Class 11 : Chemistry (In English) – Chapter 6: Equilibrium
EXPLANATION & SUMMARY
🔵 Introduction
Chemical reactions don’t always go to completion 🔁. Some stop midway, giving a mixture of reactants and products. This balance is called equilibrium ⚖. Equilibrium is central to chemistry — it explains how reactions occur in reversible ways, why some reactions yield little product, and how conditions like pressure and temperature affect the outcome.
Equilibrium is studied in two parts:
Physical Equilibrium → changes of state (solid–liquid, liquid–gas, etc.).
Chemical Equilibrium → reversible reactions in chemistry.
🟢 Equilibrium in Physical Processes
Examples:
Ice ⇌ Water (solid–liquid equilibrium).
H₂O(l) ⇌ H₂O(g) (liquid–vapor equilibrium).
Solute ⇌ Solution (saturated solution equilibrium).
Characteristics:
✔ Dynamic (forward and backward processes occur simultaneously).
✔ Rate of forward = rate of backward reaction.
✔ Observable properties constant (pressure, concentration).
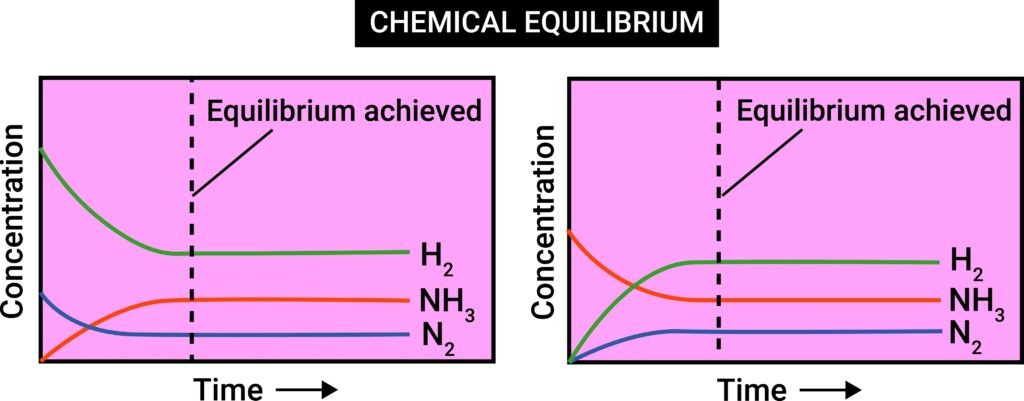
🔵 Equilibrium in Chemical Processes
Many chemical reactions are reversible. Example:
N₂ + 3H₂ ⇌ 2NH₃
When forward reaction equals backward reaction → chemical equilibrium.
📌 Characteristics:
Dynamic state.
Reached only in closed system.
Concentrations constant at equilibrium.
Equilibrium state depends on temperature, pressure, concentration.
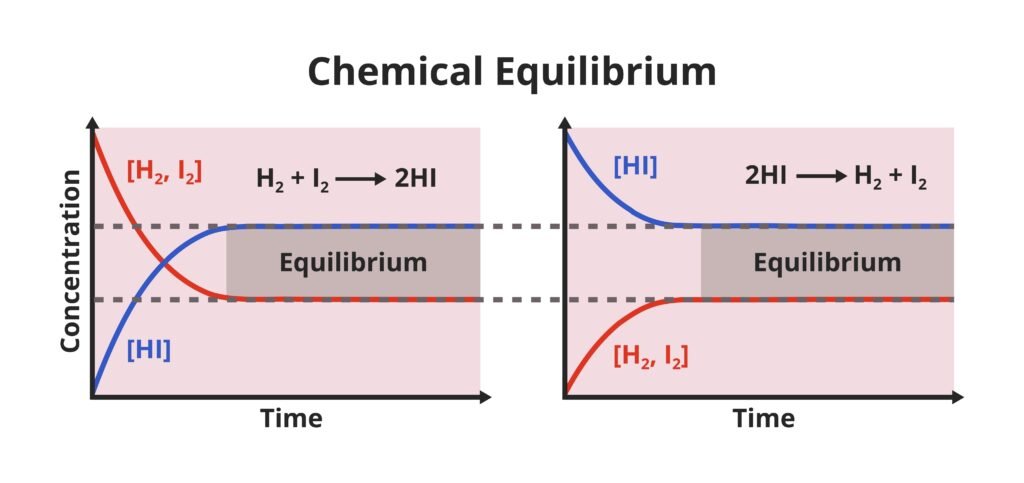
🟡 Law of Mass Action (Guldberg & Waage, 1864)
For a general reaction:
aA + bB ⇌ cC + dD
Rate ∝ [reactants]^coefficients.
At equilibrium,
Kc = [C]^c [D]^d / [A]^a [B]^b
This is the equilibrium constant (Kc).
🔴 Equilibrium Constant
Two forms:
Kc (concentration-based).
Kp (pressure-based, for gases).
Relation between Kp and Kc:
Kp = Kc (RT)^Δn
where Δn = (moles of gaseous products − moles of gaseous reactants).
🟢 Significance of K
If K is large (>>1): equilibrium lies towards products.
If K is small (<<1): equilibrium lies towards reactants.
If K ≈ 1: appreciable concentrations of both reactants and products.
🔵 Reaction Quotient (Q)
Same as K expression but for initial concentrations.
If Q < K → forward reaction dominates.
If Q > K → backward reaction dominates.
If Q = K → equilibrium attained.
🟡 Le Chatelier’s Principle (1884)
📌 If a system at equilibrium is disturbed, it shifts to counteract the disturbance.
Change in Concentration
Increase reactants → shift forward.
Increase products → shift backward.
Change in Pressure (for gases)
Increase pressure → shift towards fewer moles of gas.
Decrease pressure → shift towards more moles.
Change in Temperature
Exothermic reaction (ΔH < 0): increase T → shift backward.
Endothermic reaction (ΔH > 0): increase T → shift forward.
✔ Applications: Haber process, Contact process, industrial synthesis.
🔴 Ionic Equilibrium in Aqueous Solutions
When ionic compounds dissolve, they dissociate into ions. The equilibria involving ions are crucial.
1️⃣ Electrolytes
Strong electrolytes: completely ionize (NaCl, HCl).
Weak electrolytes: partially ionize (CH₃COOH, NH₃).
2️⃣ Ionization Equilibrium of Weak Electrolytes
Example: CH₃COOH ⇌ CH₃COO⁻ + H⁺
Ionization constant (Ka) = [H⁺][CH₃COO⁻]/[CH₃COOH].
Similarly, base ionization constant (Kb).
3️⃣ Ostwald’s Dilution Law
Degree of dissociation (α) increases with dilution.
Ka = Cα² / (1 − α).
🟢 pH Concept (Sørensen, 1909)
pH = −log[H⁺]
Acidic solution: pH < 7.
Neutral: pH = 7.
Basic: pH > 7.
pOH = −log[OH⁻].
Relation: pH + pOH = 14 (at 298 K).
🔵 Ionization of Water
H₂O ⇌ H⁺ + OH⁻
Kw = [H⁺][OH⁻] = 1 × 10⁻¹⁴ (25°C).
📌 At neutrality: [H⁺] = [OH⁻] = 10⁻⁷ M.
🟡 Common Ion Effect
📌 Suppression of ionization of weak electrolyte by adding strong electrolyte containing common ion.
Example:
CH₃COOH ⇌ CH₃COO⁻ + H⁺
Addition of CH₃COONa (common ion CH₃COO⁻) suppresses ionization.
