Class 11 : Chemistry (In English) – Chapter 4: Chemical bonding and molecular Structure
EXPLANATION & SUMMARY
🔵 Introduction
All matter in the universe 🌍 is made up of atoms and molecules. But atoms cannot exist in isolation for long; they combine with each other to form stable structures called molecules and compounds. The force that holds atoms together is called a chemical bond.
The study of chemical bonding answers fundamental questions:
Why do atoms combine?
Why are some compounds stable and others reactive?
How do shapes of molecules influence properties?
This chapter builds on atomic structure and shows how bonding leads to the diversity of substances around us 🌟.
🟢 Why Do Atoms Combine?
Atoms combine to achieve stability. Noble gases (He, Ne, Ar) are stable because they have complete octets (8 electrons in outer shell). Other atoms combine to achieve similar stable configurations (octet rule).
🔑 Driving forces for combination:
Lowering of energy (stable state).
Attaining noble gas configuration (duplet for H, He).
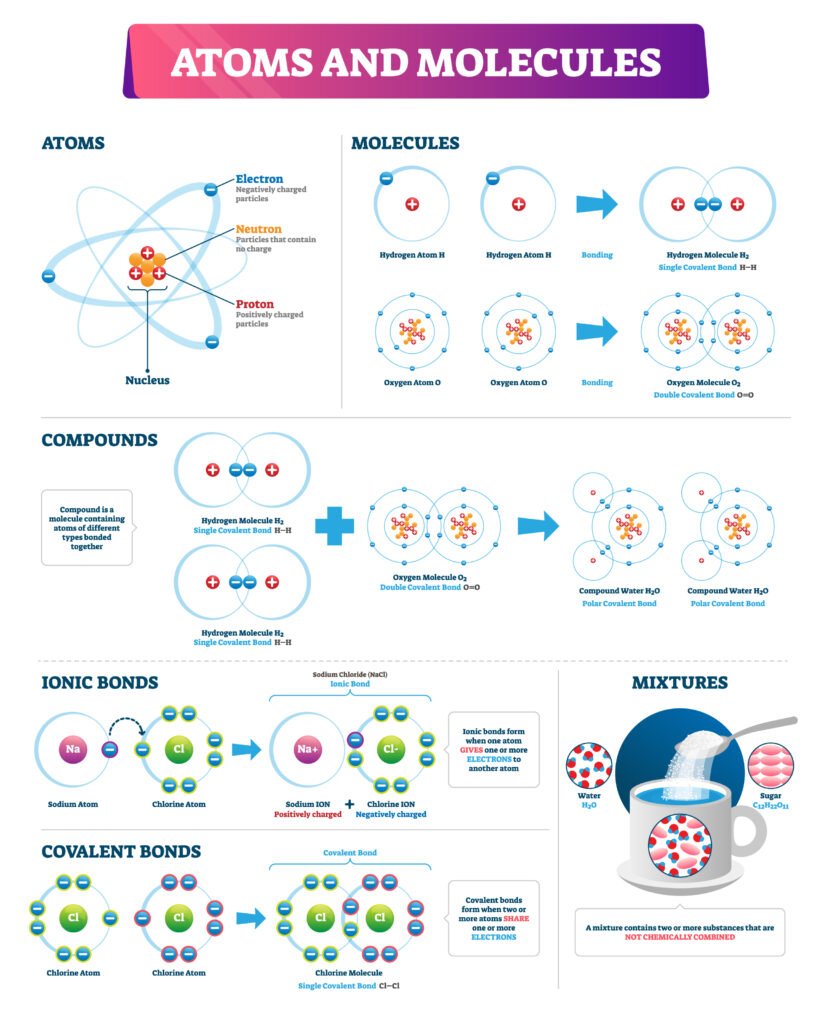
🔵 Types of Chemical Bonds
1️⃣ Ionic Bond (Electrovalent Bond)
Formed by complete transfer of electrons ⚡.
One atom loses electrons (forms cation), other gains (forms anion).
Held by strong electrostatic attraction.
Example: NaCl (Na⁺ + Cl⁻).

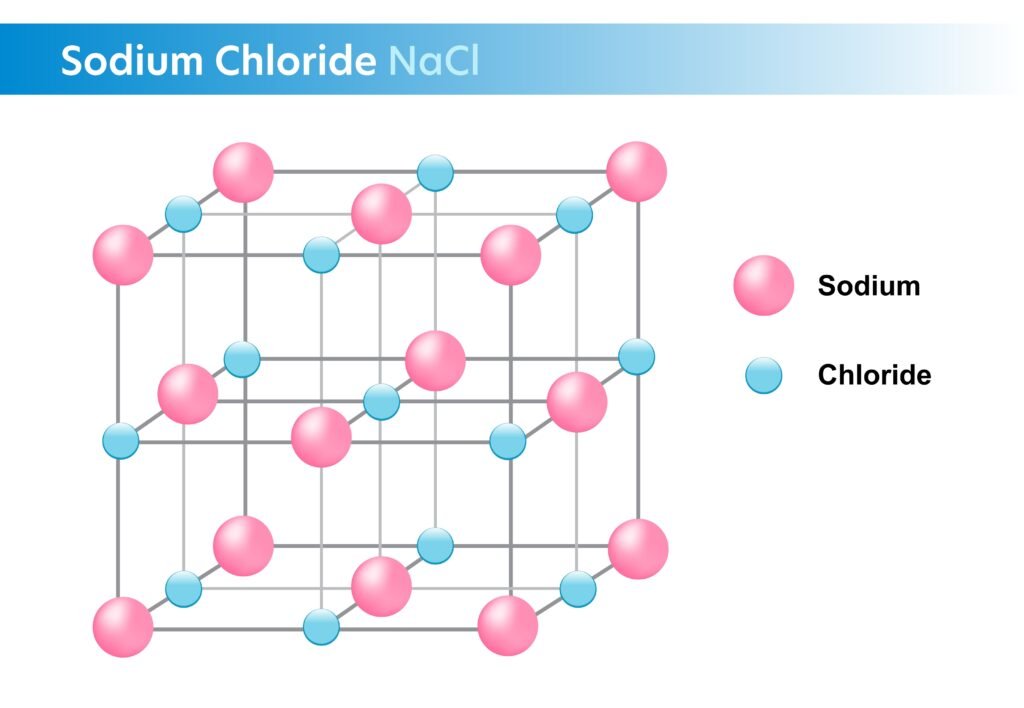
🔹 Properties:
Hard, brittle solids.
High melting and boiling points.
Conduct electricity in molten/aqueous state.
Soluble in polar solvents (e.g., water).
