Class 11 : Chemistry (In English) – Chapter 7: Redox Reaction
EXPLANATION & SUMMARY

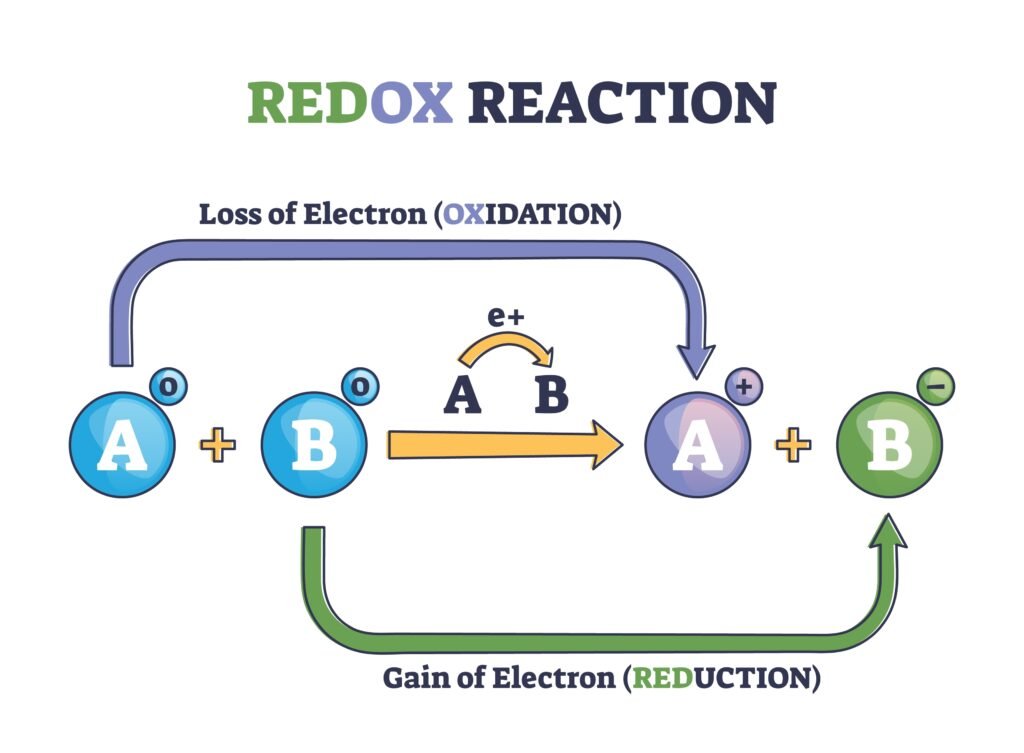
🔷 1. Introduction to Redox Reactions
Chemical reactions involving transfer of electrons are called redox reactions. The term ‘redox’ is a short form of reduction–oxidation reaction.
In every redox reaction:

🔹 One species loses electrons → Oxidation
🔹 Another species gains electrons → Reduction
These processes occur simultaneously.
🔷 2. Classical Definitions
🔸 Oxidation:
Addition of oxygen
Removal of hydrogen
Loss of electrons
Example:
2Mg + O₂ → 2MgO (Magnesium is oxidised)
🔸 Reduction:
Addition of hydrogen
Removal of oxygen
Gain of electrons
Example:
CuO + H₂ → Cu + H₂O (CuO is reduced)
🔷 3. Redox Reaction (Simultaneous Oxidation & Reduction)
Example:
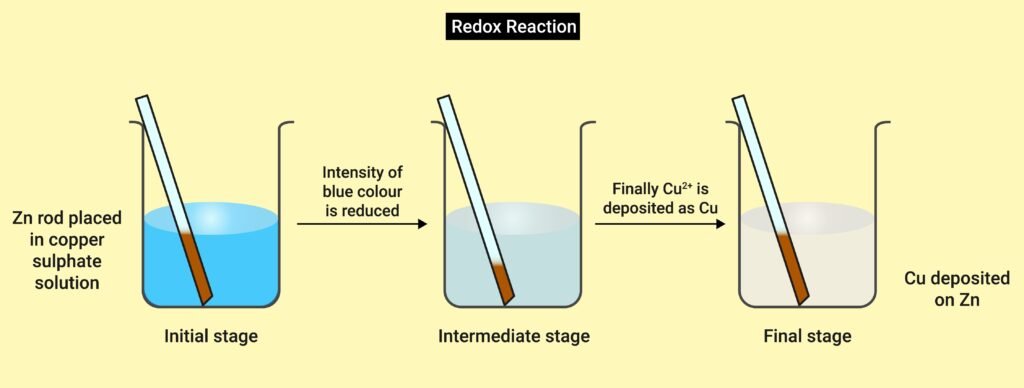
Zn + Cu²⁺ → Zn²⁺ + Cu
Here:
Zn loses electrons → Oxidised
Cu²⁺ gains electrons → Reduced
This is a redox reaction.
🔷 4. Oxidising and Reducing Agents
🔹 Oxidising Agent (Oxidant):
It accepts electrons and gets reduced.
e.g. O₂, Cl₂, KMnO₄
🔹 Reducing Agent (Reductant):
It donates electrons and gets oxidised.
e.g. H₂, Na, Zn
🔷 5. Electron Transfer Concept
Redox reactions can be described in terms of electron transfer:
🔹 Oxidation = Loss of e⁻
🔹 Reduction = Gain of e⁻
Example:
Fe²⁺ + Cu²⁺ → Fe³⁺ + Cu⁺
Fe²⁺ → Fe³⁺ = oxidation (loss of 1 e⁻)
Cu²⁺ → Cu⁺ = reduction (gain of 1 e⁻)
🔷 6. Oxidation Number
Definition: Oxidation number is the charge an atom would carry if all bonds were ionic.
