Class 11 : Chemistry (In English) – Chapter 5: Thermodynamics
EXPLANATION & SUMMARY
🔵 Introduction
Thermodynamics is the branch of science dealing with the study of energy changes 🔥 in physical and chemical processes. Every reaction around us involves energy — burning fuel 🚗, boiling water 💧, photosynthesis 🌱, respiration in our body 🫁. Energy can neither be created nor destroyed but it can change from one form to another. Thermodynamics helps us understand how energy is transferred, in what direction it flows, and whether a process is possible or not.
Chemistry uses thermodynamics mainly to study the heat changes (enthalpy) accompanying chemical reactions and the feasibility of processes.
🟢 Basic Terms in Thermodynamics
1️⃣ System and Surroundings
System: Part of universe under study (reaction mixture, vessel).
Surroundings: Everything outside the system.
Universe = System + Surroundings.
2️⃣ Types of Systems
Open system: exchange of matter + energy (boiling water in open pot).
Closed system: exchange of energy only, not matter (sealed vessel).
Isolated system: no exchange of matter or energy (thermos flask).
3️⃣ State of System
Defined by properties like pressure (P), volume (V), temperature (T), composition.
4️⃣ State Functions
Properties that depend only on state, not path taken. Examples: P, V, T, U (internal energy), H (enthalpy), G (Gibbs free energy).
5️⃣ Intensive vs Extensive Properties
Intensive: independent of mass (temperature, pressure).
Extensive: depend on mass (volume, enthalpy).
🔵 Internal Energy (U)
Total energy of system = kinetic + potential energy of particles.
Change in internal energy (ΔU) occurs due to heat (q) absorbed or work (w) done.
Equation:
ΔU = q + w
✔ Work done at constant pressure: w = −PΔV
(negative when system expands, positive when compressed).
🟢 First Law of Thermodynamics
✨ Law of Conservation of Energy: Energy can neither be created nor destroyed; it only changes form.
Equation:
ΔU = q + w
At constant volume (ΔV=0), ΔU = qᵥ.
At constant pressure, heat absorbed = enthalpy change (ΔH).
🔴 Enthalpy (H)
H = U + PV
Change in enthalpy (ΔH) = qₚ (heat at constant pressure).
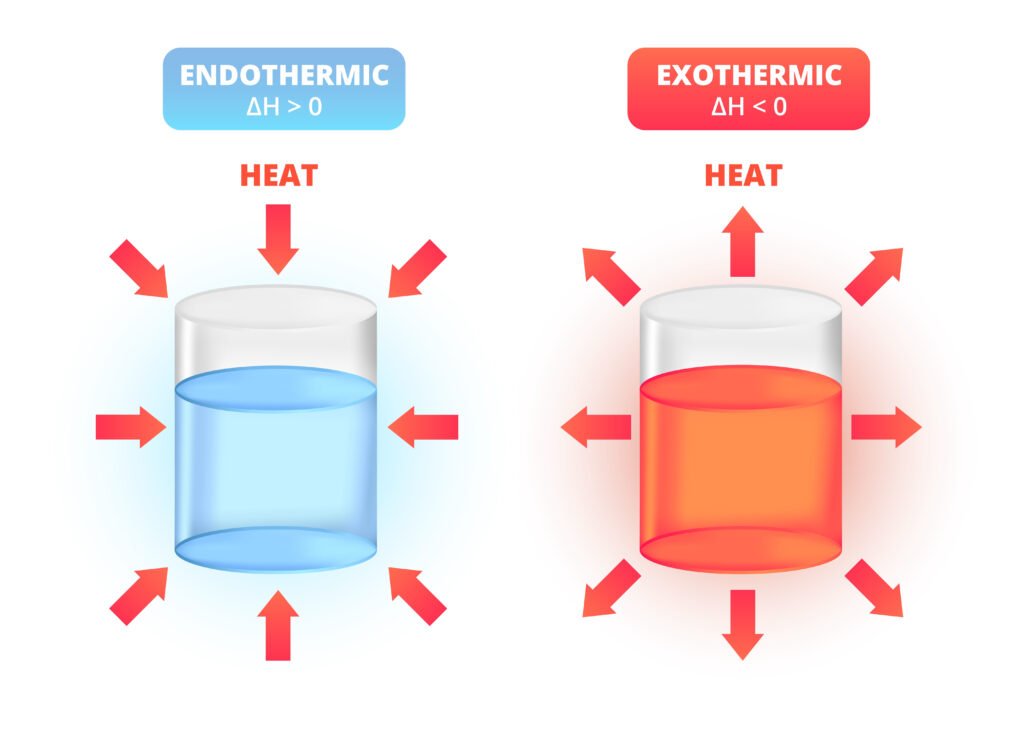
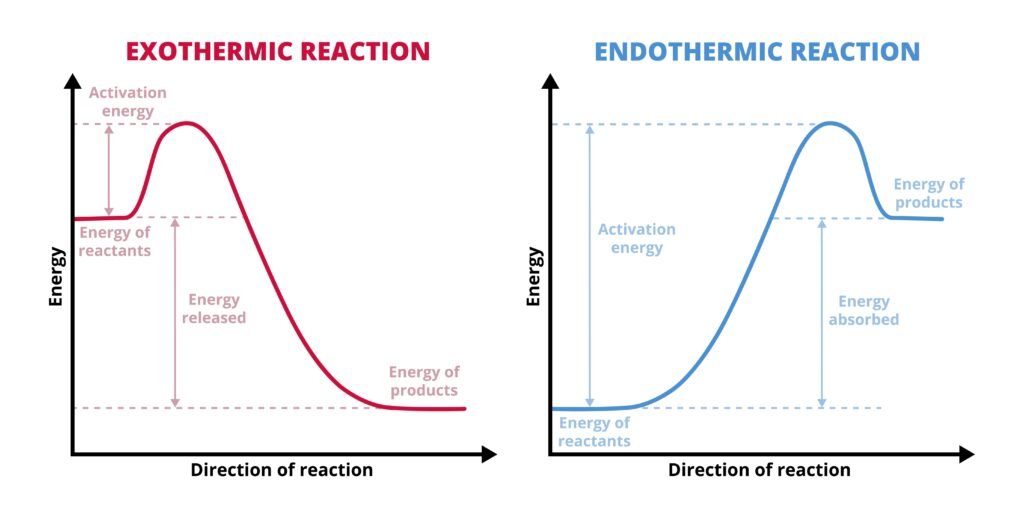
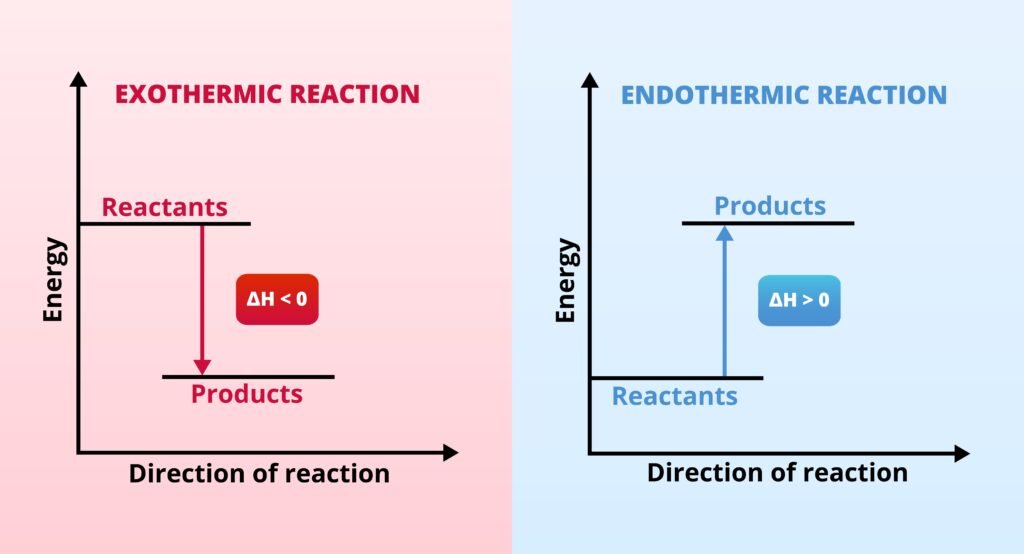
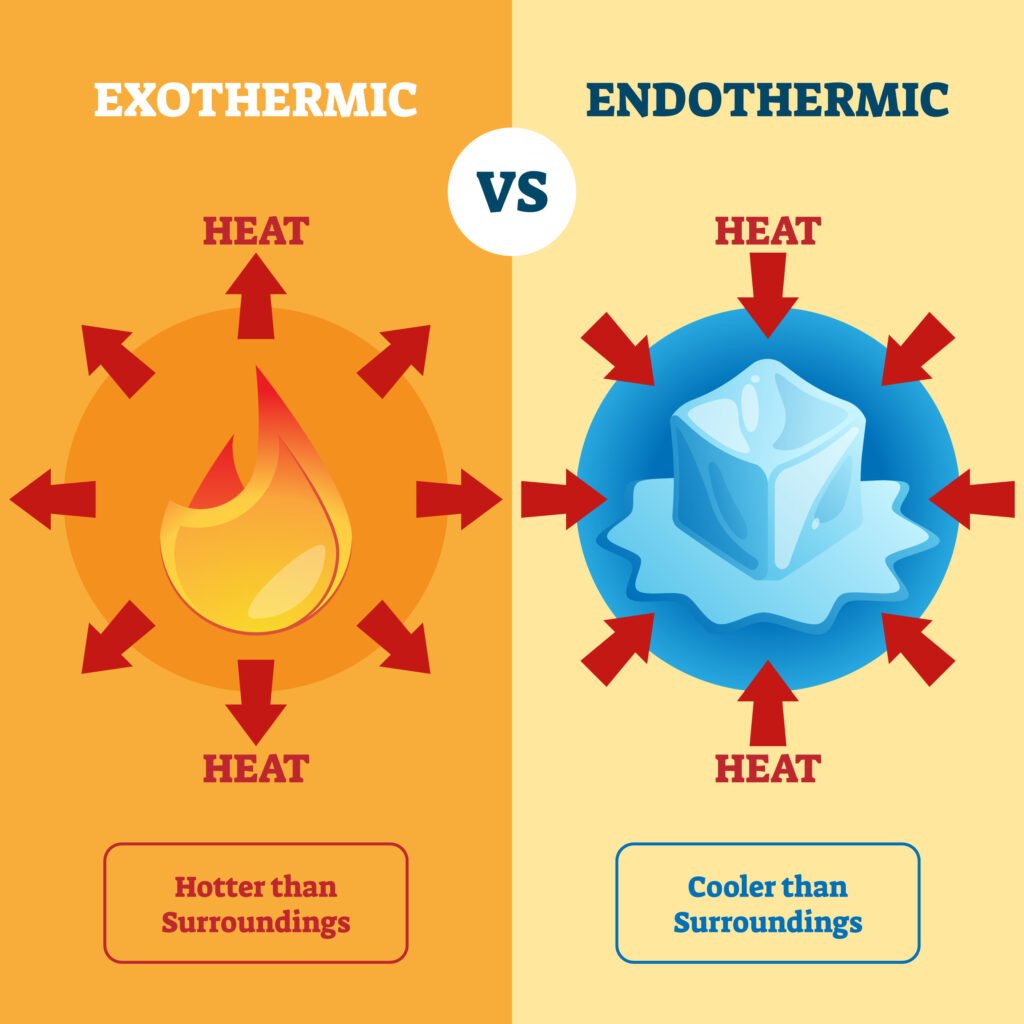
Endothermic process: ΔH > 0 (absorbs heat, e.g., melting ice ❄).