Used in buffer solutions and salt solubility control.
🔴 Buffer Solutions
Solutions which resist change in pH when small amounts of acid/base are added.
Types:
Acidic buffer: weak acid + salt (CH₃COOH + CH₃COONa).
Basic buffer: weak base + salt (NH₄OH + NH₄Cl).
📌 Henderson–Hasselbalch equation:
pH = pKa + log([salt]/[acid]).
🟢 Salt Hydrolysis
When salt reacts with water to form acidic/basic solutions.
Examples:
NaCl (strong acid + strong base) → neutral.
CH₃COONa (weak acid + strong base) → basic.
NH₄Cl (strong acid + weak base) → acidic.
🔵 Solubility Product (Ksp)
For sparingly soluble salts, solubility is small but equilibrium is established.
Example: AgCl ⇌ Ag⁺ + Cl⁻
Ksp = [Ag⁺][Cl⁻].
📌 If ionic product (Qsp) > Ksp → precipitation occurs.
🟡 Applications of Equilibrium Concepts
✔ Industrial synthesis: Haber (NH₃), Contact (H₂SO₄).
✔ Medicine: pH of blood (7.35–7.45) maintained by buffers.
✔ Environmental: Ocean pH, solubility of gases in water.
✔ Analytical: Precipitation titrations, solubility calculations.
✨ Conclusion
Equilibrium is a balance ⚖ between opposing forces. It explains why reactions don’t go to completion, how conditions influence yields, and why pH, buffers, solubility matter in chemistry and biology. It’s a foundation for understanding advanced topics like kinetics, electrochemistry, and biochemistry.
📌 Equilibrium – Key Takeaways
🔵 Physical Equilibria
Solid–liquid, liquid–vapor, solute–solution.
Dynamic, forward = backward rate.
🟢 Chemical Equilibria
Reversible reactions in closed systems.
Law of Mass Action → Kc expression.
Equilibrium constant: Kc, Kp; relation Kp = Kc(RT)^Δn.
🔴 Factors Affecting Equilibrium
Concentration, pressure, temperature.
Le Chatelier’s Principle: system shifts to oppose change.
🟡 Ionic Equilibrium
Strong vs weak electrolytes.
Ionization constants (Ka, Kb).
pH = −log[H⁺], pOH = −log[OH⁻], Kw = 10⁻¹⁴.
🌟 Special Effects
Common ion effect → ionization suppression.
Buffers resist pH changes (Henderson–Hasselbalch).
Salt hydrolysis: acidic, basic, neutral salts.
Solubility product (Ksp) → precipitation criteria.
📚 Significance
Industrial yields (NH₃, H₂SO₄).
Biological systems (blood pH).
Analytical chemistry (titrations, solubility).
📝 Quick Recap:
✔ Equilibrium is a dynamic balance in reversible reactions.
✔ Physical equilibria include melting, vaporization, dissolution.
✔ Chemical equilibria described by Kc, Kp; position depends on conditions.
✔ Le Chatelier’s Principle predicts shifts due to stress.
✔ Ionic equilibrium introduces pH, buffers, salt hydrolysis, Ksp.
✔ Common ion effect and buffer action crucial in labs and biology.
✔ Equilibrium is central to industry, medicine, and environment.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
🔵 Question 6.1:
A liquid is in equilibrium with its vapour in a sealed container at a fixed temperature. The volume of the container is suddenly increased.
a) What is the initial effect of the change on vapour pressure?
b) How do rates of evaporation and condensation change initially?
c) What happens when equilibrium is restored finally and what will be the final vapour pressure?
🟢 Answer:
a) ➤ On increasing volume, vapour pressure decreases initially because the same number of vapour molecules now occupy a larger volume, so pressure falls.
b) ➤ Rate of evaporation remains constant (depends only on temperature) but rate of condensation decreases (fewer vapour molecules collide with liquid surface).
c) ➤ Since evaporation > condensation, more liquid evaporates. Vapour pressure increases again until new equilibrium is reached.
✔ Final vapour pressure = same as initial because it depends only on temperature, not volume.
🔵 Question 6.2:
What is Kc for the following equilibrium when the equilibrium concentration of each substance is: [SO₃] = 1.90 M, [SO₂] = 0.60 M, [O₂] = 0.82 M?
Reaction:
2SO₂(g) + O₂(g) ⇌ 2SO₃(g)
🟢 Answer:
➡ Expression: Kc = [SO₃]² / ([SO₂]² × [O₂])
➡ Substitute: Kc = (1.90)² / ((0.60)² × 0.82)
➡ Simplify: Kc = 3.61 / (0.36 × 0.82) = 3.61 / 0.2952
✔ Kc = 12.23
🔵 Question 6.3:
At a certain temperature and total pressure of 10⁵ Pa, iodine vapour contains 40% by volume of I atoms.
Reaction:
I₂(g) ⇌ 2I(g)
Calculate Kp for the equilibrium.
🟢 Answer:
Let total pressure = 10⁵ Pa
Fraction of I = 0.4 → I₂ = 0.6
Assume total moles = 1 →
I₂ = 0.6 mol, I = 0.4 mol
Total = 1 mol
Partial pressures:
p(I₂) = 0.6 × 10⁵ = 6 × 10⁴ Pa
p(I) = 0.4 × 10⁵ = 4 × 10⁴ Pa
Kp = (p(I))² / p(I₂)
= (4 × 10⁴)² / (6 × 10⁴)
= (16 × 10⁸) / (6 × 10⁴)
✔ Kp = 2.67 × 10⁴ Pa
🔵 Question 6.4:
Write the expression for the equilibrium constant, Kc, for each reaction:
(i) 2NOCl(g) ⇌ 2NO(g) + Cl₂(g)
(ii) 2Cu(NO₃)₂(s) ⇌ 2CuO(s) + 4NO₂(g) + O₂(g)
(iii) CH₃COOC₂H₅(aq) + H₂O(l) ⇌ CH₃COOH(aq) + C₂H₅OH(aq)
(iv) Fe³⁺(aq) + 3OH⁻(aq) ⇌ Fe(OH)₃(s)
(v) I₂(s) + 5F₂(g) ⇌ 2IF₅(g)
🟢 Answer:
(i) Kc = [NO]²[Cl₂] / [NOCl]²
(ii) Kc = [NO₂]⁴[O₂]
(Solids excluded)
(iii) Kc = [CH₃COOH][C₂H₅OH] / [CH₃COOC₂H₅]
(Water excluded as pure liquid)
(iv) Kc = 1 / ([Fe³⁺][OH⁻]³)
(v) Kc = [IF₅]² / [F₂]⁵
(Solid I₂ excluded)
🔵 Question 6.5:
Find the value of Kc from Kp for:
(i) 2NOCl(g) ⇌ 2NO(g) + Cl₂(g), Kp = 1.8 × 10⁻² at 500 K
(ii) CaCO₃(s) ⇌ CaO(s) + CO₂(g), Kp = 167 at 1073 K
🟢 Answer:
Formula: Kp = Kc(RT)^(Δn)
(i) Δn = (2 + 1) – 2 = 1
Kc = Kp / (RT)^1
= 1.8 × 10⁻² / (0.0821 × 500)
= 1.8 × 10⁻² / 41.05 = 4.38 × 10⁻⁴
(ii) Δn = 1 – 0 = 1
Kc = Kp / (RT)
= 167 / (0.0821 × 1073)
= 167 / 88.57 = 1.88
🔵 Question 6.6:
For equilibrium:
NO(g) + O₃(g) ⇌ NO₂(g) + O₂(g), Kc = 6.3 × 10¹⁴ at 1000 K
Find Kc for reverse reaction.