Used to:
✔ Track redox changes
✔ Balance redox reactions
🔸 Rules:
Free element = 0 (e.g. O₂, N₂)
Monoatomic ion = Charge on ion
Oxygen = −2 (except in peroxides = −1)
Hydrogen = +1 (except with metals = −1)
Fluorine = −1 always
Sum of O.N. in neutral compound = 0
In ion = equal to ionic charge
🔷 7. Redox Reactions via Oxidation Number
Change in oxidation number tells us:
✔ Which atom is oxidised
✔ Which is reduced
Example:
Zn + H₂SO₄ → ZnSO₄ + H₂
Zn: 0 → +2 (oxidised)
H⁺: +1 → 0 (reduced)
🔷 8. Types of Redox Reactions
🔸 1. Combination Reactions:
Two or more reactants → One product
e.g. 2H₂ + O₂ → 2H₂O
🔸 2. Decomposition Reactions:
One compound → Simpler substances
e.g. 2KClO₃ → 2KCl + 3O₂
🔸 3. Displacement Reactions:
One element replaces another
e.g. Zn + CuSO₄ → ZnSO₄ + Cu
🔸 4. Disproportionation Reactions:
Same element undergoes both oxidation and reduction
e.g. Cl₂ + H₂O → HCl + HOCl
🔷 9. Balancing Redox Reactions
Two methods:
🔸 A. Oxidation Number Method:
Step-wise process:
Write unbalanced equation
Assign oxidation numbers
Identify changes
Equalise increase and decrease in O.N.
Balance atoms
Balance O using H₂O, H using H⁺
For basic medium: Add OH⁻
🔸 B. Half-Reaction Method:
Steps:
Split into oxidation and reduction half
Balance atoms and charges
Equalise electrons lost/gained
Combine both half-reactions
Example (Acidic medium):
MnO₄⁻ + Fe²⁺ → Mn²⁺ + Fe³⁺
→ MnO₄⁻ + 8H⁺ + 5e⁻ → Mn²⁺ + 4H₂O
→ Fe²⁺ → Fe³⁺ + e⁻
Balance and combine.
🔷 10. Redox Reactions in Electrochemistry
Redox reactions form the basis of:
🔋 Electrochemical cells
✔ Galvanic cells (spontaneous)
✔ Electrolytic cells (non-spontaneous)
e.g. Zn + Cu²⁺ → Zn²⁺ + Cu
Used in batteries, electroplating, etc.
🔷 11. Redox Reactions and Titrations
Redox reactions are used in:
📍 Redox titrations – to determine concentration of oxidising/reducing agents
Examples:
✔ KMnO₄ vs FeSO₄
✔ Iodometry: I₂ formed and titrated with Na₂S₂O₃
Indicators used: self-indicating (like KMnO₄) or starch (in iodometry)
🔷 12. Redox Reactions in Industry
Examples:
🔹 Extraction of metals (Al, Zn used as reducing agents)
🔹 Chlor-alkali process
🔹 Bleaching powder production
🔹 Combustion of fuels
🔷 13. Disproportionation Reaction (Special Type)
In a disproportionation reaction, an element in a single oxidation state gets simultaneously oxidised and reduced.
Example:
2H₂O₂ → 2H₂O + O₂
(O.N. of O: −1 → −2 and 0)
🔷 14. Limitations of Classical Concepts
Earlier definitions (O₂ addition/H₂ removal) fail in ionic or complex reactions. Hence, electron transfer and oxidation number concepts are more universal.

🟢 SUMMARY
Redox reactions are chemical processes where oxidation and reduction occur simultaneously. Oxidation refers to loss of electrons, while reduction is gain of electrons. These reactions involve transfer of electrons between atoms or ions.
Oxidising agents gain electrons (and get reduced), while reducing agents lose electrons (and get oxidised).
Oxidation numbers are useful to identify oxidation/reduction events in complex reactions. Based on this, redox reactions are classified into four main types:
Combination
Decomposition
Displacement
Disproportionation
Balancing redox reactions can be done by two systematic methods:
Oxidation Number Method
Half-Reaction Method
Redox reactions are central to electrochemistry, including batteries, galvanic and electrolytic cells. They are also applied in titrations, where concentrations of unknown solutions are calculated using known redox pairs (e.g. KMnO₄ vs Fe²⁺).
Disproportionation reactions involve a single element undergoing both oxidation and reduction. The classical definitions of redox fail in certain reactions, making oxidation number and electron transfer definitions more accurate and universal.
Redox reactions are vital in industrial processes, metallurgy, environmental chemistry, and biological systems like respiration and photosynthesis.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
🟦 Q7.1: Assign oxidation number to the underlined elements in each of the following species:
(a) NaH
(b) H₂O₂
(c) NaClO₃
(d) Mg(NO₃)₂
(e) Cr₂O₇²⁻
(f) CH₂Cl₂
(g) CuSO₄
(h) NH₄⁺
(i) HNO₃
(j) KMnO₄
🟢 Answer:
We assign oxidation numbers using standard rules:
(a) H in NaH = −1
(b) O in H₂O₂ = −1
(c) Cl in NaClO₃ = +5
(d) N in NO₃⁻ = +5
(e) Cr in Cr₂O₇²⁻ = +6
(f) Cl in CH₂Cl₂ = −1 (each), H = +1, C = 0
(g) Cu in CuSO₄ = +2
(h) N in NH₄⁺ = −3
(i) N in HNO₃ = +5
(j) Mn in KMnO₄ = +7
🟦 Q7.2: What are the oxidation numbers of the underlined elements in the following compounds?