Exothermic process: ΔH < 0 (releases heat, e.g., combustion 🔥).
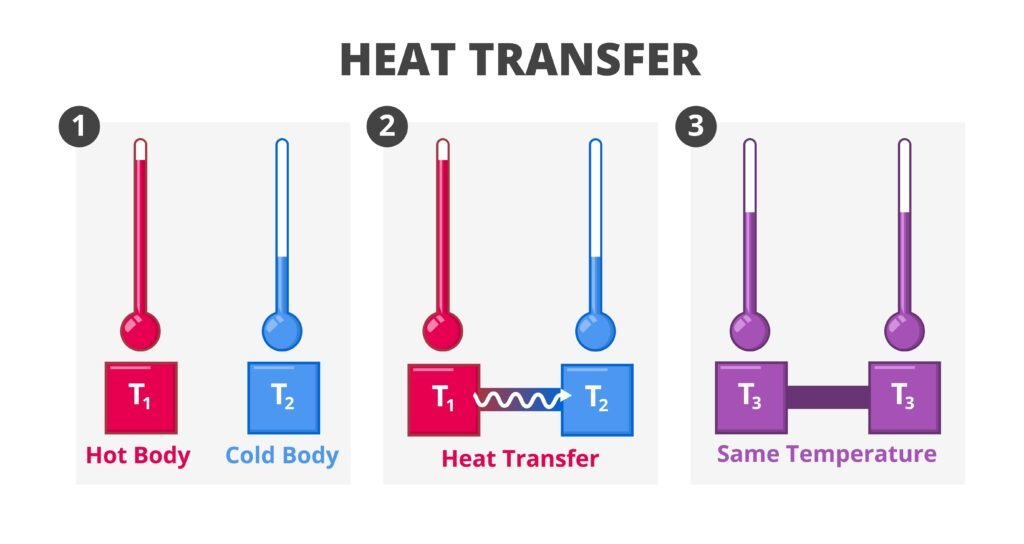
🟡 Second Law of Thermodynamics
Focuses on spontaneity. Some processes occur naturally (ice melts at 25°C, gas diffuses), others don’t (heat doesn’t flow cold → hot).
Important statements:
Clausius Statement: Heat cannot flow from cold body to hot body without external work.
Kelvin–Planck Statement: It is impossible to convert all heat into work.
Introduces entropy (S): measure of disorder/randomness.
ΔS > 0 → disorder increases (spontaneous tendency).
ΔS < 0 → disorder decreases.
🟢 Third Law of Thermodynamics
At absolute zero (0 K), entropy of a perfectly crystalline substance = 0.
This provides reference point for entropy values.
🔵 Applications of First Law
✔ Heat capacity (C): quantity of heat required to raise temperature by 1 K.
C = q / ΔT.
Molar heat capacity: per mole of substance.
✔ Relation between Cp and Cv:
Cp − Cv = R (ideal gas).
🟡 Standard Enthalpy Changes
Enthalpy of Formation
Heat change when 1 mole of compound forms from elements in standard state.
Example: ΔHf° of H₂O = −286 kJ/mol.
Enthalpy of Combustion
Heat released on complete combustion of 1 mole.
Example: CH₄ + 2O₂ → CO₂ + 2H₂O, ΔH = −890 kJ.
Enthalpy of Neutralization
Heat change when 1 mole of H⁺ neutralizes 1 mole OH⁻.
Strong acid + strong base → always −57.1 kJ/mol.
Enthalpy of Atomization
Heat change when 1 mole of gaseous atoms formed from element.
Enthalpy of Sublimation, Fusion, Vaporization
Heat change for phase transitions.
🔴 Hess’s Law of Constant Heat Summation
If a reaction occurs in several steps, total enthalpy change = sum of enthalpy changes of steps.
Example:
C (diamond) → C (graphite), ΔH = −1.9 kJ
C (graphite) + O₂ → CO₂, ΔH = −393.5 kJ
Total: C (diamond) + O₂ → CO₂, ΔH = −395.4 kJ
Applications:
Calculate ΔH when direct measurement not possible.
Useful for lattice enthalpies, bond enthalpies.
🟢 Bond Enthalpy
Energy required to break 1 mole of bonds in gaseous molecules.
For polyatomic molecules, average bond enthalpy is used.
Example: Bond enthalpy of H–H = 436 kJ/mol.
🔵 Spontaneity of Reactions
Why does ice melt at room temperature but not freeze? Thermodynamics answers this using Gibbs Free Energy (G).