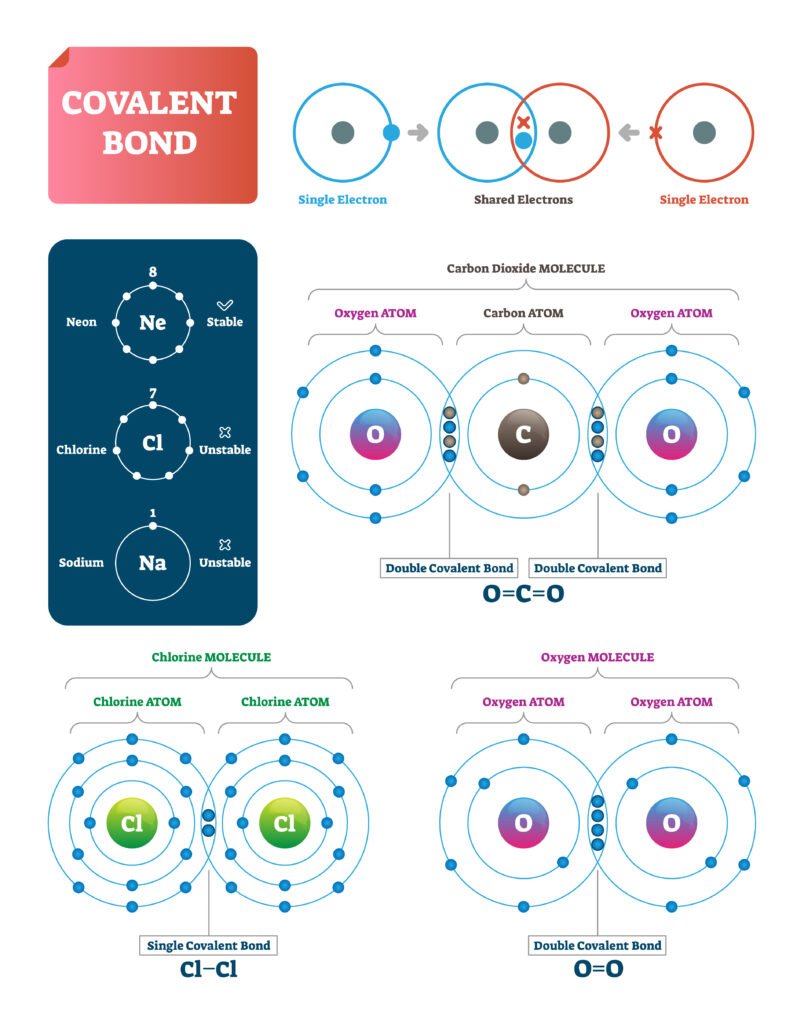
2️⃣ Covalent Bond
Formed by mutual sharing of electrons 🧩.
Shared pair constitutes the bond.
Example: H₂, O₂, CH₄.
🔹 Properties:
Gases, liquids, or soft solids.
Lower melting and boiling points.
Poor conductors of electricity.
Solubility depends on polarity.

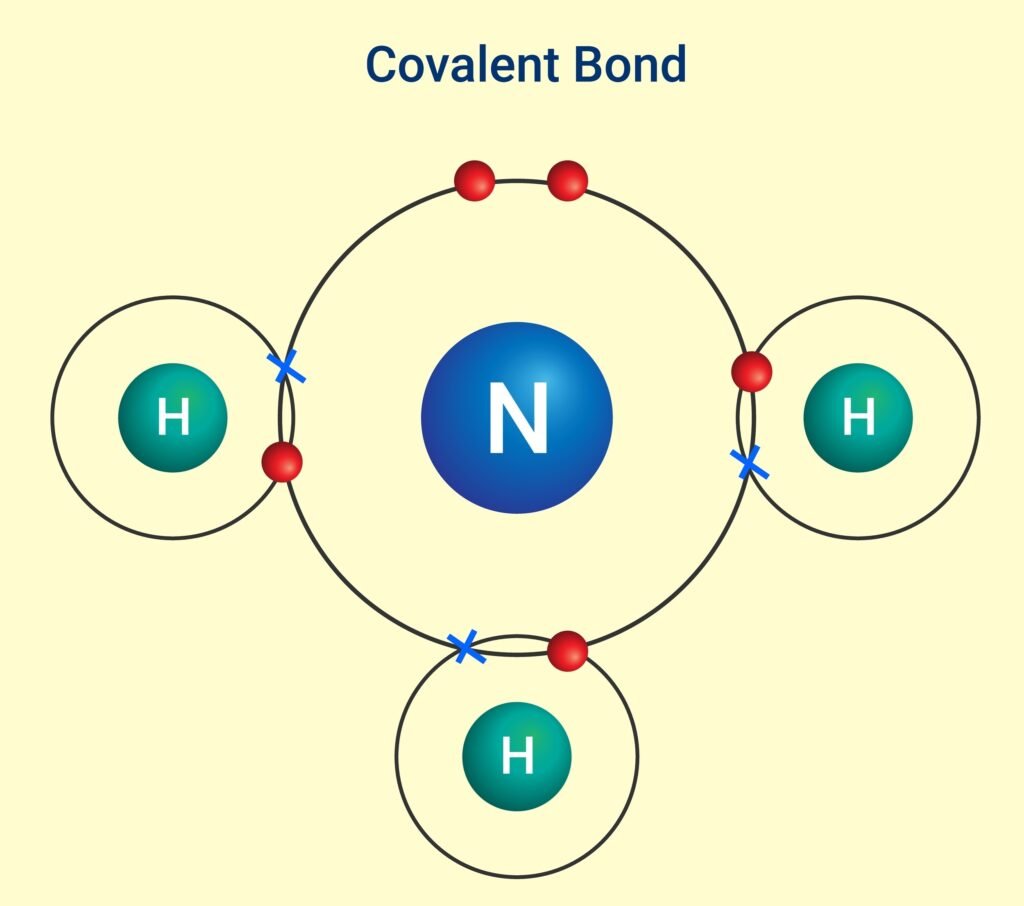
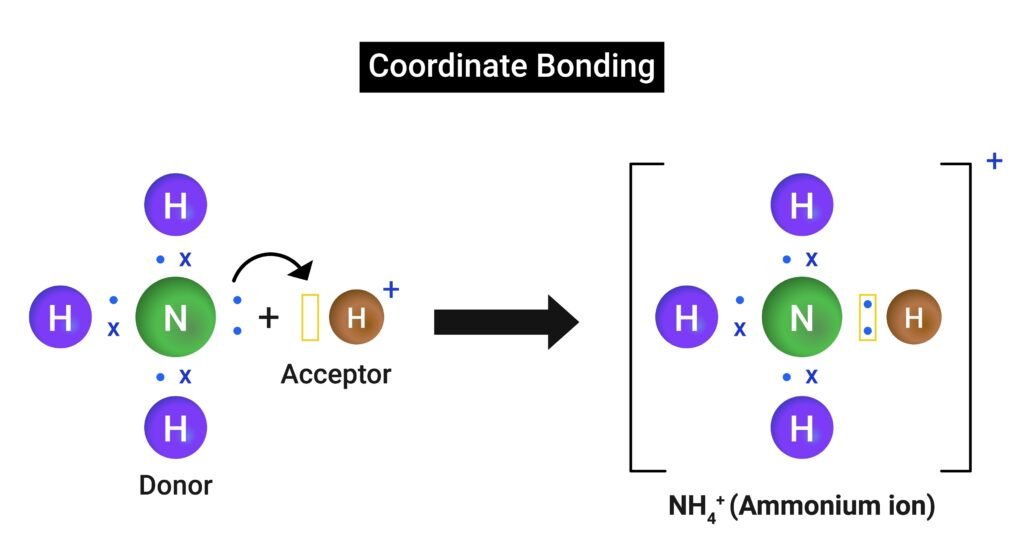
3️⃣ Coordinate (Dative) Bond
Both shared electrons contributed by one atom.