🟢 Answer:
For reverse reaction, Kc’ = 1 / Kc
✔ Kc’ = 1.59 × 10⁻¹⁵
🔵 Question 6.7:
Explain why pure liquids and solids can be ignored while writing equilibrium constant expression.
🟢 Answer:
✔ Concentration of pure solids and liquids is constant (density fixed).
✔ They are included in Kc value, but treated as 1.
Therefore, equilibrium expression includes only gases and solutes.
🔵 Question 6.8:
Reaction: 2N₂(g) + O₂(g) ⇌ 2N₂O(g)
Mixture: 0.482 mol N₂, 0.933 mol O₂ in 10 L, Kc = 2.0 × 10⁻³⁷
Determine equilibrium composition.
🟢 Answer:
➡ Initial conc: [N₂]₀ = 0.0482 M, [O₂]₀ = 0.0933 M, [N₂O]₀ = 0
Let x = formed conc of N₂O
At equilibrium:
[N₂] = 0.0482 – x
[O₂] = 0.0933 – 0.5x
[N₂O] = x
Kc = [N₂O]² / ([N₂]²[O₂])
As Kc is very small, x negligible.
So [N₂O] ≈ 0, [N₂] ≈ 0.0482 M, [O₂] ≈ 0.0933 M
✔ Reaction hardly proceeds.
🔵 Question 6.9:
2NO(g) + Br₂(g) ⇌ 2NOBr(g)
When 0.087 mol NO and 0.0437 mol Br₂ are mixed in 1 L, 0.0518 mol NOBr formed.
Find Kc.
🟢 Answer:
Initial: [NO] = 0.087, [Br₂] = 0.0437, [NOBr] = 0
Change: –0.0518 NOBr/2 = –0.0259 NO, –0.01295 Br₂
Equilibrium:
[NO] = 0.087 – 0.0518 = 0.0352
[Br₂] = 0.0437 – 0.0259 = 0.0178
[NOBr] = 0.0518
Kc = [NOBr]² / ([NO]²[Br₂])
= (0.0518)² / ((0.0352)² × 0.0178)
= 0.002684 / (0.001239 × 0.0178)
= 0.002684 / 2.2×10⁻⁵ = 122
🔵 Question 6.10:
At 450 K, Kp = 2.0 × 10¹⁰ for:
2SO₂(g) + O₂(g) ⇌ 2SO₃(g). Find Kc.
🟢 Answer:
Δn = (2) – (3) = –1
Kp = Kc(RT)^(Δn)
Kc = Kp × (RT)¹
= 2 × 10¹⁰ × (0.0821 × 450)
= 2 × 10¹⁰ × 36.945 = 7.39 × 10¹¹
🔵 Question 6.11:
HI(g) ⇌ H₂(g) + I₂(g)
At 0.2 atm total pressure, p(HI)=0.04 atm
Find Kp.
🟢 Answer:
Let dissociation = x
p(HI) = 0.2 – 2x = 0.04 → x = 0.08
p(H₂) = x = 0.08, p(I₂) = x = 0.08
Kp = (0.08 × 0.08) / (0.04)² = 0.0064 / 0.0016 = 4
🔵 Question 6.12:
N₂ + 3H₂ ⇌ 2NH₃
1.57 mol N₂, 1.92 mol H₂, 8.13 mol NH₃ in 20 L at 500 K
Kc = 1.7 × 10²
Check equilibrium.
🟢 Answer:
Concentrations:
[N₂] = 1.57/20 = 0.0785
[H₂] = 1.92/20 = 0.096
[NH₃] = 8.13/20 = 0.4065
Q = [NH₃]² / ([N₂][H₂]³)
= (0.4065)² / (0.0785 × 0.096³)
= 0.165 / (0.0785 × 0.0008847)
= 0.165 / 6.94×10⁻⁵ = 2377
Q > Kc → reaction shifts left.
🔵 Question 6.13:
Given Kc = [NH₄⁺][OH⁻] / ([NO][H₃O⁺])
Write balanced equation.
🟢 Answer:
NH₄NO ⇌ NH₄⁺ + NO + H₃O⁺ + OH⁻
But to match form, reaction is:
NH₄NO + H₂O ⇌ NH₄⁺ + NO + H₃O⁺ + OH⁻
🔵 Question 6.14:
1 mol H₂O + 1 mol CO in 10 L at 725 K
40% H₂O reacts → 0.4 mol reacts
Reaction: H₂O + CO ⇌ H₂ + CO₂
Equilibrium moles:
H₂O = 0.6, CO = 0.6, H₂ = 0.4, CO₂ = 0.4
Concentrations:
All /10 → [H₂O] = 0.06, [CO] = 0.06, [H₂] = 0.04, [CO₂] = 0.04
Kc = [H₂][CO₂]/([H₂O][CO])
= (0.04×0.04)/(0.06×0.06)
= 0.0016/0.0036 = 0.444
🔵 Question 6.15:
At 700 K, for HI ⇌ ½H₂ + ½I₂, K = 54.8
If [HI]=0.5 M, find [H₂] and [I₂].
🟢 Answer:
HI ⇌ H₂ + I₂
Let dissociation = x
Kc = [H₂][I₂]/[HI]² = x²/(0.5–2x)²
Approximate if x small: x = √(Kc)×[HI] = √(54.8)×0.5 ≈ 7.4×0.5 = 3.7
Not possible; hence solve exact:
Let [HI]=0.5–2x
Kc=54.8=(x²)/(0.5–2x)²
√54.8 = x/(0.5–2x)
7.4(0.5–2x)=x → 3.7–14.8x=x → 15.8x=3.7 → x=0.234
So [H₂]=[I₂]=0.234 M, [HI]=0.5–0.468=0.032 M
✔ [H₂]=0.234 M, [I₂]=0.234 M, [HI]=0.032 M
🔵 Question 6.16
What is the equilibrium concentration of each of the substances in the equilibrium when the initial concentration of ICl was 0.78 M?
Reaction: 2 ICl (g) ⇌ I₂ (g) + Cl₂ (g); Kc = 0.14
🟢 Answer
Let dissociation = 2x M.
Initial: [ICl] = 0.78, [I₂] = 0, [Cl₂] = 0
Change: –2x, +x, +x
Equilibrium: [ICl] = 0.78 – 2x, [I₂] = x, [Cl₂] = x
Kc = ([I₂][Cl₂]) / [ICl]² = x² / (0.78 – 2x)² = 0.14
√0.14 = x / (0.78 – 2x) → 0.374(0.78 – 2x) = x
0.291 – 0.748x = x → 1.748x = 0.291 → x = 0.166 M
✅ [I₂] = [Cl₂] = 0.166 M; [ICl] = 0.448 M
🔵 Question 6.17
For C₂H₆ (g) ⇌ C₂H₄ (g) + H₂ (g), Kp = 0.04 atm at 899 K.
If the initial pressure of C₂H₆ is 4 atm, find its equilibrium pressure.
🟢 Answer
Let dissociation = x atm.
P(C₂H₆) = 4 – x; P(C₂H₄) = x; P(H₂) = x
Kp = x² / (4 – x) = 0.04 ⇒ x ≈ 0.4 atm
✅ P(C₂H₆) = 3.6 atm, P(C₂H₄) = 0.4 atm, P(H₂) = 0.4 atm
🔵 Question 6.18
CH₃COOH (l) + C₂H₅OH (l) ⇌ CH₃COOC₂H₅ (l) + H₂O (l)
(i) Write Qc.