(a) K₂Cr₂O₇
(b) K₂CrO₄
(c) Cr₂O₃
(d) CrO₃
🟢 Answer:
(a) Cr in K₂Cr₂O₇ = +6
(b) Cr in K₂CrO₄ = +6
(c) Cr in Cr₂O₃ = +3
(d) Cr in CrO₃ = +6
🟦 Q7.3: Name the oxidising agent and the reducing agent in the following reactions:
(a) 2H₂ + O₂ → 2H₂O
(b) CuO + H₂ → Cu + H₂O
(c) MnO₂ + 4HCl → MnCl₂ + Cl₂ + 2H₂O
(d) Pb₃O₄ + 8HCl → 3PbCl₂ + Cl₂ + 4H₂O
🟢 Answer:
(a)
Oxidising Agent = O₂
Reducing Agent = H₂
(b)
Oxidising Agent = CuO
Reducing Agent = H₂
(c)
Oxidising Agent = MnO₂
Reducing Agent = HCl
(d)
Oxidising Agent = Pb₃O₄
Reducing Agent = HCl
🟦 Q7.4: What is the oxidation number of phosphorus in the following:
(a) H₃PO₃
(b) HPO₃²⁻
(c) H₄P₂O₇
(d) P₂O₅
🟢 Answer:
(a) P in H₃PO₃ = +3
(b) P in HPO₃²⁻ = +5
(c) P in H₄P₂O₇ = +5
(d) P in P₂O₅ = +5
🟦 Q7.5: Write the name of oxidising and reducing agents in the following redox reactions:
(a) Zn + Cu²⁺ → Zn²⁺ + Cu
(b) 2Na + Cl₂ → 2NaCl
(c) 2Fe²⁺ + Cl₂ → 2Fe³⁺ + 2Cl⁻
🟢 Answer:
(a)
Oxidising Agent = Cu²⁺
Reducing Agent = Zn
(b)
Oxidising Agent = Cl₂
Reducing Agent = Na
(c)
Oxidising Agent = Cl₂
Reducing Agent = Fe²⁺
🟦 Q7.6: Compute the oxidation number of sulphur, chromium and nitrogen in H₂SO₅, Cr₂O₇²⁻ and NO₃⁻.
🟢 Answer:
(a) Sulphur in H₂SO₅ = +6
(b) Chromium in Cr₂O₇²⁻ = +6
(c) Nitrogen in NO₃⁻ = +5
🟦 Q7.7: Identify the substances oxidised and the substances reduced in the following reactions:
(a) 2Na + O₂ → Na₂O₂
(b) CuO + H₂ → Cu + H₂O
🟢 Answer:
(a)
Oxidised: Na
Reduced: O₂
(b)
Oxidised: H₂
Reduced: CuO
🟦 Q7.8: Classify the following as combination, decomposition, displacement and disproportionation reactions:
(a) 2Mg + O₂ → 2MgO
(b) 2H₂O → 2H₂ + O₂
(c) Cu + 2AgNO₃ → Cu(NO₃)₂ + 2Ag
(d) Cl₂ + 2NaOH → NaCl + NaClO + H₂O
🟢 Answer:
(a) Combination reaction
(b) Decomposition reaction
(c) Displacement reaction
(d) Disproportionation reaction
🟦 Q7.9: Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them:
(a) BaCl₂ + H₂SO₄ → BaSO₄ + 2HCl
(b) 3Mg + N₂ → Mg₃N₂
(c) 4Na + O₂ → 2Na₂O
(d) 2K + 2H₂O → 2KOH + H₂
(e) CuSO₄ + Zn → Cu + ZnSO₄
🟢 Answer:
(a) Not a redox reaction
(b) Redox
Oxidising Agent = N₂
Reducing Agent = Mg
(c) Redox
Oxidising Agent = O₂
Reducing Agent = Na
(d) Redox
Oxidising Agent = H₂O
Reducing Agent = K
(e) Redox
Oxidising Agent = CuSO₄
Reducing Agent = Zn
🟦 Q7.10: Define the terms oxidation and reduction in the classical sense.
🟢 Answer:
📌 Oxidation is defined as:
The addition of oxygen, or
The removal of hydrogen.
📌 Reduction is defined as:
The removal of oxygen, or
The addition of hydrogen.
Example:
H₂S + Cl₂ → 2HCl + S
In this, H₂S is oxidised (loss of H), and Cl₂ is reduced (gain of H).
💡 Q7.11
Why does fluorine not show any positive oxidation state?
✅ Answer
📌 Fluorine is the most electronegative element in the periodic table.
📌 It has an electronic configuration of 1s² 2s² 2p⁵.
📌 Due to its small size and high electronegativity, it can only gain electrons, not lose them.
📌 Hence, it always exhibits an oxidation state of –1, never positive.
💡 Q7.12
Calculate the oxidation number of Cr in Cr₂O₇²⁻.
✅ Answer
Let oxidation number of Cr = x
There are 2 Cr atoms and 7 O atoms.
Oxidation number of O = –2
Total charge = –2
📌 2x + 7(–2) = –2
⇒ 2x – 14 = –2
⇒ 2x = +12
⇒ x = +6
🔷 So, the oxidation number of Cr is +6.