Equation:
ΔG = ΔH − TΔS
ΔG < 0 → spontaneous.
ΔG > 0 → non-spontaneous.
ΔG = 0 → equilibrium.
🟡 Important Relationships
✔ ΔH = ΔU + ΔnRT (for gaseous reactions).
✔ ΔG° = −RT ln K (relation with equilibrium constant).
✔ ΔG = wmax (maximum non-expansion work possible).
🔴 Applications of Thermodynamics in Chemistry
Predicts feasibility of reactions.
Explains why combustion is exothermic and spontaneous.
Determines enthalpies for industrial processes (Haber, Contact process).
Basis for energy cycles in biology (ATP hydrolysis).
Explains solubility, phase changes, spontaneity of diffusion.
✨ Conclusion
Thermodynamics is a guiding principle of chemistry 🔥🌊. It explains why reactions occur, how much heat is released or absorbed, and whether processes are feasible. It connects chemistry to physics, biology, and engineering, making it a universal science of energy and matter.
📌 Thermodynamics – Key Takeaways
🔵 Basic Concepts
System, surroundings, universe.
Types: open, closed, isolated.
State functions (P, V, T, U, H, G, S).
Intensive vs extensive properties.
🟢 First Law
ΔU = q + w.
Energy conserved.
Enthalpy H = U + PV, ΔH = qₚ.
Exothermic (−ΔH), endothermic (+ΔH).
🔴 Second Law
Spontaneous processes increase entropy.
Heat doesn’t flow cold → hot naturally.
Efficiency always less than 100%.
🟡 Third Law
Entropy of perfect crystal at 0 K = 0.
🌟 Enthalpy Changes
Formation, combustion, neutralization, atomization, fusion, vaporization, sublimation.
📚 Important Laws & Concepts
Hess’s Law: ΔH is additive.
Bond enthalpy: average energy to break bonds.
Heat capacities: Cp, Cv; Cp − Cv = R.
💡 Spontaneity
Gibbs Free Energy: ΔG = ΔH − TΔS.
ΔG < 0 → spontaneous.
Relation to equilibrium constant: ΔG° = −RT ln K.
✨ Applications
Predict feasibility of reactions.
Industrial processes (Haber, Contact).
Biological energy (ATP hydrolysis).
Explains combustion, phase changes, solubility.
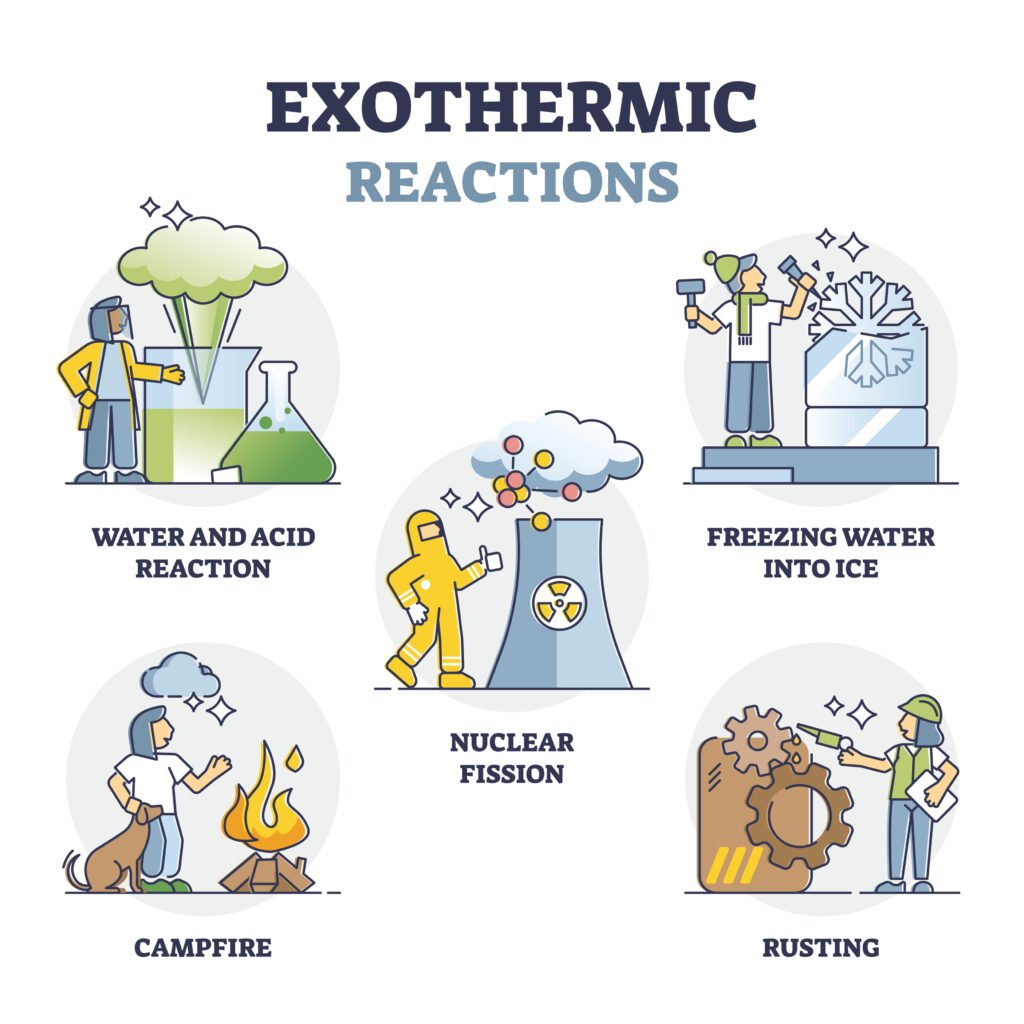
📝 Quick Recap:
✔ Thermodynamics studies energy changes in chemical/physical processes.