(ii) If 0.171 mol ester is formed from 1 mol each of reactants in 1 L at 293 K, find Kc.
(iii) If later ester = 0.214 mol, has equilibrium been reached?
🟢 Answer
(i) Qc = [ester][H₂O]/([acid][alcohol])
(ii) Equilibrium: acid = alcohol = 0.829 M; products = 0.171 M
Kc = (0.171²)/(0.829²) = 0.042
(iii) For 0.214 mol, Qc = 0.074 > Kc ⇒ shifts backward ⇒ not at equilibrium.
🔵 Question 6.19
PCl₅ (g) ⇌ PCl₃ (g) + Cl₂ (g), Kc = 8.3 × 10⁻³ at 473 K.
If 1 mol PCl₅ in 1 L partially dissociates, find equilibrium concentrations.
🟢 Answer
Let dissociation = x mol.
[PCl₅] = 1 – x, [PCl₃] = x, [Cl₂] = x
Kc = x² / (1 – x) = 8.3×10⁻³ ⇒ x ≈ 0.091 M
✅ [PCl₅] = 0.909 M, [PCl₃] = 0.091 M, [Cl₂] = 0.091 M
🔵 Question 6.20
FeO (s) + CO (g) ⇌ Fe (s) + CO₂ (g), Kp = 0.265 at 1050 K.
Initial: PCO = 1.4 atm, PCO₂ = 0.80 atm. Find equilibrium pressures.
🟢 Answer
Let change = x.
1.4 – x, 0.80 + x satisfy 0.265 = (0.80 + x)/(1.4 – x)
x = –0.339 ⇒ backward shift
✅ PCO = 1.739 atm, PCO₂ = 0.461 atm
🔵 Question 6.21
N₂ + 3H₂ ⇌ 2NH₃, Kc = 0.061.
Given [N₂] = 3 M, [H₂] = 2 M, [NH₃] = 0.5 M. Is system at equilibrium?
🟢 Answer
Qc = 0.25 / (3 × 8) = 0.0104 < Kc ⇒ reaction moves forward.
🔵 Question 6.22
2BrCl ⇌ Br₂ + Cl₂, Kc = 3.2×10⁻³; initial [BrCl] = 3.3×10⁻³ M.
Find equilibrium concentrations.
🟢 Answer
x = 1.87×10⁻⁴ M
✅ [Br₂] = [Cl₂] = 1.87×10⁻⁴ M; [BrCl] = 2.93×10⁻³ M
🔵 Question 6.23
C (s) + CO₂ (g) ⇌ 2CO (g); mixture at 1 atm has 90.55 % CO by mass. Find Kp.
🟢 Answer
Mole fractions: X_CO = 0.938, X_CO₂ = 0.062
Kp = (0.938)² / 0.062 = 14.2 atm
🔵 Question 6.24
NO + ½O₂ ⇌ NO₂; ΔG°(NO₂)=52, ΔG°(NO)=87 kJ mol⁻¹.
Find ΔG° and K.
🟢 Answer
ΔG° = 52 – 87 = –35 kJ
K = e^(35000 / (8.314×298)) ≈ 1.3×10⁶
✔ ΔG° = –35 kJ, K = 1.3×10⁶
🔵 Question 6.25
How does decreasing pressure affect equilibrium when gas moles differ?
🟢 Answer
Shift to side with more gas moles.
Example: N₂O₄ ⇌ 2NO₂ → shifts right.
🔵 Question 6.26
Which equilibria shift on increasing pressure?
🟢 Answer
Shift to side with fewer moles.
→ Reactions (iv) forward; (ii, iii, vi) backward; (i, v) no effect.
🔵 Question 6.27
H₂ + Br₂ ⇌ 2HBr, Kp = 1.6×10⁵, initial P(HBr)=10 bar.
Find equilibrium composition.
🟢 Answer
Let dissociation = 2x.
(10 – 2x)²/x² = 1.6×10⁵ ⇒ x ≈ 0.025 bar
✅ P(HBr) = 9.95 bar; P(H₂) = P(Br₂) = 0.025 bar
🔵 Question 6.28
CH₄ + H₂O ⇌ CO + 3H₂ (endothermic)
🟢 Answer
Kp = (P_CO × P_H₂³)/(P_CH₄ × P_H₂O)
↑P → left; ↑T → right; catalyst → no shift.
🔵 Question 6.29
2H₂ + CO ⇌ CH₃OH
🟢 Answer
Add H₂ → right; add CH₃OH → left; remove CO → left; remove CH₃OH → right.
🔵 Question 6.30
PCl₅ ⇌ PCl₃ + Cl₂, Kp = 8.3×10⁻³, ΔH° = +124 kJ.
🟢 Answer
(a) Kc = [PCl₃][Cl₂]/[PCl₅]
(b) Reverse K = 120.5
(c) Add PCl₅ → forward; ↑P → left; ↑T → right (K increases)
🔵 Question 6.31
Explain the following terms:
(a) Conjugate acid-base pair
(b) Amphoteric species
(c) Lewis acids and bases
🟢 Answer:
(a) Conjugate acid-base pair:
When an acid donates a proton (H⁺), the species formed is its conjugate base; similarly, when a base accepts a proton, the species formed is its conjugate acid.
➡ Example: HCl + H₂O ⇌ H₃O⁺ + Cl⁻
Here, HCl (acid) → Cl⁻ (conjugate base), H₂O (base) → H₃O⁺ (conjugate acid).
Pair: (HCl / Cl⁻), (H₂O / H₃O⁺)
(b) Amphoteric species:
A species that can act as both acid and base.
➡ Example: HCO₃⁻ acts as acid (donates H⁺) and base (accepts H⁺).
(c) Lewis acids and bases:
Lewis acid: Electron pair acceptor (e.g., BF₃, AlCl₃).
Lewis base: Electron pair donor (e.g., NH₃, OH⁻).
➡ Example: BF₃ + NH₃ → F₃B←NH₃
🔵 Question 6.32
The ionization constant of HF is 6.8×10⁻⁴. Calculate the degree of dissociation of HF in 0.1 M solution and pH.
🟢 Answer:
Let HF ⇌ H⁺ + F⁻
Initial = 0.1, 0, 0
Change = –α×0.1, +α×0.1, +α×0.1
Ka = (Cα²)/(1–α) ≈ Cα² (since α small)
6.8×10⁻⁴ = 0.1α²
α² = 6.8×10⁻³ → α = 0.082
✔ Degree of dissociation = 8.2%
[H⁺] = Cα = 0.1 × 0.082 = 8.2×10⁻³ M
pH = –log(8.2×10⁻³) = 2.09
🔵 Question 6.33
Calculate pH of 1×10⁻⁸ M HCl solution.
🟢 Answer:
H⁺ comes from both acid and water:
Let total [H⁺] = x
x = [H⁺ from HCl] + [H⁺ from H₂O] = 1×10⁻⁸ + (Kw/x)
Kw = 1×10⁻¹⁴
⇒ x² – 1×10⁻⁸x – 1×10⁻¹⁴ = 0
Solve: x = (1×10⁻⁸ + √(1×10⁻¹⁶ + 4×10⁻¹⁴)) / 2
x = (1×10⁻⁸ + 2×10⁻⁷)/2 ≈ 1.05×10⁻⁷ M
pH = –log(1.05×10⁻⁷) = 6.98
✔ Slightly < 7 (acidic)
🔵 Question 6.34
The ionization constant of phenol is 1×10⁻¹⁰. What is the concentration of phenolate ion in 0.05 M phenol solution? Also find pH.