💡 Q7.13
Balance the following redox reaction in acidic medium:
Cr₂O₇²⁻ + I⁻ → Cr³⁺ + I₂
✅ Answer
📌 Step 1: Identify oxidation & reduction
Cr changes from +6 → +3 (Reduction)
I changes from –1 → 0 (Oxidation)
📌 Step 2: Write half reactions
Oxidation: 2I⁻ → I₂ + 2e⁻
Reduction: Cr₂O₇²⁻ + 14H⁺ + 6e⁻ → 2Cr³⁺ + 7H₂O
📌 Step 3: Multiply to equalize electrons
Oxidation ×3: 6I⁻ → 3I₂ + 6e⁻
Add both:
Cr₂O₇²⁻ + 14H⁺ + 6I⁻ → 2Cr³⁺ + 7H₂O + 3I₂
🔷 Final Balanced Reaction:
Cr₂O₇²⁻ + 14H⁺ + 6I⁻ → 2Cr³⁺ + 7H₂O + 3I₂
💡 Q7.14
What is the oxidation number of each element in KMnO₄?
✅ Answer
Let oxidation number of Mn = x
K = +1
O = –2
Equation:
+1 + x + 4(–2) = 0
⇒ 1 + x – 8 = 0
⇒ x = +7
🔷 Oxidation numbers:
K = +1, Mn = +7, O = –2
💡 Q7.15
Identify the oxidizing and reducing agents in the reaction:
Zn + Cu²⁺ → Zn²⁺ + Cu
✅ Answer
📌 Zn loses 2 electrons → Oxidation
Zn is the reducing agent
📌 Cu²⁺ gains 2 electrons → Reduction
Cu²⁺ is the oxidizing agent
💡 Q7.16
Balance the following redox reaction in basic medium:
MnO₄⁻ + SO₃²⁻ → MnO₂ + SO₄²⁻
✅ Answer
📌 Oxidation: SO₃²⁻ → SO₄²⁻ + 2e⁻
📌 Reduction: MnO₄⁻ + 2H₂O + 3e⁻ → MnO₂ + 4OH⁻
📌 Multiply and add:
3SO₃²⁻ → 3SO₄²⁻ + 6e⁻
2MnO₄⁻ + 4H₂O + 6e⁻ → 2MnO₂ + 8OH⁻
📌 Add both:
2MnO₄⁻ + 3SO₃²⁻ + 4H₂O → 2MnO₂ + 3SO₄²⁻ + 8OH⁻
💡 Q7.17
Define disproportionation reaction with example.
✅ Answer
📌 A disproportionation reaction is a type of redox reaction where the same species undergoes both oxidation and reduction.
Example:
2H₂O₂ → 2H₂O + O₂
(Oxidation: –1 to 0, Reduction: –1 to –2)
💡 Q7.18
Why is the reaction between H₂ and Cl₂ considered a redox reaction?
✅ Answer
📌 In H₂ + Cl₂ → 2HCl:
H is oxidized: 0 → +1
Cl is reduced: 0 → –1
🔷 Since oxidation and reduction occur simultaneously, it is a redox reaction.
💡 Q7.19
Write the balanced redox reaction for the reaction of hydrogen peroxide with acidified potassium iodide.
✅ Answer
📌 H₂O₂ + 2H⁺ + 2I⁻ → I₂ + 2H₂O
📌 H₂O₂ acts as an oxidizing agent, converting I⁻ → I₂
💡 Q7.20
Find the oxidation number of each element in H₂SO₄.
✅ Answer
Let oxidation number of S = x
H = +1, O = –2
📌 2(+1) + x + 4(–2) = 0
⇒ 2 + x – 8 = 0
⇒ x = +6
🔷 Oxidation Numbers:
H = +1, S = +6, O = –2
💡 Q7.21
Justify that the reaction:
4Na(s) + O₂(g) → 2Na₂O(s)
is a redox reaction.
✅ Answer
📌 Step 1: Assign oxidation numbers
Na in Na (elemental) = 0
O in O₂ = 0
Na in Na₂O = +1
O in Na₂O = –2
📌 Step 2: Identify changes
Na: 0 → +1 → Oxidation
O: 0 → –2 → Reduction
🔷 Conclusion: One species is oxidized and another is reduced, so this is a redox reaction.
💡 Q7.22
What is the oxidation number of Cl in the following?
(a) Cl₂O₇
(b) ClO₄⁻
(c) ClO₃⁻
(d) ClO₂⁻
(e) ClO⁻
✅ Answer
Let x be the oxidation number of Cl
O = –2
(a) Cl₂O₇:
2x + 7(–2) = 0
⇒ 2x – 14 = 0
⇒ x = +7
(b) ClO₄⁻:
x + 4(–2) = –1
⇒ x – 8 = –1
⇒ x = +7
(c) ClO₃⁻:
x + 3(–2) = –1
⇒ x – 6 = –1
⇒ x = +5
(d) ClO₂⁻:
x + 2(–2) = –1
⇒ x – 4 = –1
⇒ x = +3
(e) ClO⁻:
x – 2 = –1
⇒ x = +1
💡 Q7.23
Identify the oxidant and reductant in the following reaction:
MnO₄⁻ + C₂O₄²⁻ → Mn²⁺ + CO₂
✅ Answer
📌 Mn in MnO₄⁻ = +7
Mn in Mn²⁺ = +2 → Reduction (MnO₄⁻ is oxidant)
📌 C in C₂O₄²⁻ = +3
C in CO₂ = +4 → Oxidation (C₂O₄²⁻ is reductant)
🔷 Oxidant: MnO₄⁻
🔷 Reductant: C₂O₄²⁻
💡 Q7.24
What is meant by the term auto-oxidation?