✔ First Law: energy conserved (ΔU = q + w).
✔ Enthalpy (ΔH): heat at constant pressure; exothermic vs endothermic.
✔ Second Law: entropy (S) increases in spontaneous processes.
✔ Third Law: entropy of perfect crystal at 0 K = 0.
✔ Key tools: Hess’s Law, bond enthalpies, Gibbs Free Energy.
✔ ΔG = ΔH − TΔS decides spontaneity.
✔ Basis for industrial, biological, and natural processes.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
🔵 Question 5.1
A thermodynamic state function is a quantity
(i) used to determine heat changes
(ii) whose value is independent of path
(iii) used to determine pressure–volume work
(iv) whose value depends on temperature only
🟢 Answer: ✅ (ii) — whose value is independent of path
✏ Explanation:
A state function depends only on the initial and final states, not on the path.
Examples: Internal energy (U), Enthalpy (H), Entropy (S), Pressure (P), Volume (V), Temperature (T).
In contrast, heat (q) and work (w) are path functions.
🔵 Question 5.2
For a process to occur under adiabatic conditions, the correct condition is
(i) ΔT = 0 (ii) Δp = 0 (iii) q = 0 (iv) w = 0
🟢 Answer: ✅ (iii) q = 0
✏ Explanation:
Adiabatic process means no heat exchange with surroundings.
So q = 0, but temperature and pressure can change as work is done.
🔵 Question 5.3
The enthalpies of all elements in their standard states are
(i) unity (ii) zero (iii) < 0 (iv) different
🟢 Answer: ✅ (ii) zero
✏ Explanation:
By convention, standard enthalpy of formation (ΔHf°) of an element in its standard state (298 K, 1 bar) is zero.
Example: ΔHf°(O₂, g) = 0; ΔHf°(N₂, g) = 0.
🔵 Question 5.4
If ΔU° = −X kJ mol⁻¹ for combustion of CH₄, the relation is
🟢 Answer: ✅ ΔH° < ΔU°
✏ Explanation:
Relation: ΔH = ΔU + Δn₍gas₎ × R × T
Reaction: CH₄(g) + 2 O₂(g) → CO₂(g) + 2 H₂O(l)
Δn₍gas₎ = 1 − 3 = −2
⇒ ΔH = ΔU − 2RT → ΔH° < ΔU°
🔵 Question 5.5
Find ΔHf° of CH₄(g) from data (kJ mol⁻¹):
ΔHcomb(CH₄) = −890.3
ΔHcomb(C) = −393.5
ΔHcomb(H₂) = −285.8
🟢 Answer:
✏ Step 1: Formation reaction:
C(s) + 2 H₂(g) → CH₄(g)
✏ Step 2: Use Hess’s Law:
ΔHf°(CH₄) = [ΔHcomb(C) + 2 × ΔHcomb(H₂)] − ΔHcomb(CH₄)
✏ Step 3: Substitute values:
= [ (−393.5) + 2(−285.8) ] − (−890.3)
= (−393.5 − 571.6) + 890.3
= −965.1 + 890.3 = −74.8 kJ mol⁻¹
✔ ΔHf°(CH₄) = −74.8 kJ mol⁻¹
🔵 Question 5.6
Reaction: A + B → C + D + q, with ΔS > 0
🟢 Answer: ✅ Possible at any temperature
✏ Explanation:
Exothermic ⇒ ΔH < 0; also ΔS > 0
Gibbs equation: ΔG = ΔH − TΔS
Both terms favour spontaneity ⇒ ΔG < 0 at all T ⇒ spontaneous always.
🔵 Question 5.7
System absorbs q = 701 J, does w = 394 J.
🟢 Answer:
ΔU = q − w = 701 − 394 = +307 J
✔ Internal energy increases by 307 J.
🔵 Question 5.8
NH₂CN(s) + 3/2 O₂(g) → N₂(g) + CO₂(g) + H₂O(l), ΔU = −742.7 kJ mol⁻¹
🟢 Answer:
✏ Step 1: Find Δn₍gas₎ = (1 + 1) − 1.5 = 0.5
✏ Step 2: Use relation: ΔH = ΔU + ΔnRT
= −742.7 + 0.5 × 8.314 × 298 / 1000
= −742.7 + 1.24 = −741.5 kJ mol⁻¹
✔ ΔH = −741.5 kJ mol⁻¹
🔵 Question 5.9
Heat to raise 60 g Al from 35 °C to 55 °C (Cₘ = 24 J mol⁻¹ K⁻¹)
🟢 Answer:
✏ Step 1: moles, n = 60 / 27 = 2.22 mol
✏ Step 2: ΔT = 55 − 35 = 20 K
✏ Step 3: q = n × Cₘ × ΔT = 2.22 × 24 × 20 = 1065.6 J ≈ 1.07 kJ
✔ q = +1.07 kJ absorbed by Al.
