) 6.0012
🟢 Answer 1.18
(i) 4.8 × 10⁻³
(ii) 2.34 × 10⁵
(iii) 8.008 × 10³
(iv) 5.000 × 10²
(v) 6.0012 × 10⁰
🔵 Question 1.19
How many significant figures are present in the following:
(i) 0.0025 (ii) 208 (iii) 5005 (iv) 126,000 (v) 500.0 (vi) 2.0034
🟢 Answer 1.19
(i) 2
(ii) 3
(iii) 4
(iv) 3 (trailing zeros not significant unless decimal is shown)
(v) 4 (trailing zero after decimal is significant)
(vi) 5
🔵 Question 1.20
Round up the following up to three significant figures:
(i) 34.216 (ii) 10.4107 (iii) 0.04597 (iv) 2808
🟢 Answer 1.20
(i) 34.2
(ii) 10.4
(iii) 0.0460
(iv) 2810
🔵 Question 1.21
The following data are obtained when dinitrogen and dioxygen react together to form different compounds:
Mass of dinitrogen Mass of dioxygen
14 g 16 g
14 g 32 g
28 g 32 g
28 g 80 g
(a) Which law of chemical combination is obeyed by the above experimental data? Give its statement.
(b) Fill in the blanks in the following conversions:
(i) 1 km = ………. mm = ………. pm
(ii) 1 mg = ………. kg = ………. ng
(iii) 1 mL = ………. L = ………. dm³
🟢 Answer 1.21
(a) Law of Multiple Proportions
▶ Statement: When two elements combine to form more than one compound, the masses of one element that combine with a fixed mass of the other are in simple whole number ratios.
✔ Here, for 14 g N: O = 16 g, 32 g → 1:2
✔ For 28 g N: O = 32 g, 80 g → 4:10 → 2:5
(b)
(i) 1 km = 1,000,000 mm = 1 × 10¹⁵ pm
(ii) 1 mg = 1 × 10⁻⁶ kg = 1 × 10⁶ ng
(iii) 1 mL = 1 × 10⁻³ L = 1 × 10⁻³ dm³
🔵 Question 1.22
If the speed of light is 3.0 × 10⁸ m s⁻¹, calculate the distance covered by light in 2.00 ns.
🟢 Answer 1.22
Time = 2.00 ns = 2.00 × 10⁻⁹ s
Speed = 3.0 × 10⁸ m/s
Distance = Speed × Time
= (3.0 × 10⁸) × (2.00 × 10⁻⁹)
= 0.60 m
🔵 Question 1.23
In a reaction:
A + B₂ → AB₂
Identify the limiting reagent, if any, in the following mixtures:
(i) 300 atoms of A + 200 molecules of B
(ii) 2 mol A + 3 mol B
(iii) 100 atoms of A + 100 molecules of B
(iv) 5 mol A + 2.5 mol B
(v) 2.5 mol A + 5 mol B
🟢 Answer 1.23
Balanced reaction: 1 A + 1 B₂ → 1 AB₂
→ Mole ratio = A : B = 1:1
(i) 300 A : 200 B ⇒ B is limiting
(ii) 2 A : 3 B ⇒ A is limiting
(iii) 100 A : 100 B ⇒ No limiting reagent
(iv) 5 A : 2.5 B ⇒ Ratio 2:1 ⇒ B is limiting
(v) 2.5 A : 5 B ⇒ Ratio 1:2 ⇒ A is limiting
🔵 Question 1.24
Dinitrogen and dihydrogen react with each other to produce ammonia:
N₂(g) + 3H₂(g) → 2NH₃(g)
(i) Calculate the mass of ammonia produced if 2.00 × 10³ g of dinitrogen reacts with 1.00 × 10³ g of dihydrogen.
(ii) Will any of the reactants remain unreacted?
(iii) If yes, which one and what would be its mass?