✅ Answer
📌 Auto-oxidation is a process in which a substance undergoes simultaneous oxidation and reduction.
📌 Example:
H₂O₂ → H₂O + O₂
Here, oxygen in H₂O₂ has oxidation number –1
Becomes –2 in H₂O → Reduction
Becomes 0 in O₂ → Oxidation
🔷 Hence, it’s called auto-oxidation or disproportionation.
💡 Q7.25
Balance the following redox reaction in acidic medium:
MnO₄⁻ + Fe²⁺ → Mn²⁺ + Fe³⁺
✅ Answer
📌 Oxidation: Fe²⁺ → Fe³⁺ + e⁻
📌 Reduction: MnO₄⁻ + 8H⁺ + 5e⁻ → Mn²⁺ + 4H₂O
📌 Multiply oxidation half by 5:
5Fe²⁺ → 5Fe³⁺ + 5e⁻
📌 Add both:
MnO₄⁻ + 5Fe²⁺ + 8H⁺ → Mn²⁺ + 5Fe³⁺ + 4H₂O
💡 Q7.26
Assign oxidation numbers to each element in HNO₃.
✅ Answer
Let oxidation number of N = x
H = +1, O = –2
📌 +1 + x + 3(–2) = 0
⇒ 1 + x – 6 = 0
⇒ x = +5
🔷 Oxidation Numbers:
H = +1, N = +5, O = –2
💡 Q7.27
Differentiate between oxidation and reduction in terms of electron transfer.
✅ Answer
📌 Oxidation:
Loss of electrons → Increase in oxidation number
Example: Na → Na⁺ + e⁻
📌 Reduction:
Gain of electrons → Decrease in oxidation number
Example: Cl₂ + 2e⁻ → 2Cl⁻
💡 Q7.28
What is the oxidation number of Mn in Mn₃O₄?
✅ Answer
Let x = oxidation number of Mn
Mn₃O₄ = 3Mn + 4O → net charge = 0
3x + 4(–2) = 0
⇒ 3x – 8 = 0
⇒ x = +8/3
🔷 Oxidation state of Mn is fractional: +8/3
(average of Mn⁺² and Mn⁺⁴)
💡 Q7.29
Balance the redox reaction:
Cl₂ → Cl⁻ + ClO₃⁻
✅ Answer
📌 Oxidation: Cl₂ → ClO₃⁻
📌 Reduction: Cl₂ → Cl⁻
📌 Balanced Equation:
3Cl₂ + 3H₂O → 5Cl⁻ + ClO₃⁻ + 6H⁺
💡 Q7.30
Which oxidation state is exhibited by nitrogen in the following compounds?
(a) N₂
(b) NO
(c) NO₂
(d) NH₃
✅ Answer
(a) N₂: 0 (elemental form)
(b) NO: +2
(c) NO₂: +4
(d) NH₃: –3
💡 Q7.31
Balance the following redox reaction using the ion-electron method:
Cr₂O₇²⁻ + SO₂ → Cr³⁺ + SO₄²⁻ (in acidic medium)
✅ Answer
📌 Oxidation:
SO₂ → SO₄²⁻
S: +4 → +6
Change = 2 e⁻ lost
📌 Reduction:
Cr₂O₇²⁻ → Cr³⁺
Cr: +6 → +3
Change = 3 e⁻ per Cr, total 6 e⁻ (2 Cr)
📌 Balance electrons:
Multiply oxidation by 3 and reduction by 1
🔁 Balanced Equation:
Cr₂O₇²⁻ + 3SO₂ + 2H₂O → 2Cr³⁺ + 3SO₄²⁻ + 4H⁺
💡 Q7.32
Which among the following does not show disproportionation reaction?
(A) Cl₂
(B) F₂
(C) Br₂
(D) I₂
✅ Answer
📌 Fluorine (F₂) is the most electronegative element and cannot show positive oxidation state.
So, it cannot undergo disproportionation.
✔️ Correct Answer: (B) F₂
💡 Q7.33
Name a compound in which nitrogen shows +1 oxidation state.
✅ Answer
📌 Example: N₂O (Nitrous oxide or laughing gas)
Let x = oxidation number of N
2x + (–2) = 0
⇒ x = +1
✔️ Nitrogen in N₂O has oxidation state +1
💡 Q7.34
Write two examples of disproportionation reactions.
✅ Answer
📌 Example 1:
2H₂O₂ → 2H₂O + O₂
(Oxidation: –1 to 0, Reduction: –1 to –2)
📌 Example 2:
Cl₂ + H₂O → HCl + HOCl
(Cl: 0 → –1 and +1)
💡 Q7.35
What is meant by a redox couple?
✅ Answer
📌 A redox couple refers to an oxidized and a reduced form of the same species involved in a redox reaction.
📌 Examples:
Fe³⁺ / Fe²⁺
Cu²⁺ / Cu
🔷 A redox couple helps define electrode potentials.
💡 Q7.36
What is a reducing agent?
✅ Answer
📌 A reducing agent is a substance that donates electrons to another species and gets oxidized in the process.
📌 Example:
Zn + Cu²⁺ → Zn²⁺ + Cu
Zn acts as reducing agent.