🔵 Question 5.10
Find ΔH for freezing 1 mol H₂O (10 °C → ice 0 °C)
Data: ΔHfusion = 6.03 kJ mol⁻¹; Cp(liq) = 75.3 J mol⁻¹ K⁻¹
🟢 Answer:
✏ Step 1: Cool water: q₁ = 75.3 × (−10)/1000 = −0.753 kJ
✏ Step 2: Freeze: q₂ = −6.03 kJ
✏ Step 3: Total ΔH = q₁ + q₂ = −6.78 kJ mol⁻¹
✔ ΔH = −6.78 kJ mol⁻¹ (exothermic)
🔵 Question 5.11
ΔHcomb(C) = −393.5 kJ mol⁻¹. Heat evolved for 35.2 g CO₂?
🟢 Answer:
✏ Step 1: n = 35.2 / 44 = 0.8 mol
✏ Step 2: q = n × ΔH = 0.8 × (−393.5) = −314.8 kJ
✔ Heat released = 314.8 kJ
🔵 Question 5.12
Calculate ΔH for the reaction:
N₂O(g) + 3 CO(g) → N₂(g) + 3 CO₂(g)
Given enthalpies of formation (kJ mol⁻¹):
CO = −110.5, CO₂ = −393.5, N₂O = +81, N₂ = 0
🟢 Answer:
✏ Formula: ΔH = ΣΔHf(products) − ΣΔHf(reactants)
= [0 + 3(−393.5)] − [81 + 3(−110.5)]
= (−1180.5) − (−250.5) = −930.0 kJ mol⁻¹
✔ Reaction is exothermic; ΔH = −930 kJ mol⁻¹
🔵 Question 5.13
Given: N₂ + 3 H₂ → 2 NH₃, ΔH = −92.4 kJ mol⁻¹
Find ΔHf° of NH₃
🟢 Answer:
The equation forms 2 mol NH₃,
So enthalpy per mole = (−92.4) / 2 = −46.2 kJ mol⁻¹
✔ ΔHf°(NH₃,g) = −46.2 kJ mol⁻¹
🔵 Question 5.14
Find ΔHf°(CH₃OH,l) from:
CH₃OH(l)+ 3/2 O₂ → CO₂ + 2 H₂O(l), ΔH = −726 kJ
ΔHf°(CO₂)=−393.5, ΔHf°(H₂O)=−286 kJ mol⁻¹
🟢 Answer:
ΔHf°(CH₃OH) = [ΔHf(CO₂)+2ΔHf(H₂O)] − ΔH
= [−393.5 + 2(−286)] − (−726)
= (−965.5) + 726 = −239.5 kJ mol⁻¹
✔ ΔHf°(CH₃OH,l) = −239.5 kJ mol⁻¹
🔵 Question 5.15
Find bond enthalpy of C–Cl in CCl₄(l) using:
ΔvapH = +30.5, ΔatH(C) = +715, ΔdissH(Cl₂) = +242 kJ mol⁻¹,
ΔHf(CCl₄,l) = −135.5 kJ mol⁻¹
🟢 Answer:
To break into atoms:
CCl₄(l) → C(g) + 4 Cl(g)
ΔH = ΔvapH + ΔatH(C) + 2ΔdissH(Cl₂) − ΔHf(CCl₄)
= 30.5 + 715 + 484 + 135.5 = 1365 kJ
Bond enthalpy = 1365 / 4 = 341 kJ mol⁻¹
✔ E(C–Cl) = 341 kJ mol⁻¹
🔵 Question 5.16
In an isolated system, ΔU = 0. What about ΔS?
🟢 Answer:
No energy or matter exchange; but entropy increases for spontaneous change.
✔ ΔS > 0 for spontaneous processes.
🔵 Question 5.17
At 298 K: 2A + B → C, ΔH = 400 kJ, ΔS = +0.2 kJ K⁻¹
Find T where reaction becomes spontaneous.
🟢 Answer:
ΔG = ΔH − TΔS
Spontaneous when ΔG < 0 ⇒ T > ΔH / ΔS
= 400 / 0.2 = 2000 K
✔ Reaction spontaneous at T > 2000 K.
🔵 Question 5.18
2 Cl(g) → Cl₂(g): predict signs of ΔH and ΔS.
🟢 Answer:
Bond formation releases energy ⇒ ΔH < 0
2 mol → 1 mol ⇒ disorder decreases ⇒ ΔS < 0
✔ ΔH < 0, ΔS < 0
🔵 Question 5.19
2 A(g)+B(g) → 2 D(g); ΔU = −10.5 kJ, ΔS = −44.1 J K⁻¹.
Find ΔG at 298 K and spontaneity.
🟢 Answer:
Convert ΔS = −0.0441 kJ K⁻¹
Δn = 2 − 3 = −1 ⇒ ΔH = ΔU + ΔnRT = −10.5 − 2.48 = −12.98 kJ
ΔG = ΔH − TΔS = −12.98 − 298(−0.0441) = +0.17 kJ
✔ ΔG = +0.17 kJ > 0, reaction non-spontaneous.
🔵 Question 5.20
K = 10, T = 300 K, R = 8.314 J mol⁻¹ K⁻¹.
Find ΔG°.
🟢 Answer:
ΔG° = −RT ln K
= −8.314 × 300 × 2.303 × 1 = −5.74 kJ mol⁻¹
✔ ΔG° = −5.74 kJ mol⁻¹
🔵 Question 5.21
Thermodynamic stability of NO(g):
½ N₂ + ½ O₂ → NO, ΔH = +90 kJ
NO + ½ O₂ → NO₂, ΔH = −74 kJ
🟢 Answer:
Formation of NO is endothermic ⇒ less stable
Converts exothermically to NO₂ ⇒ NO₂ more stable
✔ NO is thermodynamically unstable.