Example: NH₄⁺ ion (from NH₃ + H⁺).

🟡 Valence Bond Theory (VBT)
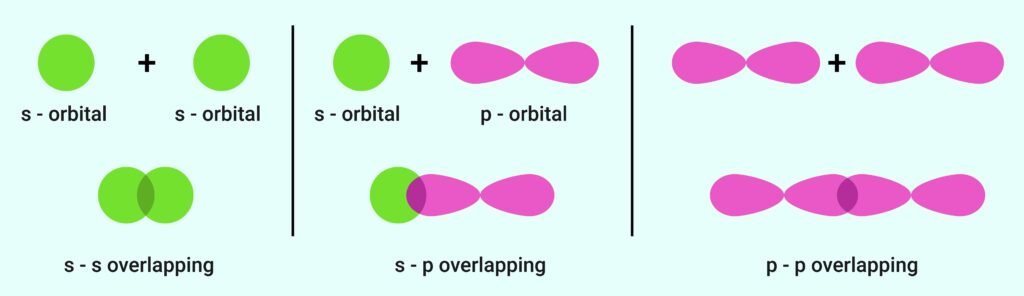
A chemical bond forms when atomic orbitals of atoms overlap.
Greater overlap → stronger bond.
Overlap types:
s–s overlap (H₂).
s–p overlap (HF).
p–p overlap (O₂).
Bond types:
Sigma (σ) bond → formed by head-on overlap, stronger.
Pi (π) bond → formed by sideways overlap, weaker.
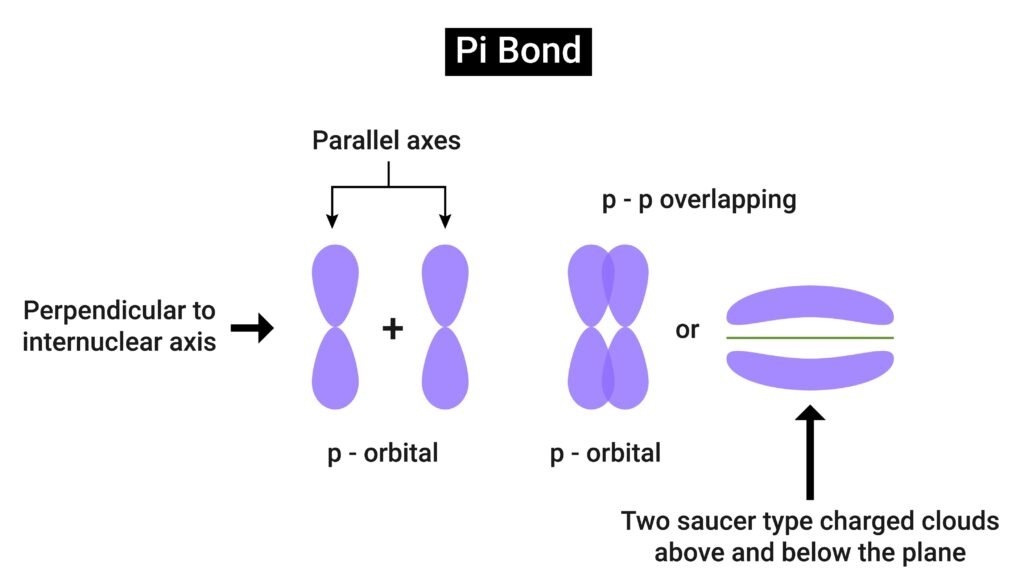
📌 Example: In O₂, one σ bond + one π bond.

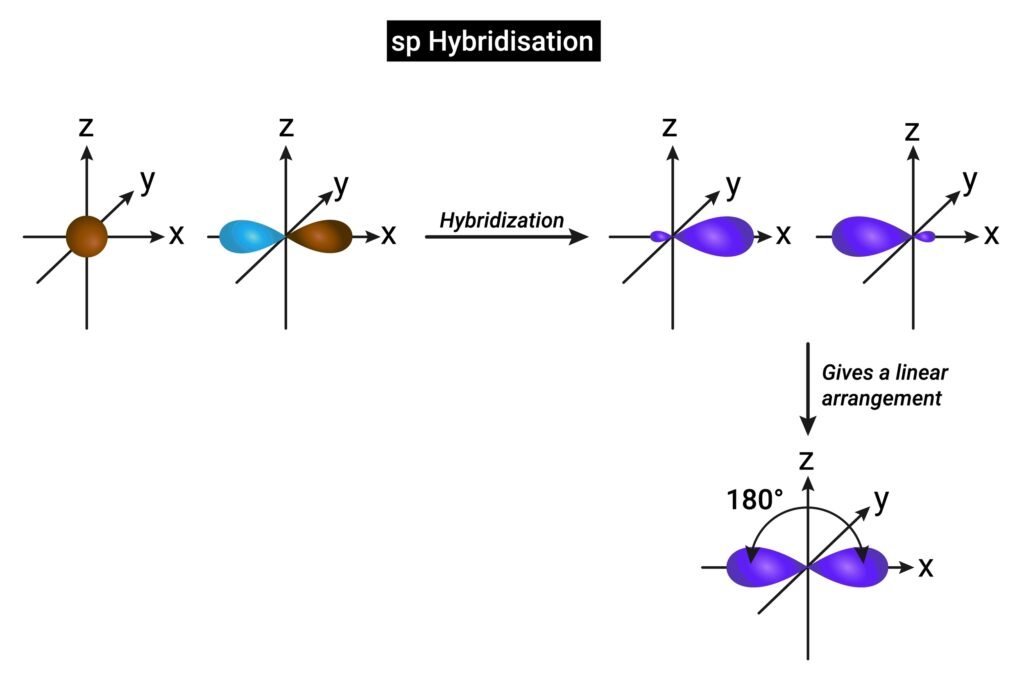
🔴 Hybridization
Hybridization is mixing of atomic orbitals of similar energies to form equivalent hybrid orbitals. It explains shape of molecules.
✔ Types of hybridization:
sp → linear (BeCl₂, 180°).
sp² → trigonal planar (BF₃, 120°).
sp³ → tetrahedral (CH₄, 109.5°).

sp³d → trigonal bipyramidal (PCl₅, 90° & 120°).