💡 Q7.37
Identify the redox changes in the following reaction:
Zn + CuSO₄ → ZnSO₄ + Cu
✅ Answer
📌 Zn: 0 → +2 (Oxidation)
📌 Cu²⁺: +2 → 0 (Reduction)
🔷 Zn is oxidized → reducing agent
🔷 Cu²⁺ is reduced → oxidizing agent
💡 Q7.38
Name a compound in which oxygen has a positive oxidation state.
✅ Answer
📌 Example: OF₂ (Oxygen difluoride)
Oxygen = +2
Fluorine = –1 (being more electronegative)
📌 1x + 2(–1) = 0
⇒ x = +2
✔️ So, oxygen has +2 oxidation state in OF₂
💡 Q7.39
Which species undergoes oxidation in the reaction:
H₂S + Cl₂ → S + HCl
✅ Answer
📌 Sulfur in H₂S: –2
Sulfur in S: 0 → Oxidation
📌 Chlorine: 0 → –1 → Reduction
✔️ Oxidized species: H₂S (Sulfur)
💡 Q7.40
Why is H₂O₂ both an oxidizing and a reducing agent?
✅ Answer
📌 Oxidation number of O in H₂O₂ = –1
📌 Acts as oxidizing agent:
H₂O₂ + 2H⁺ + 2e⁻ → 2H₂O
(–1 to –2)
📌 Acts as reducing agent:
H₂O₂ → O₂ + 2H⁺ + 2e⁻
(–1 to 0)
✔️ Hence, H₂O₂ shows dual behavior.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
🔵 Q1. What is the oxidation number of chlorine in NaClO₄?
(A) +7
(B) +5
(C) +3
(D) +1
✅ Answer: (A) +7
🔵 Q2. Identify the reducing agent in the reaction:
Zn + CuSO₄ → ZnSO₄ + Cu
(A) Zn
(B) Cu²⁺
(C) SO₄²⁻
(D) Cu
✅ Answer: (A) Zn
🔵 Q3. Which of the following is not a redox reaction?
(A) H₂ + Cl₂ → 2HCl
(B) BaCl₂ + H₂SO₄ → BaSO₄ + 2HCl
(C) CuO + H₂ → Cu + H₂O
(D) Fe₂O₃ + 3CO → 2Fe + 3CO₂
✅ Answer: (B) BaCl₂ + H₂SO₄ → BaSO₄ + 2HCl
🔵 Q4. The equivalent weight of a substance in a redox reaction depends on:
(A) Molecular weight only
(B) Atomic number
(C) Number of electrons lost or gained
(D) Volume of gas evolved
✅ Answer: (C) Number of electrons lost or gained
🔵 Q5. Oxidation involves:
(A) Gain of electrons
(B) Gain of protons
(C) Loss of protons
(D) Loss of electrons
✅ Answer: (D) Loss of electrons
🔵 Q6. In acidic medium, the oxidation number of Mn in MnO₄⁻ changes to what value in Mn²⁺?
(A) +4
(B) +6
(C) +2
(D) 0
✅ Answer: (C) +2
🔵 Q7. Assertion (A): Oxidation number of carbon in CH₄ is –4.
Reason (R): Carbon is more electronegative than hydrogen.
(A) A and R both are correct, and R is the correct explanation of A.
(B) A and R both are correct, but R is not the correct explanation of A.
(C) A is correct, R is incorrect.
(D) A is incorrect, R is correct.
✅ Answer: (C) A is correct, R is incorrect.
🔵 Q8. Which of the following reactions is a disproportionation reaction?
(A) Cl₂ + 2NaBr → 2NaCl + Br₂
(B) 2F₂ + 2OH⁻ → OF₂ + 2F⁻ + H₂O
(C) Zn + HCl → ZnCl₂ + H₂
(D) Fe + CuSO₄ → FeSO₄ + Cu
✅ Answer: (B) 2F₂ + 2OH⁻ → OF₂ + 2F⁻ + H₂O
🔵 Q9. What is the oxidation number of Fe in K₄[Fe(CN)₆]?
(A) +3
(B) +2
(C) +4
(D) 0
✅ Answer: (B) +2
🔵 Q10. What is the change in oxidation number of sulfur in the reaction:
S₈ + 12O₂ → 8SO₃
(A) 0 to +6
(B) 0 to +4
(C) –2 to +6
(D) +2 to +6
✅ Answer: (A) 0 to +6
🔵 Q11. Very Short Answer: Define redox reaction.
✅ Answer: A redox reaction is a chemical reaction in which oxidation and reduction occur simultaneously.
🔵 Q12. Very Short Answer: What is meant by oxidation number?
✅ Answer: Oxidation number is the hypothetical charge assigned to an atom assuming that the bonding is purely ionic.
🔵 Q13. Very Short Answer: Name one oxidizing agent used in acidic medium.
✅ Answer: Potassium permanganate (KMnO₄)
🔵 Q14. Very Short Answer: Give one example of a disproportionation reaction.
✅ Answer: 2H₂O₂ → 2H₂O + O₂
🔵 Q15. Very Short Answer: What is the oxidation number of nitrogen in NO₃⁻?
✅ Answer: +5
🔵 Q16. Case-based MCQ:
Case: Consider the redox reaction:
Cr₂O₇²⁻ + 14H⁺ + 6e⁻ → 2Cr³⁺ + 7H₂O
Q: What is the change in oxidation number of Cr?
(A) 0 to +3
(B) +6 to +3
(C) +3 to +6
(D) –2 to 0
✅ Answer: (B) +6 to +3
🔵 Q17. Case-based MCQ:
Case: In the reaction Cu + 2AgNO₃ → Cu(NO₃)₂ + 2Ag,
Q: What role does copper play?