🔵 Question 5.22
Entropy change in surroundings for
H₂(g) + ½ O₂(g) → H₂O(l), ΔH = −286 kJ at 298 K.
🟢 Answer:
ΔS₍surr₎ = −ΔH / T = −(−286) / 298 = +0.96 kJ K⁻¹ = +960 J K⁻¹
✔ ΔS₍surr₎ = +960 J K⁻¹
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
✳ Section A (Q1–Q16) – MCQs (1 mark each, 16 × 1 = 16 marks)
Options:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Question 1. Thermodynamics is concerned with:
Rate of reactions
Heat and work
Mechanism of reactions
Atomic structure
Answer: 2
Question 2. Which of the following is a state function?
Work
Heat
Enthalpy
Path
Answer: 3
Question 3. The internal energy of a system is denoted by:
H
U
S
G
Answer: 2
Question 4. Which law states that energy can neither be created nor destroyed?
Zeroth law
First law
Second law
Third law
Answer: 2
Question 5. Work done at constant pressure is related to:
ΔU
ΔH
q_v
q_p
Answer: 4
Question 6. ΔH – ΔU = nRΔT is valid for:
Constant temperature
Gaseous reactions
Reversible reactions
Ionic reactions
Answer: 2
Question 7. The enthalpy of neutralization of strong acid and strong base is approximately:
–57 kJ mol⁻¹
–117 kJ mol⁻¹
–100 kJ mol⁻¹
–34 kJ mol⁻¹
Answer: 1
Question 8. Hess’s law is based on:
First law of thermodynamics
Second law of thermodynamics
Third law of thermodynamics
Zeroth law
Answer: 1
Question 9. Entropy is a measure of:
Heat content
Work capacity
Randomness
Free energy
Answer: 3
Question 10. Which process has ΔS < 0?
Melting of ice
Vaporization of water
Condensation of steam
Dissolution of salt
Answer: 3
Question 11. Gibbs free energy is represented by:
G = H + TS
G = H – TS
G = U + PV
G = U – TS
Answer: 2
Question 12. At equilibrium ΔG is:
Positive
Negative
Zero
Infinite
Answer: 3
Question 13. Which law defines temperature?
Zeroth law
First law
Second law
Third law
Answer: 1
Question 14. (Assertion–Reason)
Assertion (A): Heat is a path function.
Reason (R): Heat depends on path taken, not on initial and final states.
Answer: 1
Question 15. (Assertion–Reason)
Assertion (A): Enthalpy is a state function.
Reason (R): Its value depends only on initial and final states.
Answer: 1
Question 16. Standard enthalpy of formation of an element in its standard state is:
Zero
Positive
Negative
One
Answer: 1
⚡ Section B (Q17–Q21) – Very Short Answer (2 marks each, 5 × 2 = 10 marks)
Q17. Define system and surroundings.
🟦 System → part of universe under study.
🟩 Surroundings → rest of the universe that interacts with system.
Q18. Write first law of thermodynamics in mathematical form.
🟦 ΔU = q + w
🟩 Where ΔU = change in internal energy, q = heat supplied, w = work done.
Q19. State Hess’s law.
🟦 The enthalpy change of a reaction is the same, whether reaction occurs in one step or in multiple steps.
🟩 It depends only on initial and final states.
Q20. Calculate ΔH if ΔU = 50 J, Δn = 2 mol, R = 8.314 J mol⁻¹ K⁻¹, T = 300 K.
➤ Formula: ΔH = ΔU + ΔnRT
➤ Substitution: = 50 + (2 × 8.314 × 300)
➤ Calculation: = 50 + 4988.4 = 5038.4 J
✅ Final Answer: ΔH = 5038.4 J
Q21. Why entropy increases when ice melts?
🟦 Ice → ordered solid, water → disordered liquid.
🟩 Increase in randomness → ΔS > 0.
🧪 Section C (Q22–Q28) – Short Answer (3 marks each, 7 × 3 = 21 marks)
Q22. State three limitations of first law of thermodynamics.
🟦 It does not indicate feasibility of process.
🟨 It does not predict direction of process.
🟩 It does not give information about rate of process.
Q23. Differentiate between extensive and intensive properties with examples.
🟦 Extensive → depend on amount of substance (mass, volume, enthalpy).
🟨 Intensive → independent of amount (temperature, pressure, density).
🟩 Examples: Heat capacity (extensive), specific heat (intensive).
Q24. Explain standard enthalpy of formation with example.
🟦 Heat change when 1 mol of compound is formed from its elements in standard state.
🟨 Example: C(graphite) + O₂(g) → CO₂(g), ΔH = –393.5 kJ mol⁻¹.
🟩 Standard state = 298 K, 1 bar pressure.
Q25. Derive relation between ΔH and ΔU.
➤ At constant pressure: ΔH = ΔU + pΔV.
➤ For gases, pΔV = ΔnRT.
✅ Final: ΔH = ΔU + ΔnRT.
Q26. State second law of thermodynamics in terms of entropy.
🟦 For a spontaneous process, total entropy of system and surroundings increases.
🟩 ΔS_universe = ΔS_system + ΔS_surroundings > 0.