🟢 Answer:
C₆H₅OH ⇌ C₆H₅O⁻ + H⁺
Ka = 1×10⁻¹⁰ = (x²)/(0.05) ⇒ x² = 5×10⁻¹² ⇒ x = 2.24×10⁻⁶
✔ [C₆H₅O⁻] = 2.24×10⁻⁶ M
pH = –log(2.24×10⁻⁶) = 5.65
🔵 Question 6.35
What is the pH of 0.002 M solution of NaOH?
🟢 Answer:
NaOH is strong base → [OH⁻] = 0.002 M
pOH = –log(0.002) = 2.70
pH = 14 – 2.70 = 11.30
🔵 Question 6.36
Calculate pH of 0.001 M Ba(OH)₂ solution.
🟢 Answer:
Each Ba(OH)₂ gives 2 OH⁻
[OH⁻] = 2 × 0.001 = 0.002 M
pOH = –log(0.002) = 2.70
pH = 14 – 2.70 = 11.30
🔵 Question 6.37
Calculate pH of 0.1 M solution of an acid with Ka = 1.0×10⁻⁴.
🟢 Answer:
HA ⇌ H⁺ + A⁻
Ka = Cα² = 1×10⁻⁴ = 0.1α² → α² = 1×10⁻³ → α = 0.0316
[H⁺] = Cα = 0.1 × 0.0316 = 3.16×10⁻³ M
pH = –log(3.16×10⁻³) = 2.50
🔵 Question 6.38
Calculate pH of 0.01 M H₂SO₄ solution (considering full dissociation of first H⁺ and partial for second).
Ka₂ = 1.2×10⁻²
🟢 Answer:
After first dissociation: [H⁺] = 0.01, [HSO₄⁻] = 0.01
HSO₄⁻ ⇌ H⁺ + SO₄²⁻
Let additional H⁺ = x
Ka₂ = (0.01 + x)(x)/(0.01 – x) ≈ (0.01)(x)/0.01 = x = 1.2×10⁻²
Total [H⁺] = 0.01 + 0.012 = 0.022 M
pH = –log(0.022) = 1.66
🔵 Question 6.39
What is the pH of 10⁻⁸ M NaOH solution?
🟢 Answer:
Let [OH⁻] = x = 10⁻⁸
Total [OH⁻] = 10⁻⁸ + (Kw/x) = 10⁻⁸ + 10⁻⁶ = 1.01×10⁻⁶
pOH = –log(1.01×10⁻⁶) = 5.996
pH = 14 – 5.996 = 8.00
✔ Slightly basic.
🔵 Question 6.40
Calculate pH of 0.2 M solution of HCN (Ka = 4.8×10⁻¹⁰).
🟢 Answer:
Ka = Cα² ⇒ α² = 4.8×10⁻¹⁰ / 0.2 = 2.4×10⁻⁹
α = 4.9×10⁻⁵
[H⁺] = 0.2 × 4.9×10⁻⁵ = 9.8×10⁻⁶
pH = –log(9.8×10⁻⁶) = 5.01
🔵 Question 6.41
The pH of a weak acid solution is 3.0. Calculate its ionization constant if concentration is 0.1 M.
🟢 Answer:
pH = 3 → [H⁺] = 1×10⁻³
Ka = [H⁺]² / (C – [H⁺]) = (1×10⁻³)² / (0.1 – 0.001) = 1×10⁻⁶ / 0.099 ≈ 1.01×10⁻⁵
🔵 Question 6.42
Find pH of 0.05 M CH₃COOH (Ka = 1.8×10⁻⁵).
🟢 Answer:
Ka = Cα² ⇒ α² = 1.8×10⁻⁵ / 0.05 = 3.6×10⁻⁴ ⇒ α = 0.019
[H⁺] = 0.05 × 0.019 = 9.5×10⁻⁴
pH = –log(9.5×10⁻⁴) = 3.02
🔵 Question 6.43
Find pH of a solution obtained by mixing 50 mL of 0.1 M HCl with 50 mL of 0.1 M NaOH.
🟢 Answer:
Moles HCl = 0.005, Moles NaOH = 0.005 → neutralize completely
Solution = 100 mL, [H⁺] = 0
Neutral → pH = 7.00
🔵 Question 6.44
Find pH of a solution obtained by mixing 25 mL of 0.1 M HCl with 50 mL of 0.1 M NaOH.
🟢 Answer:
Moles HCl = 0.0025, Moles NaOH = 0.005 → Excess OH⁻ = 0.0025 mol
Total volume = 75 mL = 0.075 L
[OH⁻] = 0.0025 / 0.075 = 0.0333 M
pOH = –log(0.0333) = 1.48
pH = 14 – 1.48 = 12.52
🔵 Question 6.45
Calculate pH of a solution obtained by mixing 40 mL of 0.1 M NaOH with 10 mL of 0.1 M HCl.
🟢 Answer:
Moles NaOH = 0.004, HCl = 0.001 → Excess OH⁻ = 0.003 mol
Total volume = 50 mL = 0.05 L
[OH⁻] = 0.003 / 0.05 = 0.06 M
pOH = –log(0.06) = 1.22
pH = 14 – 1.22 = 12.78
🔵 Question 6.46
Calculate the pH of a buffer solution containing 0.1 M NH₃ and 0.1 M NH₄Cl. (Kb for NH₃ = 1.77 × 10⁻⁵)
🟢 Answer:
We use Henderson–Hasselbalch equation (for basic buffer):
pOH = pKb + log([salt]/[base])
➡ pKb = –log(1.77 × 10⁻⁵) = 4.75
➡ Ratio [salt]/[base] = 0.1 / 0.1 = 1
So pOH = 4.75 + log(1) = 4.75
pH = 14 – 4.75 = 9.25
✔ pH = 9.25
🔵 Question 6.47
Calculate the pH of a buffer solution containing 0.05 M acetic acid and 0.1 M sodium acetate. (Ka = 1.8 × 10⁻⁵)
🟢 Answer:
Use Henderson–Hasselbalch (acidic buffer):
pH = pKa + log([salt]/[acid])
pKa = –log(1.8 × 10⁻⁵) = 4.74
Ratio = 0.1 / 0.05 = 2
pH = 4.74 + log(2) = 4.74 + 0.30 = 5.04
✔ pH = 5.04
🔵 Question 6.48
How much NaOH must be added to 0.5 L of 0.1 M acetic acid to make pH = 4.74? (Ka = 1.8 × 10⁻⁵)
🟢 Answer:
For pH = pKa → ratio [salt]/[acid] = 1
Initial moles acid = 0.5 × 0.1 = 0.05 mol
We need [salt] = [acid] ⇒ convert half acid into salt
Moles NaOH required = 0.025 mol
Mass = 0.025 × 40 = 1.0 g
✔ Add 1.0 g NaOH
🔵 Question 6.49
What is the pH of a solution obtained by mixing 100 mL of 0.1 M CH₃COOH and 100 mL of 0.1 M NaOH? (Ka = 1.8 × 10⁻⁵)
🟢 Answer:
Moles CH₃COOH = 0.01, NaOH = 0.01 → neutralize completely
→ forms 0.01 mol CH₃COONa in 0.2 L → [salt] = 0.05 M
Buffer with no free acid/base, pH ≈ pKa + log([salt]/[acid])
[acid] ≈ 0 → treat as weak base hydrolysis:
Kb = Kw / Ka = 10⁻¹⁴ / 1.8×10⁻⁵ = 5.56×10⁻¹⁰
[OH⁻] = √(Kb × C) = √(5.56×10⁻¹⁰ × 0.05) = 5.3×10⁻⁶
pOH = 5.28 → pH = 8.72
✔ pH = 8.72
🔵 Question 6.50
Find pH of a mixture of 50 mL 0.1 M NH₄OH and 25 mL 0.1 M HCl. (Kb = 1.77 × 10⁻⁵)
🟢 Answer:
Moles NH₄OH = 0.005, HCl = 0.0025
Excess base = 0.0025 mol; salt formed = 0.0025 mol
Total volume = 75 mL = 0.075 L
[base] = 0.0025 / 0.075 = 0.0333 M
[salt] = 0.0333 M
pOH = pKb + log([salt]/[base]) = 4.75 + log(1) = 4.75
pH = 14 – 4.75 = 9.25
✔ pH = 9.25
🔵 Question 6.51
What is the pH of a solution obtained by mixing 50 mL of 0.1 M NaOH and 25 mL of 0.1 M HCl?