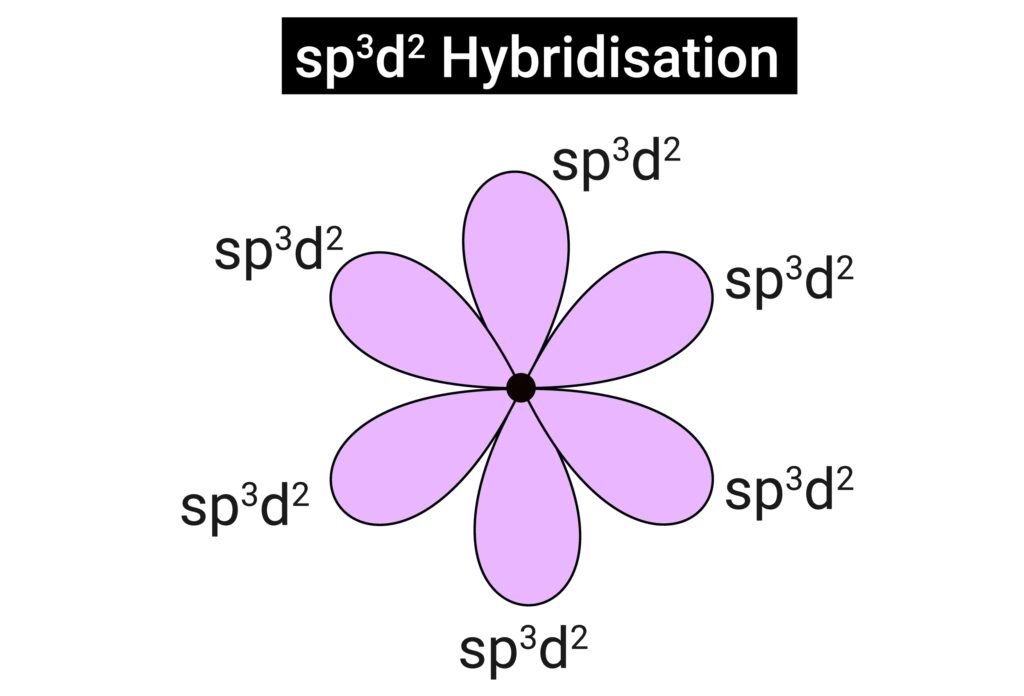
sp³d² → octahedral (SF₆, 90°).
💡 Lone pairs distort shapes:
NH₃ → sp³, pyramidal (107°).
H₂O → sp³, bent (104.5°).
🟢 Molecular Orbital Theory (MOT)
VBT had limitations → could not explain magnetism & bond order properly.
MOT by Hund & Mulliken (1932):
Atomic orbitals combine to form molecular orbitals (MOs).
Bonding MOs (lower energy) stabilize; antibonding MOs destabilize.
Bond order = ½ (Nb − Na).
✔ Examples:
H₂ → BO=1 (stable).
He₂ → BO=0 (unstable).
O₂ → BO=2 → paramagnetic (explained magnetism correctly).
🔵 Shapes of Molecules: VSEPR Theory
Valence Shell Electron Pair Repulsion (VSEPR) theory states that electron pairs (bonding + lone) around central atom repel each other. To minimize repulsion, they adopt maximum separation, giving molecular shape.
✔ Examples:
BeCl₂ → linear (180°).
BF₃ → trigonal planar (120°).
CH₄ → tetrahedral (109.5°).
NH₃ → pyramidal (107°).
H₂O → bent (104.5°).
PCl₅ → trigonal bipyramidal.
SF₆ → octahedral.
🟡 Bond Parameters
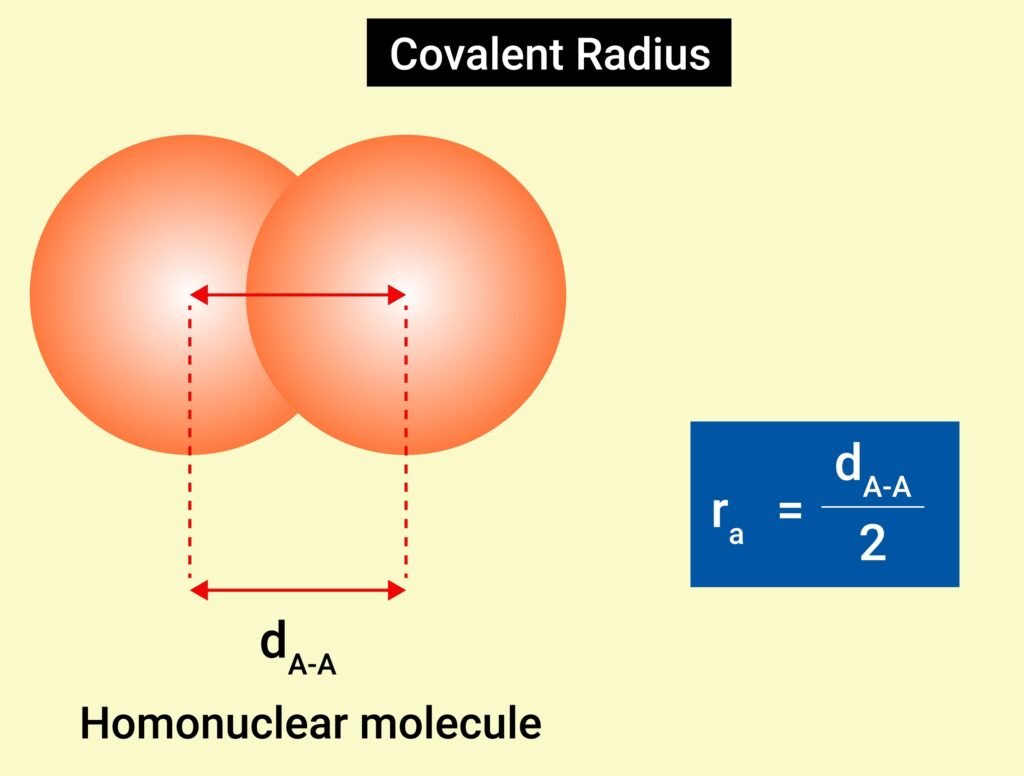
Bond Length
Distance between nuclei of bonded atoms.
Covalent radius trend: decreases with bond multiplicity.
Example: C–C single bond (154 pm), C=C (134 pm), C≡C (120 pm).
Bond Angle
Angle between orbitals of bonded atoms.
Depends on repulsions, hybridization.
Bond Enthalpy
Energy required to break one mole of bonds.
Multiple bonds stronger → higher enthalpy.
Bond Order
Number of bonds between atoms.
Higher BO → shorter, stronger bond.

🔴 Polarity of Bonds
Covalent bond may be polar (unequal sharing) or nonpolar (equal sharing).
Dipole moment (μ) = Q × d measures polarity.
Molecules with symmetrical shape may be nonpolar even if bonds are polar (e.g., CO₂).
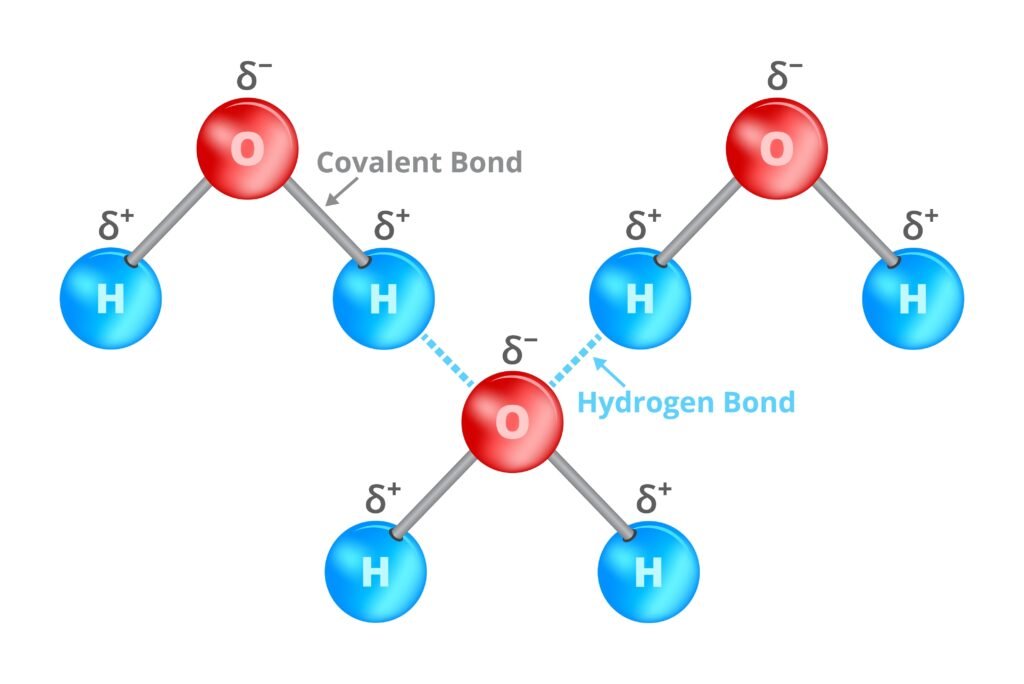
🟢 Hydrogen Bonding
Special bond between hydrogen attached to electronegative atom (N, O, F) and another electronegative atom.