🟢 Answer 1.24
(i) Molar mass N₂ = 28 g, H₂ = 2 g, NH₃ = 17 g
Moles of N₂ = 2000 / 28 ≈ 71.43
Moles of H₂ = 1000 / 2 = 500
From balanced reaction: 1 mol N₂ reacts with 3 mol H₂
Required H₂ = 71.43 × 3 = 214.29 mol
Available H₂ = 500 mol → Excess
So N₂ is limiting
NH₃ produced = 2 × 71.43 = 142.86 mol
Mass = 142.86 × 17 = 2428.57 g
(ii) Yes, hydrogen remains unreacted
(iii) Used H₂ = 214.29 mol = 428.58 g
Unreacted = 1000 – 428.58 = 571.42 g
🔵 Question 1.25
How are 0.50 mol Na₂CO₃ and 0.50 M Na₂CO₃ different?
🟢 Answer 1.25
0.50 mol Na₂CO₃ means amount of substance
0.50 M Na₂CO₃ means concentration: 0.50 mol in 1 L solution
✔ So first is fixed quantity; second is per unit volume
🔵 Question 1.26
If 10 volumes of dihydrogen gas reacts with five volumes of dioxygen gas, how many volumes of water vapour would be produced?
🟢 Answer 1.26
Reaction: 2H₂ + O₂ → 2H₂O
Mole ratio: 2:1 → 2
Volume ratio = 10:5 ⇒ H₂:O₂
✔ Matches 2:1
Product = 10 volumes of H₂ → 10 volumes of H₂O
🔵 Question 1.27
Convert the following into basic units:
(i) 28.7 pm (ii) 15.15 pm (iii) 25365 mg
🟢 Answer 1.27
(i) 28.7 pm = 28.7 × 10⁻¹² m = 2.87 × 10⁻¹¹ m
(ii) 15.15 pm = 1.515 × 10⁻¹¹ m
(iii) 25365 mg = 25.365 g = 0.025365 kg
🔵 Question 1.28
Which one of the following will have the largest number of atoms?
(i) 1 g Au (ii) 1 g Na (iii) 1 g Li (iv) 1 g Cl₂
🟢 Answer 1.28
Atoms = Mass / Molar mass × Avogadro’s number
✔ Smaller molar mass → more atoms
→ Li (M = 6.94) gives maximum atoms
✅ Answer: (iii) 1 g Li
🔵 Question 1.29
Calculate the molarity of a solution of ethanol in water, in which the mole fraction of ethanol is 0.040 (assume the density of water to be one).
🟢 Answer 1.29
Mole fraction = n₁ / (n₁ + n₂)
Assume water = 1 kg = 1000 g = 55.56 mol
Let n₁ = ethanol = 0.040 × (n₁ + 55.56)
Solve: n₁ ≈ 2.31 mol
Volume = 1 L → Molarity = 2.31 M
🔵 Question 1.30
What will be the mass of one ¹²C atom in g?
🟢 Answer 1.30
1 mol ¹²C atoms = 12 g
Number of atoms in 1 mol = 6.022×10²³
Mass of one atom = 12 / 6.022×10²³ = 1.993 × 10⁻²³ g
🔵 Question 1.31
How many significant figures should be present in the answer of the following calculations?
(i) 0.02856 × 298.15 × 0.112
(ii) 5 × 5.364
(iii) 0.0125 + 0.7864 + 0.0215
🟢 Answer 1.31
(i) Least significant figures = 3 (from 0.112)
→ Final answer must have 3 significant figures
(ii) 5 has 1 significant figure, 5.364 has 4
→ Answer must have 1 significant figure
(iii) Addition: least decimal places = 4 (all have 4)
→ Final answer must have 4 decimal places
🔵 Question 1.32
Use the data given in the following table to calculate the molar mass of naturally occurring argon isotopes:
Isotope Isotopic molar mass Abundance
³⁶Ar 35.96755 g mol⁻¹ 0.337%
³⁸Ar 37.96272 g mol⁻¹ 0.063%
⁴⁰Ar 39.9624 g mol⁻¹ 99.600%
🟢 Answer 1.32
Molar mass =
(35.96755×0.337 + 37.96272×0.063 + 39.9624×99.6) / 100
= (12.124 + 2.392 + 3976.6) / 100
= 3991.116 / 100 = 39.911 g/mol
🔵 Question 1.33
Calculate the number of atoms in each of the following:
(i) 52 mol of Ar (ii) 52 u of He (iii) 52 g of He
🟢 Answer 1.33
(i) 52 mol Ar = 52 × 6.022×10²³ = 3.13 × 10²⁵ atoms
(ii) 52 u He = 1 atom of He = 1 atom
(iii) Molar mass of He = 4 g/mol
→ Moles = 52 / 4 = 13 mol
→ Atoms = 13 × 6.022×10²³ = 7.83 × 10²⁴ atoms
🔵 Question 1.34
A welding fuel gas contains carbon and hydrogen only.