🟢 Answer:
Moles NaOH = 0.005, HCl = 0.0025
Excess OH⁻ = 0.0025 mol
Total volume = 75 mL = 0.075 L
[OH⁻] = 0.0025 / 0.075 = 0.0333 M
pOH = –log(0.0333) = 1.48
pH = 14 – 1.48 = 12.52
✔ pH = 12.52
🔵 Question 6.52
Calculate the pH of a solution prepared by mixing 25 mL of 0.1 M NaOH with 50 mL of 0.1 M HCl.
🟢 Answer:
Moles NaOH = 0.0025, HCl = 0.005 → excess acid = 0.0025 mol
Total volume = 75 mL = 0.075 L
[H⁺] = 0.0025 / 0.075 = 0.0333 M
pH = –log(0.0333) = 1.48
✔ pH = 1.48
🔵 Question 6.53
What is the pH of a buffer prepared by mixing 250 mL of 0.1 M NH₄OH and 250 mL of 0.1 M NH₄Cl? (Kb = 1.77×10⁻⁵)
🟢 Answer:
Equal moles of base and salt → [salt]/[base] = 1
pOH = pKb = 4.75
pH = 14 – 4.75 = 9.25
✔ pH = 9.25
🔵 Question 6.54
Calculate pH of a solution obtained by mixing 40 mL 0.1 M HCl and 10 mL 0.1 M NaOH.
🟢 Answer:
Moles HCl = 0.004, NaOH = 0.001 → excess acid = 0.003
Total volume = 50 mL = 0.05 L
[H⁺] = 0.003 / 0.05 = 0.06 M
pH = –log(0.06) = 1.22
✔ pH = 1.22
🔵 Question 6.55
Calculate pH of a solution obtained by mixing 10 mL 0.1 M HCl and 40 mL 0.1 M NaOH.
🟢 Answer:
Moles HCl = 0.001, NaOH = 0.004 → excess OH⁻ = 0.003
Total volume = 50 mL = 0.05 L
[OH⁻] = 0.003 / 0.05 = 0.06 M
pOH = –log(0.06) = 1.22
pH = 14 – 1.22 = 12.78
✔ pH = 12.78
🔵 Question 6.56
Calculate the pH of a 0.05 M NH₄Cl solution. (Kb for NH₃ = 1.77×10⁻⁵)
🟢 Answer:
NH₄Cl → weak acid (hydrolysis of NH₄⁺):
Ka = Kw / Kb = 10⁻¹⁴ / 1.77×10⁻⁵ = 5.65×10⁻¹⁰
[H⁺] = √(Ka × C) = √(5.65×10⁻¹⁰ × 0.05) = 5.31×10⁻⁶
pH = –log(5.31×10⁻⁶) = 5.28
✔ pH = 5.28
🔵 Question 6.57
Calculate the pH of 0.1 M Na₂CO₃ solution. (Kb₁ for CO₃²⁻ = 2.1×10⁻⁴)
🟢 Answer:
CO₃²⁻ + H₂O ⇌ HCO₃⁻ + OH⁻
[OH⁻] = √(Kb₁ × C) = √(2.1×10⁻⁴ × 0.1) = 4.58×10⁻³
pOH = –log(4.58×10⁻³) = 2.34
pH = 14 – 2.34 = 11.66
✔ pH = 11.66
🔵 Question 6.58
Calculate the pH of 0.1 M NaHCO₃ solution. (Ka₁ = 4.3×10⁻⁷, Ka₂ = 4.8×10⁻¹¹)
🟢 Answer:
HCO₃⁻ is amphiprotic → pH = ½(pKa₁ + pKa₂)
pKa₁ = 6.37, pKa₂ = 10.32
pH = ½(6.37 + 10.32) = 8.35
✔ pH = 8.35
🔵 Question 6.59
What is the pH of 0.1 M CH₃COONa solution? (Ka = 1.8×10⁻⁵)
🟢 Answer:
CH₃COO⁻ hydrolysis: Kb = Kw / Ka = 10⁻¹⁴ / 1.8×10⁻⁵ = 5.56×10⁻¹⁰
[OH⁻] = √(Kb × C) = √(5.56×10⁻¹⁰ × 0.1) = 7.45×10⁻⁶
pOH = 5.13 → pH = 14 – 5.13 = 8.87
✔ pH = 8.87
🔵 Question 6.60
Calculate the pH of 0.05 M NH₄NO₃ solution. (Kb for NH₃ = 1.77×10⁻⁵)
🟢 Answer:
NH₄⁺ hydrolysis: Ka = Kw / Kb = 10⁻¹⁴ / 1.77×10⁻⁵ = 5.65×10⁻¹⁰
[H⁺] = √(Ka × C) = √(5.65×10⁻¹⁰ × 0.05) = 5.31×10⁻⁶
pH = –log(5.31×10⁻⁶) = 5.28
✔ pH = 5.28
🔵 Question 6.61
Calculate the pH of 0.1 M NH₄Cl and 0.05 M NaOH mixture. (Kb for NH₃ = 1.77 × 10⁻⁵)
🟢 Answer:
This forms a basic buffer (NH₄⁺/NH₃).
Use pOH = pKb + log([salt]/[base])
pKb = –log(1.77 × 10⁻⁵) = 4.75
Ratio = 0.1 / 0.05 = 2
pOH = 4.75 + log(2) = 4.75 + 0.30 = 5.05
pH = 14 – 5.05 = 8.95
✔ pH = 8.95
🔵 Question 6.62
Find pH of a buffer containing 0.1 M CH₃COOH and 0.1 M CH₃COONa when 0.01 mol HCl is added to 1 L. (Ka = 1.8 × 10⁻⁵)
🟢 Answer:
Initial moles acid = 0.1; salt = 0.1
HCl converts salt → acid:
New moles acid = 0.11; salt = 0.09
pH = pKa + log([salt]/[acid])
pKa = 4.74
pH = 4.74 + log(0.09/0.11) = 4.74 + log(0.818) = 4.74 – 0.087 = 4.65
✔ pH = 4.65
🔵 Question 6.63
Find pH of same buffer when 0.01 mol NaOH is added instead.
🟢 Answer:
NaOH reacts with acid → salt:
New acid = 0.09, salt = 0.11
pH = 4.74 + log(0.11/0.09) = 4.74 + 0.087 = 4.83
✔ pH = 4.83
🔵 Question 6.64
Calculate solubility of AgCl (Ksp = 1.6 × 10⁻¹⁰).