Two types:
Intermolecular (between molecules, e.g., H₂O high boiling).
Intramolecular (within molecule, e.g., o-nitrophenol).
Consequences: higher boiling points, solubility, structure stabilization (DNA double helix 🧬).
🔵 Resonance
When one structure is not enough to explain bonding, a resonance hybrid (average of multiple structures) is considered.
Examples:
Ozone (O₃) ↔ two resonating structures.
Benzene (C₆H₆) ↔ Kekulé structures.
🟡 Applications of Bonding Concepts
✔ Explains stability of molecules.
✔ Determines shape and polarity (important in solubility, boiling point).
✔ Accounts for magnetism (O₂ paramagnetism explained by MOT).
✔ Explains unique properties of water, ammonia, HF (hydrogen bonding).
✔ Foundation for advanced chemistry: organic reactions, complex compounds, biochemistry.
✨ Conclusion
The chapter on Chemical Bonding and Molecular Structure is the heart of chemistry. It connects atomic structure to molecular properties, enabling us to predict shapes, polarity, reactivity, and strength of compounds. From simple molecules like H₂ to complex biological macromolecules, bonding explains the architecture of matter 🔬.
🔹 II. Lesson Summary
📌 Chemical Bonding and Molecular Structure – Key Takeaways
🔵 Why Bonding?
Atoms combine to attain stability (octet rule, duplet for H/He).
🟢 Types of Bonds
Ionic: electron transfer (NaCl).
Covalent: electron sharing (CH₄).
Coordinate: both electrons from one atom (NH₄⁺).
🔴 Theories
VBT: overlap of orbitals → σ and π bonds.
Hybridization: mixing orbitals → sp, sp², sp³, etc.
MOT: bonding/antibonding orbitals, bond order, magnetism.
VSEPR: electron pair repulsion → molecular shapes.
🟡 Bond Parameters
Length: distance between nuclei.
Angle: between bonds.
Enthalpy: energy needed to break.
Order: number of bonds (1, 2, 3).
🌟 Special Topics
Polarity: dipole moment, symmetry.
Hydrogen bonding: inter vs intra, explains high boiling, DNA stability.
Resonance: delocalized bonds (O₃, benzene).
📚 Molecular Shapes Examples
Linear: BeCl₂.
Trigonal planar: BF₃.
Tetrahedral: CH₄.
Pyramidal: NH₃.
Bent: H₂O.
Trigonal bipyramidal: PCl₅.
Octahedral: SF₆.
✨ Importance
Predicts structure, polarity, reactivity.
Explains water anomalies, O₂ magnetism.
Basis of organic, inorganic, and biological chemistry.
🔹 III. Quick Recap
📝 Quick Recap:
✔ Atoms bond to achieve octet stability.
✔ Bonds: ionic (transfer), covalent (sharing), coordinate (donation).
✔ VBT: orbital overlap → σ, π bonds.
✔ Hybridization: sp, sp², sp³ → shapes explained.
✔ MOT: bonding/antibonding orbitals, bond order, magnetism.
✔ VSEPR: molecular shapes depend on electron repulsions.
✔ Bond parameters: length, angle, enthalpy, order.
✔ Polarity, resonance, hydrogen bonding → unique behaviors.
✔ Explains stability, properties, reactivity of compounds.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
🔵 Question 4.1
Explain the formation of a chemical bond.
🟢 Answer:
✔ A chemical bond is a force that holds two atoms together to form a stable molecule.
✔ Atoms form bonds to achieve a stable electronic configuration (usually octet).
✔ Driving factors:
Lower potential energy
Attainment of noble gas configuration
✔ Main types:
Ionic bond – transfer of electrons (NaCl)
Covalent bond – sharing of electrons (H₂, O₂)
Coordinate bond – shared pair from one atom (NH₄⁺)
✨ Conclusion: Atoms combine to become more stable.
🔵 Question 4.2
Write Lewis dot symbols for atoms of the following elements:
Mg, Na, B, O, N, Br
🟢 Answer:
Lewis symbol = dots = number of valence electrons.
Mg (2 valence e⁻): Mg··
Na (1 e⁻): Na·
B (3 e⁻): B···
O (6 e⁻): O······
N (5 e⁻): N·····
Br (7 e⁻): Br·······
🔵 Question 4.3
Write Lewis symbols for the following atoms and ions:
S and S²⁻; Al and Al³⁺; H and H⁻
🟢 Answer:
S: 6 valence e⁻ → S······
S²⁻: gains 2 e⁻ → 8 dots → S········
Al: 3 valence e⁻ → Al···
Al³⁺: loses 3 e⁻ → no dots
H: 1 e⁻ → H·
H⁻: 2 e⁻ → H··
🔵 Question 4.4
Draw Lewis structures for:
H₂S, SiCl₄, BeF₂, CO₃²⁻, HCOOH
🟢 Answer:
✔ H₂S:
S has 6 e⁻ → bonds with 2 H → H–S–H, two lone pairs on S
✔ SiCl₄:
Si (4 e⁻) + 4 Cl (7 each) → four single bonds
Structure:
Cl
│
Cl–Si–Cl
│
Cl
✔ BeF₂:
Be (2 e⁻) forms 2 single bonds → F–Be–F, linear
✔ CO₃²⁻:
24 valence e⁻, one C double-bonded to one O, single to two O⁻
Resonance: three equivalent forms
✔ HCOOH (formic acid):
H–C(=O)–OH
🔵 Question 4.5
Define octet rule. Write its significance and limitations.
🟢 Answer:
✔ Octet rule: Atoms tend to gain/lose/share electrons to have 8 in outer shell.
✔ Significance: Explains stability and formation of molecules like NaCl, H₂O.
✔ Limitations:
Fails for odd-electron molecules (NO)
Fails for electron-deficient (BF₃)
Fails for expanded octet (SF₆)
🔵 Question 4.6
Write favourable factors for formation of ionic bond.