Burning a small sample of it in oxygen gives 3.38 g CO₂, 0.690 g H₂O.
Volume = 10.0 L, mass = 11.6 g (STP)
Calculate:
(i) Empirical formula (ii) Molar mass of the gas (iii) Molecular formula
🟢 Answer 1.34
(i) Moles C = 3.38 g CO₂ → 3.38 / 44 = 0.0768 mol C
Moles H = 0.690 g H₂O → 0.690 / 18 = 0.0383 mol H₂O
→ H atoms = 0.0383 × 2 = 0.0766 mol
Ratio C:H ≈ 1:1 → Empirical formula = CH
(ii) Molar mass = Mass / Volume at STP
= 11.6 g / 10 L = 1.16 g/L
At STP, 1 mol = 22.4 L → Molar mass = 22.4 × 1.16 = 25.98 g/mol
(iii) Empirical mass = 13 (C + H)
n = 26 / 13 = 2 → Molecular formula = C₂H₂
🔵 Question 1.35
Calcium carbonate reacts with aqueous HCl to give CaCl₂ and CO₂:
CaCO₃ + 2HCl → CaCl₂ + CO₂ + H₂O
What mass of CaCO₃ is required to react completely with 25 mL of 0.75 M HCl?
🟢 Answer 1.35
Moles of HCl = 0.75 × 25/1000 = 0.01875 mol
From reaction: 2 mol HCl reacts with 1 mol CaCO₃
→ Moles of CaCO₃ = 0.01875 / 2 = 0.009375 mol
Mass = 0.009375 × 100 = 0.9375 g
🔵 Question 1.36
Chlorine is prepared by treating manganese dioxide with hydrochloric acid:
4HCl + MnO₂ → 2H₂O + MnCl₂ + Cl₂
How many grams of HCl are required to react with 5.0 g of manganese dioxide?
🟢 Answer 1.36
Molar mass of MnO₂ = 54.94 + 2×16 = 86.94 g/mol
Moles = 5.0 / 86.94 ≈ 0.0575 mol
From reaction: 1 mol MnO₂ → 4 mol HCl
→ Required HCl = 0.0575 × 4 = 0.23 mol
Mass = 0.23 × 36.46 = 8.39 g
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
🔷 Section A: Q1 to Q18 (1 mark each)
🔵 Question 1.
Which of the following is a pure substance?
(A) Milk
(B) Air
(C) Water
(D) Soil
🟢 Answer: (C) Water
🔵 Question 2.
Which of the following has the highest number of atoms?
(A) 1 g of H₂
(B) 1 g of O₂
(C) 1 g of Li
(D) 1 g of Cl₂
🟢 Answer: (C) 1 g of Li
🔵 Question 3.
SI unit of amount of substance is:
(A) Gram
(B) Mole
(C) Kilogram
(D) Liter
🟢 Answer: (B) Mole
🔵 Question 4.
What is the molar mass of CaCl₂?
(A) 75.5 g/mol
(B) 95.5 g/mol
(C) 110.98 g/mol
(D) 111.0 g/mol
🟢 Answer: (D) 111.0 g/mol
🔵 Question 5.
1 amu is equal to:
(A) 1.66 × 10⁻²³ g
(B) 1.66 × 10⁻²⁴ g
(C) 1.66 × 10⁻²⁷ kg
(D) 1.66 × 10⁻²⁶ kg
🟢 Answer: (C) 1.66 × 10⁻²⁷ kg
🔵 Question 6.