(A) Reducing agent
(B) Oxidizing agent
(C) Catalyst
(D) Neutral compound
✅ Answer: (A) Reducing agent
🔵 Q18. Case-based MCQ:
Case: The following reaction occurs in acidic solution:
MnO₄⁻ + 5Fe²⁺ + 8H⁺ → Mn²⁺ + 5Fe³⁺ + 4H₂O
Q: How many electrons are transferred in total?
(A) 3
(B) 4
(C) 5
(D) 6
✅ Answer: (C) 5
🔷 Q19. Write the oxidation half-reaction and the reduction half-reaction for the reaction:
Zn + Cu²⁺ → Zn²⁺ + Cu
📌 Answer:
🔹 Oxidation half-reaction:
Zn → Zn²⁺ + 2e⁻
🔹 Reduction half-reaction:
Cu²⁺ + 2e⁻ → Cu
🔷 Q20. Calculate the oxidation number of Mn in MnO₄⁻.
📌 Answer:
Let oxidation number of Mn = x
Oxygen = –2 × 4 = –8
Net charge on MnO₄⁻ = –1
So,
x – 8 = –1
⇒ x = +7
✅ Oxidation number of Mn = +7
🔷 Q21. What is disproportionation reaction? Give an example.
📌 Answer:
🔹 A disproportionation reaction is a type of redox reaction in which a single substance undergoes both oxidation and reduction.
🔹 Example:
2H₂O₂ → 2H₂O + O₂
Here, O in H₂O₂ has oxidation number –1. It gets oxidised to 0 in O₂ and reduced to –2 in H₂O.
🔷 Q22. Differentiate between oxidising agent and reducing agent.
📌 Answer:
🔹 Oxidising agent: Accepts electrons and gets reduced.
🔹 Reducing agent: Donates electrons and gets oxidised.
🟢 Example:
In the reaction: CuO + H₂ → Cu + H₂O
• CuO is oxidising agent
• H₂ is reducing agent
🔷 Q23. What is meant by the term oxidation number? How is it different from valency?
📌 Answer:
🔹 Oxidation number: It is the apparent charge assigned to an atom in a molecule/ion.
🔹 Valency: It is the combining capacity of an element.
🟢 Difference:
Oxidation number can be positive or negative.
Valency is always positive and whole number.
Oxidation number varies with compound; valency is fixed for most elements.
🔶 Q24. Balance the redox reaction:
Fe²⁺ + Cr₂O₇²⁻ + H⁺ → Fe³⁺ + Cr³⁺ + H₂O (acidic medium)
📌 Answer:
🔹 Step 1: Oxidation half (Fe²⁺ → Fe³⁺)
Fe²⁺ → Fe³⁺ + e⁻
🔹 Step 2: Reduction half (Cr₂O₇²⁻ → Cr³⁺)
Cr₂O₇²⁻ + 14H⁺ + 6e⁻ → 2Cr³⁺ + 7H₂O
🔹 Step 3: Multiply oxidation by 6:
6Fe²⁺ → 6Fe³⁺ + 6e⁻
🔹 Step 4: Add both:
Cr₂O₇²⁻ + 14H⁺ + 6Fe²⁺ → 2Cr³⁺ + 7H₂O + 6Fe³⁺
✅ Final balanced redox equation.
🔶 Q25. Explain the concept of redox reactions in terms of electron transfer. Give an example.
📌 Answer:
🔹 Redox reactions involve transfer of electrons:
• Oxidation: Loss of electrons
• Reduction: Gain of electrons
🟢 Example:
Zn + Cu²⁺ → Zn²⁺ + Cu
• Zn is oxidised (loses 2e⁻)
• Cu²⁺ is reduced (gains 2e⁻)
✅ Hence, electron transfer governs redox processes.
🔶 Q26. Determine the oxidation number of N in NO₃⁻ and NH₄⁺.
📌 Answer:
🔹 In NO₃⁻:
Let x be oxidation number of N
x + 3(–2) = –1
⇒ x – 6 = –1
⇒ x = +5
🔹 In NH₄⁺:
x + 4(+1) = +1
⇒ x + 4 = +1
⇒ x = –3
✅ Oxidation numbers:
• N in NO₃⁻ = +5
• N in NH₄⁺ = –3
🔶 Q27. Define equivalent weight of an oxidising agent. Give its formula.
📌 Answer:
🔹 Equivalent weight of an oxidising agent =
(Molecular weight) / (Number of electrons gained per molecule)
🟢 Example: KMnO₄ in acid medium
Mn⁷⁺ → Mn²⁺ (gain of 5e⁻)
Equivalent weight = Molecular weight / 5
✅ Used in redox titrations.
🔶 Q28. Identify the oxidised and reduced species in the following reaction:
Cl₂ + 2NaOH → NaCl + NaClO + H₂O
📌 Answer:
Cl₂ → NaCl → Cl⁻ (oxidation number 0 to –1) → reduction
Cl₂ → NaClO → Cl⁺ (oxidation number 0 to +1) → oxidation
✅ So, Cl₂ is both oxidised and reduced → disproportionation reaction
🔷 Q29. Case-Based Question
Read the passage and answer the questions:
Hydrogen peroxide (H₂O₂) is a powerful oxidising as well as reducing agent. It decomposes to form water and oxygen. In acidic medium, it can oxidise iodide to iodine, whereas in basic medium, it undergoes disproportionation.
(i) Write the redox reaction of H₂O₂ with KI in acidic medium.