🟢 Answer:
✔ Low ionization enthalpy (cation formation easy)
✔ High electron gain enthalpy (anion formation easy)
✔ High lattice enthalpy (stabilizes solid)
Example: NaCl – Na⁺ + Cl⁻ → strong ionic bond
🔵 Question 4.7
Discuss shape of the following molecules using VSEPR model:
BeCl₂, BCl₃, SiCl₄, AsF₅, H₂S, PH₃
🟢 Answer:
BeCl₂: 2 bond pairs, 0 lone → linear
BCl₃: 3 bond pairs → trigonal planar
SiCl₄: 4 bond pairs → tetrahedral
AsF₅: 5 bond pairs → trigonal bipyramidal
H₂S: 2 bond + 2 lone → bent
PH₃: 3 bond + 1 lone → trigonal pyramidal
🔵 Question 4.8
Although NH₃ and H₂O are distorted tetrahedral, bond angle in water < ammonia. Discuss.
🟢 Answer:
✔ NH₃: 1 lone pair → bond angle 107°
✔ H₂O: 2 lone pairs → more repulsion → bond angle 104.5°
💡 More lone pairs → smaller angle.
🔵 Question 4.9
How do you express bond strength in terms of bond order?
🟢 Answer:
✔ Bond order (B.O.) = ½ (bonding – antibonding electrons)
✔ ↑ Bond order → ↑ bond strength → ↓ bond length
e.g. N₂ (B.O. = 3) > O₂ (B.O. = 2) > F₂ (B.O. = 1)
🔵 Question 4.10
Define bond length.
🟢 Answer:
✔ Bond length = equilibrium distance between nuclei of two bonded atoms.
Measured in pm.
e.g. H–H = 74 pm
Shorter bond → stronger.
🔵 Question 4.11
Explain resonance with reference to CO₃²⁻.
🟢 Answer:
✔ CO₃²⁻ has 3 equivalent structures; true structure is resonance hybrid.
✔ Each C–O bond = same length, B.O. = 4/3.
💡 Resonance increases stability.
🔵 Question 4.12
H₃PO₃ can be represented by two structures. Can these be canonical forms?
🟢 Answer:
❌ No.
Because in (1) and (2), bonding arrangement differs (one has P=O, other has P–O–H), atoms linked differently → not resonance → tautomers, not canonical forms.
🔵 Question 4.13
Write resonance structures of SO₃, NO₂, NO₃⁻.
🟢 Answer:
✔ SO₃: three equivalent structures, S double bonded with each O once.
✔ NO₂: two forms with one double, one single bond.
✔ NO₃⁻: three equivalent forms; each N–O bond = 1⅓ order.
🔵 Question 4.14
Use Lewis symbols to show electron transfer in:
(a) K and S (b) Ca and O (c) Al and N
🟢 Answer:
(a) K· + :S: → K₂S (K⁺, S²⁻)
(b) Ca·· + :O: → CaO (Ca²⁺, O²⁻)
(c) Al··· + :N: → AlN (Al³⁺, N³⁻)
Each forms stable ionic compounds by complete transfer.
🔵 Question 4.15
Although both CO₂ and H₂O are triatomic molecules, the shape of H₂O molecule is bent while that of CO₂ is linear. Explain on the basis of dipole moment.
🟢 Answer:
✔ CO₂: O=C=O is linear, bond dipoles equal & opposite → cancel out → μ = 0 (non-polar)
✔ H₂O: H–O–H is bent, bond dipoles do not cancel → μ ≠ 0 (polar)
✨ Therefore, CO₂ is non-polar, H₂O is polar.
🔵 Question 4.16
Write the significance/applications of dipole moment.
🟢 Answer:
✔ Measures polarity of a molecule
✔ Helps to predict shape (μ = 0 → symmetrical, linear)
✔ Differentiates polar and non-polar molecules
✔ Explains bond character (μ ∝ % ionic character)
🔵 Question 4.17
Define electronegativity. How does it differ from electron gain enthalpy?
🟢 Answer:
✔ Electronegativity = tendency of an atom to attract shared pair of electrons in a bond.
✔ Electron gain enthalpy = energy change when an isolated atom gains electron.
💡 Difference:
EN → property of bonded atom
ΔegH → property of isolated atom
🔵 Question 4.18
Explain with an example polar covalent bond.
🟢 Answer:
✔ When shared pair is unequally shared → bond has partial charges.
Example: H–Cl
→ Cl more electronegative → δ⁻, H → δ⁺
→ Polar covalent bond
🔵 Question 4.19
Arrange the bonds in order of increasing ionic character:
LiF, K₂O, N₂, SO₂, ClF₃
🟢 Answer:
✔ Greater ΔEN → more ionic.
ΔEN: N₂ (0) < SO₂ < ClF₃ < K₂O < LiF
✅ Order: N₂ < SO₂ < ClF₃ < K₂O < LiF
🔵 Question 4.20
Write correct Lewis structure of acetic acid (CH₃COOH).
🟢 Answer:
✔ Structure:
H
│
H–C–H
│
C=O
│
O–H
💡 Contains 4 single bonds and 1 double bond.
🔵 Question 4.21
Apart from tetrahedral, CH₄ can’t be square planar. Why?
🟢 Answer:
✔ Square planar needs d orbitals, but C lacks d orbitals (only s & p).
✔ sp³ hybridization → tetrahedral only.
🔵 Question 4.22
Explain why BeH₂ has zero dipole moment though Be–H bonds are polar.
🟢 Answer:
✔ BeH₂ is linear.
✔ Bond dipoles equal & opposite → cancel.
→ μ = 0 though each bond is polar.
🔵 Question 4.23
Which has higher dipole moment: NH₃ or NF₃?
🟢 Answer:
✔ NH₃: μ vectors of bonds + lone pair in same direction → μ high (1.46 D)
✔ NF₃: μ vectors oppose → partial cancellation → μ lower (0.24 D)
✅ NH₃ > NF₃
🔵 Question 4.24
What is meant by hybridization? Describe sp, sp², sp³ hybrid orbitals.
🟢 Answer:
✔ Hybridization: Mixing of atomic orbitals of similar energy to form new equivalent orbitals.
sp: 1s + 1p → 2 linear orbitals (180°)
sp²: 1s + 2p → 3 trigonal planar (120°)
sp³: 1s + 3p → 4 tetrahedral (109.5°)
🔵 Question 4.25
Describe change in hybridisation of Al in:
AlCl₃ + Cl⁻ → AlCl₄⁻
🟢 Answer:
✔ AlCl₃ → sp² (3 bonds)
✔ AlCl₄⁻ → accepts lone pair → sp³ (4 bonds)
✨ Hybridisation changes from sp² → sp³
🔵 Question 4.26
Change in hybridisation of B and N in:
BF₃ + NH₃ → F₃B←NH₃
🟢 Answer:
✔ B (in BF₃): sp² → after bonding → sp³
✔ N (in NH₃): sp³ → remains sp³
🔵 Question 4.27
Draw formation of double and triple bonds in C₂H₄ and C₂H₂.
🟢 Answer:
✔ C₂H₄ (Ethene):
Each C: sp² → 1 σ (C–C) + 1 π
→ structure: H₂C=CH₂
✔ C₂H₂ (Ethyne):
Each C: sp → 1 σ + 2 π
→ structure: HC≡CH
🔵 Question 4.28
Total number of σ and π bonds:
(a) C₂H₂ (b) C₂H₄
🟢 Answer:
(a) C₂H₂:
1 C–C σ + 2 π, 2 C–H σ → σ = 3, π = 2
(b) C₂H₄:
1 C–C σ + 1 π, 4 C–H σ → σ = 5, π = 1
🔵 Question 4.2
Considering x-axis as the internuclear axis, which out of the following will not form a σ (sigma) bond and why?