Which of the following obeys the Law of Multiple Proportions?
(A) H₂ and O₂
(B) CO and CO₂
(C) N₂ and NH₃
(D) NaCl and KCl
🟢 Answer: (B) CO and CO₂
🔵 Question 7.
Which prefix represents 10⁻¹⁵?
(A) micro
(B) femto
(C) nano
(D) pico
🟢 Answer: (B) femto
🔵 Question 8.
Write the number of significant figures in 500.0
🟢 Answer: 4 significant figures
🔵 Question 9.
State Avogadro’s law.
🟢 Answer:
Equal volumes of all gases under same conditions of temperature and pressure contain equal number of molecules.
🔵 Question 10.
Define empirical formula.
🟢 Answer:
It is the simplest whole-number ratio of atoms of each element present in a compound.
🔵 Question 11.
Which law of chemical combination is demonstrated by the reaction of 16 g of oxygen with 2 g of hydrogen to form 18 g of water?
🟢 Answer:
Law of Conservation of Mass
🔵 Question 12.
Give one point of difference between precision and accuracy.
🟢 Answer:
Precision refers to closeness among repeated measurements, while accuracy refers to closeness to the true value.
🔵 Question 13.
Calculate number of molecules in 1.0 g of water.
🟢 Answer:
Molar mass of H₂O = 18 g/mol
Moles = 1 / 18 = 0.0556 mol
Molecules = 0.0556 × 6.022 × 10²³ = 3.35 × 10²² molecules
🔵 Question 14.
Give the SI unit of molar mass.
🟢 Answer:
kg mol⁻¹ (commonly g mol⁻¹ in practice)
🔵 Question 15.
Write the molecular formula of a compound whose empirical formula is CH and molar mass is 26.
🟢 Answer:
Empirical mass = 13 → n = 26 / 13 = 2
Molecular formula = CH × 2 = C₂H₂
🔵 Question 16.
Which one is the limiting reagent when 5 mol H₂ reacts with 2 mol O₂?
🟢 Answer:
Reaction: 2H₂ + O₂ → 2H₂O
Mole ratio: 2:1 → 5 mol H₂ needs 2.5 mol O₂
Available = 2 mol O₂ ⇒ O₂ is limiting reagent
🔵 Question 17.
What is the mass percent of carbon in CO₂?
🟢 Answer:
Mass of carbon = 12, Molar mass = 44
% C = (12 / 44) × 100 = 27.27%
🔵 Question 18.
What is the mass of one atom of carbon-12?
🟢 Answer:
Mass = 12 / 6.022 × 10²³ = 1.993 × 10⁻²³ g
🟨 Section B: Q19 to Q23 (2 marks each)
🔵 Question 19.
Calculate the number of molecules in 5.6 litres of oxygen gas at STP.
🟢 Answer:
At STP, 1 mol gas = 22.4 L
Moles = 5.6 / 22.4 = 0.25 mol
Molecules = 0.25 × 6.022 × 10²³ = 1.506 × 10²³ molecules
🔵 Question 20.
A compound contains 92.3% carbon and 7.7% hydrogen. Its molar mass is 78 g/mol. Determine its empirical and molecular formula.
🟢 Answer:
Step 1: Assume 100 g → C = 92.3 g, H = 7.7 g
Moles:
C = 92.3 / 12.01 ≈ 7.69
H = 7.7 / 1.008 ≈ 7.65
Ratio ≈ 1:1
Empirical formula = CH
Empirical mass = 13
n = 78 / 13 = 6
Molecular formula = C₆H₆
🔵 Question 21.
Differentiate between homogeneous and heterogeneous mixtures with one example each.
🟢 Answer:
✔️ Homogeneous mixture: Same composition throughout (e.g., sugar in water)
✔️ Heterogeneous mixture: Composition varies (e.g., sand in water)
🔵 Question 22.
Calculate the number of atoms present in 0.5 mol of calcium.