Q27. Define Gibbs free energy. Give criterion for spontaneity.
🟦 G = H – TS.
🟨 ΔG < 0 → process spontaneous. 🟩 ΔG = 0 → equilibrium; ΔG > 0 → non-spontaneous.
Q28. Explain enthalpy of neutralization with example.
🟦 Heat change when 1 mol of H⁺ reacts with 1 mol of OH⁻ to form water.
🟨 Strong acid + strong base → ΔH = –57 kJ mol⁻¹.
🟩 Example: HCl + NaOH → NaCl + H₂O.
🧭 Section D (Q29–Q30) – Case-Based Questions (4 marks each, 2 × 4 = 8 marks)
Q29. Read the passage and answer the questions:
The combustion of methane is highly exothermic. The enthalpy change depends on whether the process is carried out directly or in steps, but the overall enthalpy remains the same. This demonstrates Hess’s law.
(a) Write the chemical equation for combustion of methane. (1 mark)
(b) State Hess’s law. (1 mark)
(c) Explain why enthalpy change remains the same for direct and indirect routes. (2 marks)
🧪 Answer:
(a) CH₄(g) + 2O₂(g) → CO₂(g) + 2H₂O(l), ΔH = –890 kJ mol⁻¹
(b) Hess’s law: Enthalpy change is independent of path, depends only on initial and final states.
(c) Because enthalpy is a state function, it depends only on states, not on process steps.
Q30. Read the passage and answer the questions:
Entropy is a measure of disorder. For spontaneous processes such as melting of ice or expansion of gas, entropy increases. According to the second law of thermodynamics, ΔS_universe > 0 for spontaneity.
(a) Define entropy. (1 mark)
(b) What happens to entropy during freezing of water? (1 mark)
(c) State second law of thermodynamics in terms of entropy. (2 marks)
🧪 Answer:
(a) Entropy is a measure of randomness/disorder of a system.
(b) Entropy decreases because order increases when liquid turns into solid.
(c) For spontaneous process: ΔS_universe = ΔS_system + ΔS_surroundings > 0.
⚡ Section E (Q31–Q33) – Long Answer (5 marks each, 3 × 5 = 15 marks)
Q31. (a) Derive the relation between ΔH and ΔU for a gaseous reaction.
OR
(b) Calculate ΔH for the reaction: N₂(g) + 3H₂(g) → 2NH₃(g), given ΔU = –92 kJ, Δn = –2, T = 298 K, R = 8.314 J mol⁻¹ K⁻¹.
🧪 Answer (a):
➤ By definition: H = U + pV → ΔH = ΔU + Δ(pV).
➤ For gases: Δ(pV) = ΔnRT.
➤ So, ΔH = ΔU + ΔnRT.
✅ Relation derived: ΔH = ΔU + ΔnRT.
Answer (b):
➤ Formula: ΔH = ΔU + ΔnRT.
➤ Substitution: ΔU = –92 kJ, Δn = (2 – 4) = –2, T = 298 K.
➤ ΔH = –92 kJ + (–2 × 8.314 × 298)/1000 kJ.
➤ ΔH = –92 kJ – 4.95 kJ = –96.95 kJ.
✅ Final Answer: ΔH = –96.95 kJ.
Q32. (a) State and explain the second law of thermodynamics using Gibbs free energy.
OR
(b) Calculate ΔG for a reaction at 298 K if ΔH = –40 kJ mol⁻¹ and ΔS = –100 J mol⁻¹ K⁻¹. Comment on spontaneity.
🧪 Answer (a):
🟦 Second law: Natural processes tend to occur spontaneously.
🟨 In terms of Gibbs energy: ΔG = ΔH – TΔS.
🟩 If ΔG < 0 → process spontaneous. 🧪 ΔG = 0 → equilibrium, ΔG > 0 → non-spontaneous.
🎯 Thus Gibbs energy criterion predicts spontaneity.
Answer (b):
➤ Formula: ΔG = ΔH – TΔS.
➤ Convert ΔS: –100 J mol⁻¹ K⁻¹ = –0.100 kJ mol⁻¹ K⁻¹.
➤ ΔG = –40 – (298 × –0.100).
➤ ΔG = –40 + 29.8 = –10.2 kJ mol⁻¹.
✅ Final Answer: ΔG = –10.2 kJ mol⁻¹ → reaction is spontaneous.
Q33. (a) State and explain the third law of thermodynamics. Mention its significance.
OR
(b) Define enthalpy of formation, enthalpy of combustion, and enthalpy of neutralization with examples.
🧪 Answer (a):
🟦 Third law: Entropy of a perfectly crystalline substance is zero at 0 K.
🟨 Significance: Provides absolute scale of entropy values.
🟩 Used to calculate standard entropy changes and predict spontaneity of reactions.
🎯 Helps in determination of equilibrium constants.
Answer (b):
🟦 Enthalpy of formation: Heat change when 1 mol compound formed from elements (C + O₂ → CO₂, ΔH = –393.5 kJ mol⁻¹).
🟨 Enthalpy of combustion: Heat change when 1 mol substance completely burns (CH₄ + 2O₂ → CO₂ + 2H₂O, ΔH = –890 kJ mol⁻¹).
🟩 Enthalpy of neutralization: Heat change when 1 mol H⁺ reacts with 1 mol OH⁻ (HCl + NaOH → NaCl + H₂O, ΔH = –57 kJ mol⁻¹).
————————————————————————————————————————————————————————————————————————————