(ii) Identify the oxidised and reduced species.
(iii) What is the oxidation number of O in H₂O₂?
(iv) Write the reaction of H₂O₂ undergoing disproportionation in basic medium.
📌 Answer:
(i) 2KI + H₂O₂ + H₂SO₄ → I₂ + K₂SO₄ + 2H₂O
(ii)
🔹 I⁻ → I₂ (oxidation)
🔹 H₂O₂ → H₂O (reduction)
(iii)
🔹 In H₂O₂, oxidation number of O = –1
(iv)
🔹 2H₂O₂ → 2H₂O + O₂
(This is a disproportionation reaction: O is both oxidised and reduced)
🔷 Q30. Case-Based Question
The oxidation number concept helps in balancing redox reactions and identifying oxidising/reducing agents.
Consider the reaction:
Fe₂O₃ + CO → Fe + CO₂
(i) Assign oxidation numbers to all atoms.
(ii) Identify the oxidising and reducing agents.
(iii) Balance the redox reaction using oxidation number method.
(iv) Classify the type of redox process.
📌 Answer:
(i)
🔹 Fe in Fe₂O₃ = +3
🔹 O = –2
🔹 C in CO = +2
🔹 C in CO₂ = +4
🔹 Fe in Fe = 0
(ii)
🔹 Fe₂O₃ is oxidising agent
🔹 CO is reducing agent
(iii)
Fe₂O₃ + 3CO → 2Fe + 3CO₂
(iv)
This is a redox reaction involving oxidation of carbon and reduction of iron.
🔷 Q31. Case-Based Question
A redox titration involves KMnO₄ and FeSO₄ in acidic medium. KMnO₄ acts as oxidising agent and Fe²⁺ is reducing agent.
(i) Write the balanced redox reaction.
(ii) What is the role of H₂SO₄?
(iii) Calculate the n-factor of KMnO₄ in this reaction.
(iv) What colour change is observed at endpoint?
📌 Answer:
(i)
MnO₄⁻ + 8H⁺ + 5Fe²⁺ → Mn²⁺ + 5Fe³⁺ + 4H₂O
(ii)
🔹 H₂SO₄ provides acidic medium necessary for KMnO₄ to act as a strong oxidising agent.
(iii)
🔹 Mn⁷⁺ → Mn²⁺ (change = 5)
✅ n-factor = 5
(iv)
🔹 Purple (KMnO₄) to Colourless (Mn²⁺)
At endpoint, a permanent pink colour appears due to excess KMnO₄.
🔶 Q32. Long Answer (5 Marks)
Balance the following redox reaction by ion-electron method in acidic medium:
Cr₂O₇²⁻ + Fe²⁺ + H⁺ → Cr³⁺ + Fe³⁺ + H₂O
📌 Answer:
🔹 Step 1: Write half-reactions
Oxidation: Fe²⁺ → Fe³⁺ + e⁻
Reduction: Cr₂O₇²⁻ + 14H⁺ + 6e⁻ → 2Cr³⁺ + 7H₂O
🔹 Step 2: Multiply oxidation half by 6
6Fe²⁺ → 6Fe³⁺ + 6e⁻
🔹 Step 3: Add both
Cr₂O₇²⁻ + 14H⁺ + 6Fe²⁺ → 2Cr³⁺ + 7H₂O + 6Fe³⁺
✅ Final Balanced Redox Equation
🔶 Q33. Explain the types of redox reactions with one example each.
📌 Answer:
🔹 1. Combination Reaction:
H₂ + Cl₂ → 2HCl
🔹 2. Decomposition Reaction:
2H₂O₂ → 2H₂O + O₂
🔹 3. Displacement Reaction:
Zn + CuSO₄ → ZnSO₄ + Cu
🔹 4. Disproportionation Reaction:
Cl₂ + 2NaOH → NaCl + NaClO + H₂O
✅ These are all types of redox reactions involving oxidation and reduction.
🔶 Q34. Balance the reaction using ion-electron method in basic medium:
MnO₄⁻ + I⁻ → MnO₂ + IO₃⁻
📌 Answer:
🔹 Step 1: Oxidation half
I⁻ → IO₃⁻ (I: –1 to +5) → Loss of 6e⁻
🔹 Step 2: Reduction half
MnO₄⁻ + 2H₂O + 3e⁻ → MnO₂ + 4OH⁻
🔹 Step 3: LCM of electrons = 6
Multiply oxidation half by 1
Multiply reduction half by 2
🔹 Step 4: Add both
2MnO₄⁻ + I⁻ + 4H₂O → 2MnO₂ + IO₃⁻ + 4OH⁻
✅ Final Balanced Equation in basic medium
🔶 Q35. Differentiate between the following:
(i) Oxidation and reduction
(ii) Redox and non-redox reactions
(iii) Oxidising and reducing agents
📌 Answer:
(i) Oxidation vs Reduction
🔹 Oxidation = Loss of electrons
🔹 Reduction = Gain of electrons
(ii) Redox vs Non-Redox
🔹 Redox: Electron transfer occurs (e.g. Zn + Cu²⁺ → Zn²⁺ + Cu)
🔹 Non-redox: No electron transfer (e.g. NaCl + AgNO₃ → AgCl + NaNO₃)
(iii) Oxidising vs Reducing Agents
🔹 Oxidising Agent: Gains electrons (gets reduced)
🔹 Reducing Agent: Loses electrons (gets oxidised)
✅ These distinctions help in identifying redox processes clearly.
————————————————————————————————————————————————————————————————————————————