🟢 Answer:
Atoms = 0.5 × 6.022 × 10²³ = 3.011 × 10²³ atoms
🔵 Question 23.
Write the postulates of Dalton’s atomic theory (any two).
🟢 Answer:
All matter consists of tiny indivisible particles called atoms.
Atoms of a given element are identical in mass and properties.
🟧 Section C: Q24 to Q28 (3 marks each)
🔵 Question 24.
Calculate the molarity of a solution prepared by dissolving 5 g of NaOH in 450 mL of solution.
(Molar mass of NaOH = 40 g/mol)
🟢 Answer:
Moles = 5 / 40 = 0.125 mol
Volume = 450 mL = 0.45 L
Molarity = 0.125 / 0.45 = 0.278 M
🔵 Question 25.
Determine the empirical formula of a compound with 40% carbon, 6.7% hydrogen, and 53.3% oxygen.
🟢 Answer:
Assume 100 g:
C = 40 g → 40 / 12.01 = 3.33 mol
H = 6.7 g → 6.7 / 1.008 = 6.65 mol
O = 53.3 g → 53.3 / 16.00 = 3.33 mol
Simplest ratio = C:H:O = 1:2:1
Empirical formula = CH₂O
🔵 Question 26.
What do you understand by limiting reagent?
Identify the limiting reagent when 6 g of Mg reacts with 6 g of HCl.
Mg + 2HCl → MgCl₂ + H₂
🟢 Answer:
✔️ Limiting reagent is the one that is completely consumed first in a reaction.
Moles of Mg = 6 / 24.3 ≈ 0.247
Moles of HCl = 6 / 36.46 ≈ 0.165
Required ratio = 1:2 → For 0.247 mol Mg → need 0.494 mol HCl
Available = 0.165 → So, HCl is limiting reagent
🔵 Question 27.
Explain the law of definite proportions with one example.
🟢 Answer:
✔️ A given compound always contains the same elements in the same fixed ratio by mass.
Example: In water, H:O = 2:16 = 1:8 always, regardless of source.
🔵 Question 28.
Calculate the mass percent of each element in CH₃COOH.
(Atomic masses: C = 12, H = 1, O = 16)
🟢 Answer:
Molar mass = 2×12 + 4×1 + 2×16 = 60 g/mol
% C = (24 / 60) × 100 = 40%
% H = (4 / 60) × 100 = 6.67%
% O = (32 / 60) × 100 = 53.33%
🟦 Section D: Q29 to Q31 (4 marks each – Case Based)
🔵 Question 29.
Read the passage and answer the following:
A student prepares an aqueous solution by dissolving 10.0 g of glucose (C₆H₁₂O₆) in 90.0 g of water.
(Atomic masses: C = 12, H = 1, O = 16)
(i) Calculate the number of moles of glucose.
(ii) Calculate the molality of the solution.
(iii) Find the mass % of glucose.
(iv) How many molecules of glucose are present?
🟢 Answer:
(i) Molar mass of C₆H₁₂O₆ = (6×12 + 12×1 + 6×16) = 180 g/mol
Moles = 10.0 / 180 = 0.0556 mol
(ii) Molality = Moles / Mass of solvent (kg) = 0.0556 / 0.090 = 0.618 mol/kg
(iii) Mass % = (10 / (10 + 90)) × 100 = 10%
(iv) Molecules = 0.0556 × 6.022 × 10²³ = 3.35 × 10²² molecules
🔵 Question 30.
Read the paragraph and answer:
Calcium carbonate decomposes on heating to give calcium oxide and carbon dioxide:
CaCO₃ → CaO + CO₂
A sample of limestone gave 2.2 g of CO₂.
(i) Write the balanced equation.
(ii) Calculate moles of CO₂ evolved.
(iii) Find the mass of CaCO₃ decomposed.
(iv) Name the law illustrated by this reaction.