(a) 1s and 1s
(b) 1s and 2pₓ
(c) 2pₓ and 2pₓ
(d) 1s and 2s
🟢 Answer:
✔ A σ bond is formed by end-to-end overlap of orbitals along the internuclear axis (x-axis).
(a) 1s–1s → yes, forms σ bond
(b) 1s–2pₓ → yes, forms σ bond
(c) 2pₓ–2pₓ → yes, forms σ bond
(d) 1s–2s → yes, forms σ bond
✅ All can form σ bond, none is excluded.
🔵 Question 4.30
Which hybrid orbitals are used by carbon atoms in the following molecules?
(a) CH₃–CH₃
(b) CH₂=CH₂
(c) CH≡CH
(d) CH₃CHO (acetaldehyde)
(e) CH₃COOH (acetic acid)
🟢 Answer:
✔ (a) CH₃–CH₃ → all C atoms sp³
✔ (b) CH₂=CH₂ → each C sp²
✔ (c) CH≡CH → each C sp
✔ (d) CH₃CHO → CH₃ (sp³), CHO carbon (sp²)
✔ (e) CH₃COOH → CH₃ (sp³), COOH carbon (sp²)
🔵 Question 4.31
What do you understand by bond pairs and lone pairs of electrons? Give one example of each.
🟢 Answer:
✔ Bond pair: electrons shared between two atoms forming a covalent bond.
Example: in H–Cl, one shared pair = bond pair.
✔ Lone pair: non-bonding pair localized on one atom.
Example: in H₂O, oxygen has 2 lone pairs.
🔵 Question 4.32
Distinguish between a σ bond and a π bond.
🟢 Answer:
Feature σ bond π bond
Formation End-to-end overlap Sidewise overlap
Strength Stronger Weaker
Rotation Free Restricted
Example H–H in H₂ C=C in C₂H₄ (one σ, one π)
🔵 Question 4.33
Explain the formation of H₂ molecule on the basis of valence bond theory (VBT).
🟢 Answer:
✔ Each H atom has 1s¹ configuration.
✔ Two 1s orbitals overlap head-on to form one σ bond.
✔ Electron density increases between nuclei → lowers energy → stable H₂.
🔵 Question 4.34
Write the important conditions required for the linear combination of atomic orbitals to form molecular orbitals.
🟢 Answer:
✔ Orbitals must have same energy.
✔ Proper symmetry about internuclear axis.
✔ Maximum overlap possible.
✨ These ensure effective bonding/antibonding MO formation.
🔵 Question 4.35
Use molecular orbital theory to explain why Be₂ molecule does not exist.
🟢 Answer:
✔ Be: 1s² 2s²
✔ MO filling: σ1s² σ1s² σ2s² σ2s²
✔ Bond order = (Nb–Na)/2 = (4–4)/2 = 0
✅ Zero bond order → Be₂ unstable, does not exist.
🔵 Question 4.36
Compare relative stability and magnetic property of O₂, O₂⁺, O₂⁻, O₂²⁻.
🟢 Answer:
Species Bond Order Magnetic Stability
O₂ 2 Paramagnetic (2 unpaired) Stable
O₂⁺ 2.5 Paramagnetic More stable
O₂⁻ 1.5 Paramagnetic Less stable
O₂²⁻ 1 Diamagnetic Least stable
✔ Higher bond order → more stability.
🔵 Question 4.37
Write the significance of ‘+’ and ‘–’ sign in representing orbitals.
🟢 Answer:
✔ They show phase of wave function (ψ).
✔ In bonding overlap: same sign → constructive, bonding MO.
✔ Opposite sign → destructive, antibonding MO.
🔵 Question 4.38
Describe hybridisation in PCl₅. Why are axial bonds longer than equatorial?
🟢 Answer:
✔ P: 3s²3p³ + promotion → 3s¹3p³3d¹ → sp³d
✔ Geometry: trigonal bipyramidal
→ 3 equatorial (120°), 2 axial (90°)
✔ Axial bonds face more repulsion → longer & weaker.
🔵 Question 4.39
Define hydrogen bond. Is it weaker or stronger than van der Waals forces?
🟢 Answer:
✔ Attractive force between H (bonded to F, O, N) and lone pair of nearby electronegative atom.
✔ Stronger than van der Waals forces, but weaker than covalent bond.
🔵 Question 4.40
What is meant by bond order? Calculate bond order of N₂, O₂, O₂⁺ and O₂²⁻.
🟢 Answer:
✔ Bond order = ½ (Nb – Na)
N₂: (10–4)/2 = 3
O₂: (10–6)/2 = 2
O₂⁺: (10–5)/2 = 2.5
O₂²⁻: (10–8)/2 = 1
✔ Higher bond order → stronger bond → shorter bond length.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
✳ Section A (Q1–Q16) – MCQs (1 mark each, 16 × 1 = 16 marks)
Options:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Question 1. Which type of bond is present in NaCl?
Covalent bond
Ionic bond
Metallic bond
Coordinate bond
Answer: 2
Question 2. Which among the following molecules has triple bond?
O₂
N₂
Cl₂
F₂
Answer: 2
Question 3. The molecule with linear shape is:
H₂O
NH₃
CO₂
BF₃
Answer: 3
Question 4. Hybridization of carbon in methane (CH₄) is:
sp
sp²
sp³
dsp²
Answer: 3
Question 5. Which among the following has bent shape?
CO₂
H₂O
BeCl₂
BF₃
Answer: 2
Question 6. Which molecule shows resonance?
CH₄
O₃
H₂O
NH₄⁺
Answer: 2
Question 7. Which bond has highest polarity?
H–H
H–F
H–Cl
H–Br
Answer: 2
Question 8. Which compound shows coordinate bond?
H₂O
NH₄⁺
Cl₂
NaCl
Answer: 2
Question 9. VSEPR theory is used to predict:
Bond length
Bond energy
Molecular shape
Bond order
Answer: 3
Question 10. Bond order of O₂ molecule according to MOT is:
1
2
3
2.5
Answer: 2
Question 11. Paramagnetic molecule is:
O₂
N₂
CO₂
H₂O
Answer: 1
Question 12. Which orbital overlap gives strongest bond?
s–s
s–p
p–p (axial)
p–p (sideways)
Answer: 3
Question 13. Dipole moment of CO₂ is zero because:
It has non-polar bonds
It has polar bonds but linear structure
It has ionic bonds
It has weak bonds
Answer: 2
Question 14. (Assertion–Reason)
Assertion (A): NH₃ has pyramidal shape.
Reason (R): Nitrogen has lone pair of electrons which repels bond pairs.
Answer: 1
Question 15. (Assertion–Reason)
Assertion (A): O₂ is paramagnetic.
Reason (R): According to MOT, O₂ has two unpaired electrons in antibonding orbitals.
Answer: 1
Question 16. Which of the following is isostructural with CO₂?
SO₂
NO₂⁻
BeCl₂
H₂O
Answer: 3
⚡ Section B (Q17–Q21) – Very Short Answer (2 marks each, 5 × 2 = 10 marks)
Q17. Define octet rule with one example.
🟦 Atoms tend to gain, lose or share electrons to acquire stable configuration of 8 electrons in valence shell.