🟢 Answer:
AgCl ⇌ Ag⁺ + Cl⁻
Let solubility = s
Ksp = s²
s = √(1.6 × 10⁻¹⁰) = 1.26 × 10⁻⁵ M
✔ Solubility = 1.26 × 10⁻⁵ mol L⁻¹
🔵 Question 6.65
Calculate solubility of PbCl₂ (Ksp = 1.7 × 10⁻⁵).
🟢 Answer:
PbCl₂ ⇌ Pb²⁺ + 2 Cl⁻
Let solubility = s
Ksp = s × (2s)² = 4s³
s³ = 1.7 × 10⁻⁵ / 4 = 4.25 × 10⁻⁶
s = (4.25 × 10⁻⁶)^(1/3) = 1.62 × 10⁻² M
✔ Solubility = 1.62 × 10⁻² mol L⁻¹
🔵 Question 6.66
Calculate solubility of CaF₂ in water (Ksp = 3.2 × 10⁻¹¹).
🟢 Answer:
CaF₂ ⇌ Ca²⁺ + 2 F⁻
Ksp = s × (2s)² = 4s³
s³ = 3.2 × 10⁻¹¹ / 4 = 8 × 10⁻¹²
s = (8 × 10⁻¹²)^(1/3) = 2.0 × 10⁻⁴ M
✔ Solubility = 2.0 × 10⁻⁴ mol L⁻¹
🔵 Question 6.67
Calculate solubility of Ag₂CrO₄ (Ksp = 1.1 × 10⁻¹²).
🟢 Answer:
Ag₂CrO₄ ⇌ 2 Ag⁺ + CrO₄²⁻
Ksp = (2s)² × s = 4s³
s³ = 1.1 × 10⁻¹² / 4 = 2.75 × 10⁻¹³
s = (2.75 × 10⁻¹³)^(1/3) = 6.5 × 10⁻⁵ M
✔ Solubility = 6.5 × 10⁻⁵ mol L⁻¹
🔵 Question 6.68
Calculate solubility of BaSO₄ (Ksp = 1.1 × 10⁻¹⁰) in 0.01 M Na₂SO₄.
🟢 Answer:
BaSO₄ ⇌ Ba²⁺ + SO₄²⁻
Ksp = s × (0.01 + s) ≈ s × 0.01
s = 1.1 × 10⁻¹⁰ / 0.01 = 1.1 × 10⁻⁸ M
✔ Solubility = 1.1 × 10⁻⁸ mol L⁻¹
🔵 Question 6.69
Calculate solubility of AgCl in 0.1 M NaCl. (Ksp = 1.6 × 10⁻¹⁰)
🟢 Answer:
Ksp = [Ag⁺][Cl⁻] ≈ s × 0.1
s = 1.6 × 10⁻¹⁰ / 0.1 = 1.6 × 10⁻⁹ M
✔ Solubility = 1.6 × 10⁻⁹ mol L⁻¹
🔵 Question 6.70
If Ksp = 1.0 × 10⁻²⁵ for Ag₂S, find solubility in pure water.
🟢 Answer:
Ag₂S ⇌ 2 Ag⁺ + S²⁻
Ksp = 4s³
s³ = 1 × 10⁻²⁵ / 4 = 2.5 × 10⁻²⁶
s = (2.5 × 10⁻²⁶)^(1/3) = 2.9 × 10⁻⁹ M
✔ Solubility = 2.9 × 10⁻⁹ mol L⁻¹
🔵 Question 6.71
What is the pH at which Fe(OH)₃ (Ksp = 4 × 10⁻³⁸) starts precipitating from 0.001 M Fe³⁺?
🟢 Answer:
Ksp = [Fe³⁺][OH⁻]³
[OH⁻]³ = 4 × 10⁻³⁸ / 10⁻³ = 4 × 10⁻³⁵
[OH⁻] = (4 × 10⁻³⁵)^(1/3) = 3.4 × 10⁻¹²
pOH = 11.47 → pH = 2.53
✔ pH = 2.53
🔵 Question 6.72
At what pH will Mg(OH)₂ (Ksp = 8.9 × 10⁻¹²) start to precipitate from 0.01 M Mg²⁺?
🟢 Answer:
Ksp = [Mg²⁺][OH⁻]²
[OH⁻]² = 8.9 × 10⁻¹² / 0.01 = 8.9 × 10⁻¹⁰
[OH⁻] = 2.98 × 10⁻⁵ → pOH = 4.53 → pH = 9.47
✔ pH = 9.47
🔵 Question 6.73
Will AgCl (Ksp = 1.6 × 10⁻¹⁰) precipitate if 10⁻⁴ M AgNO₃ and 10⁻⁴ M NaCl are mixed?
🟢 Answer:
Ionic product, Q = [Ag⁺][Cl⁻] = (10⁻⁴)(10⁻⁴) = 10⁻⁸
Q > Ksp (10⁻⁸ > 1.6 × 10⁻¹⁰) → precipitation occurs
✔ AgCl will precipitate.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
✳ Section A (Q1–Q16) – MCQs (1 mark each, 16 × 1 = 16 marks)
Options:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Question 1. Equilibrium in a chemical reaction is established when:
Forward and backward reactions stop
Forward and backward reactions have equal rates
Concentration of reactants becomes zero
Products stop forming
Answer: 2
Question 2. Which of the following is a dynamic equilibrium?
Evaporation of water in a closed vessel
Evaporation in an open vessel
Decomposition of H₂O₂ in open air
Rusting of iron
Answer: 1
Question 3. The equilibrium constant is expressed as:
K_p/K_c
Products/Reactants
Reactants/Products
None of these
Answer: 2
Question 4. For the reaction N₂(g) + 3H₂(g) ⇌ 2NH₃(g), the equilibrium constant expression K_c is:
[N₂][H₂]³/[NH₃]²
[NH₃]²/[N₂][H₂]³
[NH₃]/[N₂][H₂]
[NH₃]³/[N₂][H₂]²
Answer: 2
Question 5. The value of K_c for a reaction is 10⁵. This means:
Products are favored
Reactants are favored
Both equally favored
Equilibrium does not exist
Answer: 1
Question 6. If Q_c > K_c, the reaction will:
Proceed forward
Proceed backward
Remain at equilibrium
Stop
Answer: 2
Question 7. pH of neutral water at 25 °C is:
6
7
8
14
Answer: 2
Question 8. Kw at 25 °C is equal to:
10⁻¹⁴
10⁻⁷
10⁻¹²
10⁻¹⁰
Answer: 1
Question 9. Which acid is diprotic?
HCl
H₂SO₄
HNO₃
CH₃COOH
Answer: 2
Question 10. Common-ion effect is used in:
Neutralization
Buffer solutions
Hydrolysis
Titrations only
Answer: 2
Question 11. Which salt solution is basic?
NaCl
NH₄Cl
CH₃COONa
KHSO₄
Answer: 3
Question 12. The unit of K_c for reaction A + B ⇌ AB is:
mol L⁻¹
L mol⁻¹
Dimensionless
mol² L⁻²
Answer: 2
Question 13. Le Chatelier’s principle predicts:
Direction of reaction shift
Equilibrium constant value
Activation energy
Bond energy
Answer: 1
Question 14. (Assertion–Reason)
Assertion (A): For exothermic reactions, increasing temperature decreases K.
Reason (R): Equilibrium shifts in direction that absorbs heat.
Answer: 1
Question 15. (Assertion–Reason)
Assertion (A): Pure solids and liquids are not included in K expressions.
Reason (R): Their concentrations remain constant.