🟢 Answer:
(i) CaCO₃ → CaO + CO₂ ✅ Balanced
(ii) Moles CO₂ = 2.2 / 44 = 0.05 mol
(iii) Moles CaCO₃ = 0.05 mol
Mass = 0.05 × 100 = 5.0 g
(iv) Law of Conservation of Mass
🔵 Question 31.
Study the data and answer:
Compound A contains 63.15% silver, 8.79% nitrogen, and 28.06% oxygen.
Molar mass of compound is 169.87 g/mol.
(i) Calculate empirical formula.
(ii) Determine molecular formula.
🟢 Answer:
(i) Moles:
Ag = 63.15 / 107.87 ≈ 0.585
N = 8.79 / 14.01 ≈ 0.627
O = 28.06 / 16.00 ≈ 1.754
Divide by 0.585 → Ag:N:O ≈ 1:1:3
Empirical formula = AgNO₃
(ii) Empirical mass = 107.87 + 14.01 + 48 = 169.88
→ Molecular formula = AgNO₃
🟪 Section E: Q32 to Q35 (5 marks each – Long Answers)
🔵 Question 32.
(a) Define mole.
(b) What is Avogadro’s number?
(c) Calculate:
(i) Mass of 2.5 mol of CO₂
(ii) Volume occupied by 0.5 mol of gas at STP
(iii) Number of molecules in 11.2 L of CH₄ at STP
🟢 Answer:
(a) Mole is the amount of substance that contains as many particles as there are atoms in 12 g of carbon-12.
(b) Avogadro’s number = 6.022 × 10²³ particles/mol
(c)
(i) Molar mass CO₂ = 44 g/mol → Mass = 2.5 × 44 = 110 g
(ii) 1 mol gas = 22.4 L → Volume = 0.5 × 22.4 = 11.2 L
(iii) Mole = 11.2 / 22.4 = 0.5 mol
→ Molecules = 0.5 × 6.022×10²³ = 3.011 × 10²³ molecules
🔵 Question 33.
A compound contains 54.5% C, 9.1% H, and 36.4% O. Molar mass = 88 g/mol.
Determine:
(a) Empirical formula
(b) Molecular formula
🟢 Answer:
Assume 100 g:
C = 54.5 g → 54.5 / 12.01 ≈ 4.54 mol
H = 9.1 g → 9.1 / 1.008 ≈ 9.03 mol
O = 36.4 g → 36.4 / 16 ≈ 2.275 mol
Divide by 2.275 → C:H:O ≈ 2:4:1
Empirical formula = C₂H₄O
Empirical mass = 44
n = 88 / 44 = 2
Molecular formula = C₄H₈O₂
🔵 Question 34.
(a) Define molarity and molality.
(b) A solution is prepared by dissolving 20 g of NaOH in 250 mL of solution. Calculate its:
(i) Molarity
(ii) Molality (assume density of water = 1 g/mL)
🟢 Answer:
(a)
✔ Molarity = moles of solute / volume of solution in L
✔ Molality = moles of solute / mass of solvent in kg
(b)
Moles of NaOH = 20 / 40 = 0.5 mol
(i) Volume = 250 mL = 0.25 L → M = 0.5 / 0.25 = 2.0 M
(ii) Solvent mass ≈ 250 – 20 = 230 g = 0.230 kg
Molality = 0.5 / 0.230 = 2.17 mol/kg
🔵 Question 35.
(a) State the law of multiple proportions with one example.
(b) 2.5 g of a compound contains 0.9 g calcium, 0.7 g sulphur, and 0.9 g oxygen. Determine the empirical formula.
🟢 Answer:
(a) When two elements combine to form more than one compound, the masses of one element that combine with a fixed mass of the other bear a simple whole number ratio.
Example: CO and CO₂ — 12 g C combines with 16 g O in CO and 32 g in CO₂ → 1:2
(b) Moles:
Ca = 0.9 / 40 = 0.0225
S = 0.7 / 32.06 = 0.0218
O = 0.9 / 16 = 0.0563
Divide by smallest → Ca:S:O ≈ 1:1:2.58 ≈ 1:1:3
Empirical formula = CaSO₃
————————————————————————————————————————————————————————————————————————————