🟩 Example: NaCl forms by transfer of 1 electron from Na to Cl.
Q18. Differentiate between sigma and pi bond (two points).
🟦 Sigma bond – formed by head-on overlap; stronger.
🟩 Pi bond – formed by sideways overlap; weaker.
Q19. What is bond order? Write formula.
🟦 Bond order = (Number of bonding electrons – Number of antibonding electrons)/2.
🟩 Greater bond order → stronger bond, shorter bond length.
Q20. Calculate bond order of N₂ using MOT.
➤ Bonding electrons = 10, Antibonding electrons = 4
➤ Bond order = (10 – 4)/2 = 3
✅ Final Answer: Bond order = 3
Q21. Why does H₂O have bent shape according to VSEPR theory?
🟦 Oxygen has 2 lone pairs and 2 bond pairs.
🟩 Lone pair–bond pair repulsion > bond pair–bond pair → bent structure.
🧪 Section C (Q22–Q28) – Short Answer (3 marks each, 7 × 3 = 21 marks)
Q22. State three limitations of octet rule.
🟦 Incomplete octet (H, Be).
🟨 Expanded octet (P, S).
🟩 Odd-electron molecules (NO, NO₂).
Q23. Explain Fajan’s rule with one example.
🟦 Smaller cation and larger anion → more covalent character.
🟨 Example: LiI more covalent than LiF.
🟩 Greater polarization → greater covalency.
Q24. Write the molecular orbital electronic configuration of O₂ and predict its magnetic behaviour.
🟦 O₂ (Z = 8): (σ1s)² (σ1s)² (σ2s)² (σ2s)² (σ2pz)² (π2px = 2, π2py = 2) (π2px = 1, π2py = 1).
🟨 Two unpaired electrons present.
✅ Hence O₂ is paramagnetic.
Q25. State and explain VSEPR theory with example of BF₃.
🟦 Electron pairs around central atom repel each other.
🟨 Shape depends on minimizing repulsion.
🟩 In BF₃: 3 bond pairs → trigonal planar shape.
Q26. What are resonance structures? Give one example.
🟦 Structures with same arrangement of atoms but different arrangement of electrons.
🟨 Actual molecule is hybrid of resonance structures.
🟩 Example: O₃, NO₃⁻.
Q27. Explain formation of NH₄⁺ ion.
🟦 N atom shares 3 electrons with H atoms → NH₃.
🟨 Lone pair on N donated to H⁺ → coordinate bond.
✅ NH₄⁺ formed with tetrahedral structure.
Q28. State three differences between bonding in H₂ and He₂ according to MOT.
🟦 H₂: bond order = 1 → stable molecule.
🟨 He₂: bond order = 0 → unstable, does not exist.
🟩 Reason: antibonding orbitals cancel bonding in He₂.
🧭 Section D (Q29–Q30) – Case-Based Questions (4 marks each, 2 × 4 = 8 marks)
Q29. Read the passage and answer the questions:
The bond in hydrogen chloride (HCl) is polar covalent due to the difference in electronegativity of H and Cl. The molecule has a dipole moment of 1.08 D, showing partial charges.
(a) What type of bond is present in HCl? (1 mark)
(b) Why is the bond polar? (1 mark)
(c) Explain significance of dipole moment in HCl. (2 marks)
🧪 Answer:
(a) Polar covalent bond.
(b) Because Cl is more electronegative than H, electron pair is shifted towards Cl.
(c) Dipole moment shows extent of polarity → partial positive charge on H and negative charge on Cl.
Q30. Read the passage and answer the questions:
According to Molecular Orbital Theory (MOT), electronic configurations of molecules can be used to calculate bond order and predict stability. For example, O₂ molecule has bond order 2 and is paramagnetic.
(a) Write the formula for bond order. (1 mark)
(b) Calculate bond order of O₂. (1 mark)
(c) Why is O₂ paramagnetic according to MOT? (2 marks)
🧪 Answer:
(a) Bond order = (Number of bonding electrons – Number of antibonding electrons)/2.
(b) O₂: Bonding e⁻ = 10, Antibonding e⁻ = 6 → BO = (10 – 6)/2 = 2.
(c) O₂ has two unpaired electrons in π2px and π2py orbitals → paramagnetic.
⚡ Section E (Q31–Q33) – Long Answer (5 marks each, 3 × 5 = 15 marks)
Q31. (a) Discuss the main postulates of Valence Bond Theory (VBT) and explain hybridization in CH₄.
OR
(b) Explain the formation of σ and π bonds with neat description.
🧪 Answer (a):
🟦 VBT: A covalent bond forms due to overlap of half-filled orbitals.
🟨 Bond strength ∝ overlap extent.
🟩 Hybridization: Mixing of atomic orbitals to form new hybrid orbitals of equal energy.
🧪 In CH₄: C undergoes sp³ hybridization → 4 sp³ orbitals form 4 σ bonds with H.
🎯 Shape: Tetrahedral, bond angle 109.5°.
Answer (b):
🟦 σ-bond: head-on overlap of orbitals (s–s, s–p, p–p axial). Strongest bond.
🟨 π-bond: sideways overlap of p-orbitals after σ bond is formed.
🎯 Example: N₂ has one σ and two π bonds.
Q32. (a) State VSEPR theory. Predict the shapes of (i) BeCl₂, (ii) BF₃, (iii) NH₃, (iv) H₂O.
OR
(b) Discuss Fajan’s rule and factors influencing covalent character in ionic compounds.
🧪 Answer (a):
🟦 VSEPR theory: Shape depends on repulsions between electron pairs in valence shell of central atom.
(i) BeCl₂: 2 bond pairs → Linear (180°).
(ii) BF₃: 3 bond pairs → Trigonal planar (120°).
(iii) NH₃: 3 bond pairs + 1 lone pair → Trigonal pyramidal (107°).
(iv) H₂O: 2 bond pairs + 2 lone pairs → Bent (104.5°).
Answer (b):
🟦 Fajan’s rule: Small, highly charged cation + large, polarizable anion → more covalent character.
🟨 Factors: (i) Size of cation (smaller → more covalent),
(ii) Charge of cation (higher → more covalent),
(iii) Size of anion (larger → more covalent).
🎯 Example: AlCl₃ shows covalent character.
Q33. (a) Explain Molecular Orbital Theory with energy-level diagram up to O₂ molecule. Discuss stability of O₂ and He₂.
OR
(b) Define resonance. Explain resonance in O₃ molecule and its significance.
🧪 Answer (a):
🟦 MOT: Atomic orbitals combine to form bonding and antibonding molecular orbitals.
🟨 Filling follows Aufbau principle, Pauli’s principle, Hund’s rule.
🟩 O₂ (Z = 8): BO = 2, two unpaired electrons → stable, paramagnetic.
🧪 He₂: BO = 0 → unstable, does not exist.
🎯 MOT explains stability and magnetic behaviour better than VBT.
Answer (b):
🟦 Resonance: When a molecule cannot be represented by a single structure but two or more canonical structures.
🟨 Example: O₃ → O=O–O ↔ O–O=O.
🟩 Actual structure is resonance hybrid, with bond order = 1.5.
🎯 Resonance increases stability and delocalization of electrons.
————————————————————————————————————————————————————————————————————————————