Answer: 1
Question 16. The pH of 0.01 M HCl solution is:
1
2
3
4
Answer: 2
⚡ Section B (Q17–Q21) – Very Short Answer (2 marks each, 5 × 2 = 10 marks)
Q17. Define equilibrium constant.
🟦 The ratio of product concentration to reactant concentration at equilibrium, each raised to power of stoichiometric coefficients.
🟩 Represents position of equilibrium.
Q18. Write expression for K_p in terms of K_c.
🟦 K_p = K_c(RT)^Δn
🟩 Δn = moles of gaseous products – moles of gaseous reactants.
Q19. What is the significance of Q_c?
🟦 Q_c = [products]/[reactants] at any instant.
🟩 Comparison of Q_c and K_c tells direction of reaction shift.
Q20. Calculate pOH of a solution with [OH⁻] = 1 × 10⁻⁵ M.
➤ Formula: pOH = –log[OH⁻]
➤ Substitution: pOH = –log(1 × 10⁻⁵)
✅ Final Answer: pOH = 5
Q21. State common-ion effect with an example.
🟦 Suppression of ionization of weak electrolyte by adding strong electrolyte containing a common ion.
🟩 Example: CH₃COOH ionization suppressed by CH₃COONa.
🧪 Section C (Q22–Q28) – Short Answer (3 marks each, 7 × 3 = 21 marks)
Q22. Differentiate between homogeneous and heterogeneous equilibrium with examples.
🟦 Homogeneous: all species in same phase (N₂ + 3H₂ ⇌ 2NH₃).
🟨 Heterogeneous: species in different phases (CaCO₃(s) ⇌ CaO(s) + CO₂(g)).
🟩 Criterion: phases present.
Q23. Derive relationship between K_p and K_c.
➤ General reaction: aA + bB ⇌ cC + dD.
➤ K_p = (pC^c pD^d)/(pA^a pB^b).
➤ Using p = cRT: K_p = K_c(RT)^Δn.
✅ Relation: K_p = K_c(RT)^Δn.
Q24. Explain effect of temperature on equilibrium constant for exothermic and endothermic reactions.
🟦 Exothermic: Increase in T → K decreases, reaction shifts backward.
🟨 Endothermic: Increase in T → K increases, reaction shifts forward.
🟩 Explained by Le Chatelier’s principle.
Q25. Write differences between strong and weak electrolytes with examples.
🟦 Strong: completely ionize in solution (HCl, NaOH).
🟨 Weak: partially ionize (CH₃COOH, NH₃).
🟩 Conductivity higher for strong electrolytes.
Q26. Calculate pH of 0.001 M HNO₃ solution.
➤ Formula: pH = –log[H⁺].
➤ Substitution: pH = –log(0.001).
✅ Final Answer: pH = 3.
Q27. Explain buffer action of CH₃COOH/CH₃COONa.
🟦 Addition of H⁺ → reacts with CH₃COO⁻ to form CH₃COOH.
🟨 Addition of OH⁻ → reacts with CH₃COOH to form CH₃COO⁻.
🟩 Maintains nearly constant pH.
Q28. Write expression for hydrolysis constant of salt of weak acid and strong base.
🟦 For CH₃COONa: CH₃COO⁻ + H₂O ⇌ CH₃COOH + OH⁻.
🟨 Hydrolysis constant Kh = Kw/Ka.
🟩 pH > 7.
🧭 Section D (Q29–Q30) – Case-Based Questions (4 marks each, 2 × 4 = 8 marks)
Q29. Read the passage and answer the questions:
In the synthesis of ammonia (N₂ + 3H₂ ⇌ 2NH₃), the reaction is exothermic. According to Le Chatelier’s principle, changes in pressure, temperature, and concentration affect equilibrium.
(a) What is the effect of increasing pressure on the equilibrium? (1 mark)
(b) What is the effect of increasing temperature on equilibrium yield? (1 mark)
(c) Explain why high pressure and moderate temperature are used industrially. (2 marks)
🧪 Answer:
(a) High pressure shifts equilibrium towards ammonia (fewer gas molecules).
(b) High temperature shifts equilibrium backward (exothermic reaction).
(c) Industrially, high pressure increases yield, and moderate temperature ensures a balance of good yield and fast rate.
Q30. Read the passage and answer the questions:
The dissociation of acetic acid is suppressed by adding sodium acetate. This is the common-ion effect and is applied in buffer preparation.
(a) Write dissociation equation of acetic acid. (1 mark)
(b) What is the effect of adding CH₃COONa? (1 mark)
(c) Explain how buffer solution resists change in pH. (2 marks)
🧪 Answer:
(a) CH₃COOH ⇌ CH₃COO⁻ + H⁺.
(b) Addition of CH₃COONa increases [CH₃COO⁻], suppressing dissociation.
(c) Added H⁺ combines with CH₃COO⁻, and added OH⁻ reacts with CH₃COOH → pH remains nearly constant.
⚡ Section E (Q31–Q33) – Long Answer (5 marks each, 3 × 5 = 15 marks)
Q31. (a) State Le Chatelier’s principle. Apply it to the reaction N₂O₄ ⇌ 2NO₂ with respect to pressure and temperature changes.
OR
(b) Derive the relationship between Kp and Kc for a general gaseous reaction.
🧪 Answer (a):
🟦 Principle: If a system at equilibrium is disturbed, it shifts in a direction to counteract the change.
🟨 Pressure: Increase in pressure shifts equilibrium towards fewer gas molecules (N₂O₄).
🟩 Temperature: Since dissociation N₂O₄ → 2NO₂ is endothermic, higher T favors NO₂ formation.
Answer (b):
🟦 General reaction: aA + bB ⇌ cC + dD.
🟨 Kp = (pC^c pD^d)/(pA^a pB^b).
🟩 Using p = CRT → Kp = Kc(RT)^Δn, where Δn = (c + d – a – b).
Q32. (a) Define ionic product of water. Derive relation between Kw and pH.
OR
(b) Calculate pH of 0.01 M Ba(OH)₂ solution at 25 °C.
🧪 Answer (a):
🟦 Ionic product: Kw = [H⁺][OH⁻] = 1 × 10⁻¹⁴ at 25 °C.
🟨 In neutral water: [H⁺] = [OH⁻] = 1 × 10⁻⁷ M.
🟩 pH = –log[H⁺].
🎯 Relation: pKw = pH + pOH = 14.
Answer (b):
➤ Ba(OH)₂ → Ba²⁺ + 2OH⁻.
➤ [OH⁻] = 2 × 0.01 = 0.02 M.
➤ pOH = –log(0.02) ≈ 1.70.
➤ pH = 14 – 1.70 = 12.30.
✅ Final Answer: pH = 12.3.
Q33. (a) Explain buffer solution and derive Henderson–Hasselbalch equation.
OR
(b) Write notes on solubility product (Ksp). Derive expression for molar solubility of AgCl.
🧪 Answer (a):
🟦 Buffer: Resists pH change on addition of small acid/base.
🟨 Weak acid buffer: pH = pKa + log([salt]/[acid]).
🟩 Derivation: From Ka = [H⁺][A⁻]/[HA], rearranging gives Henderson equation.
🎯 Example: CH₃COOH/CH₃COONa buffer.
Answer (b):
🟦 Solubility product: Ksp = product of ionic concentrations of a sparingly soluble salt at equilibrium.
🟨 For AgCl ⇌ Ag⁺ + Cl⁻, Ksp = [Ag⁺][Cl⁻].
🟩 Let solubility = S → [Ag⁺] = [Cl⁻] = S.
🧮 Ksp = S² → S = √Ksp.
————————————————————————————————————————————————————————————————————————————
