Class 11 : Chemistry (In English) – Chapter 2: Structure of Atom Chapter
EXPLANATION & SUMMARY
🔵 Introduction to the Structure of Atom
Atoms are the basic building blocks of matter 🌍. Everything around us is made of atoms – the chair, the air, our body, even stars ✨. The concept of atoms is ancient; the word “atom” comes from the Greek word “atomos,” meaning indivisible. The Indian philosopher Maharishi Kanad also described the concept of “anu” centuries ago. But modern chemistry and physics have refined this idea with scientific experiments.
Originally, atoms were believed to be indivisible, but as science advanced, it became clear that atoms are made up of subatomic particles ⚛ like electrons, protons, and neutrons. Understanding how these particles are arranged gives rise to the structure of atom. This chapter explores how scientists discovered the atom’s internal structure step by step, through experiments and theories.
🟢 Discovery of Subatomic Particles
1️⃣ Electrons (e⁻)
Discovered by J. J. Thomson in 1897 using cathode ray tube experiments.
When high voltage is applied across a vacuum tube, cathode rays (stream of negatively charged particles) are produced.
These rays were deflected by electric and magnetic fields, proving that they were negatively charged.
Charge of electron: −1.602 × 10⁻¹⁹ C, mass: 9.1 × 10⁻³¹ kg.
💡 Symbol: e⁻
2️⃣ Protons (p⁺)
Discovered by E. Goldstein in 1886 via canal rays (anode rays).
These rays were deflected opposite to electrons, meaning they carried positive charge.
Charge of proton: +1.602 × 10⁻¹⁹ C, mass ≈ 1.67 × 10⁻²⁷ kg (about 1836 times heavier than electron).
3️⃣ Neutrons (n⁰)
Discovered by James Chadwick in 1932.
Neutrons are neutral, with no charge.
Mass similar to protons.
Crucial in providing stability to nucleus and in nuclear reactions.
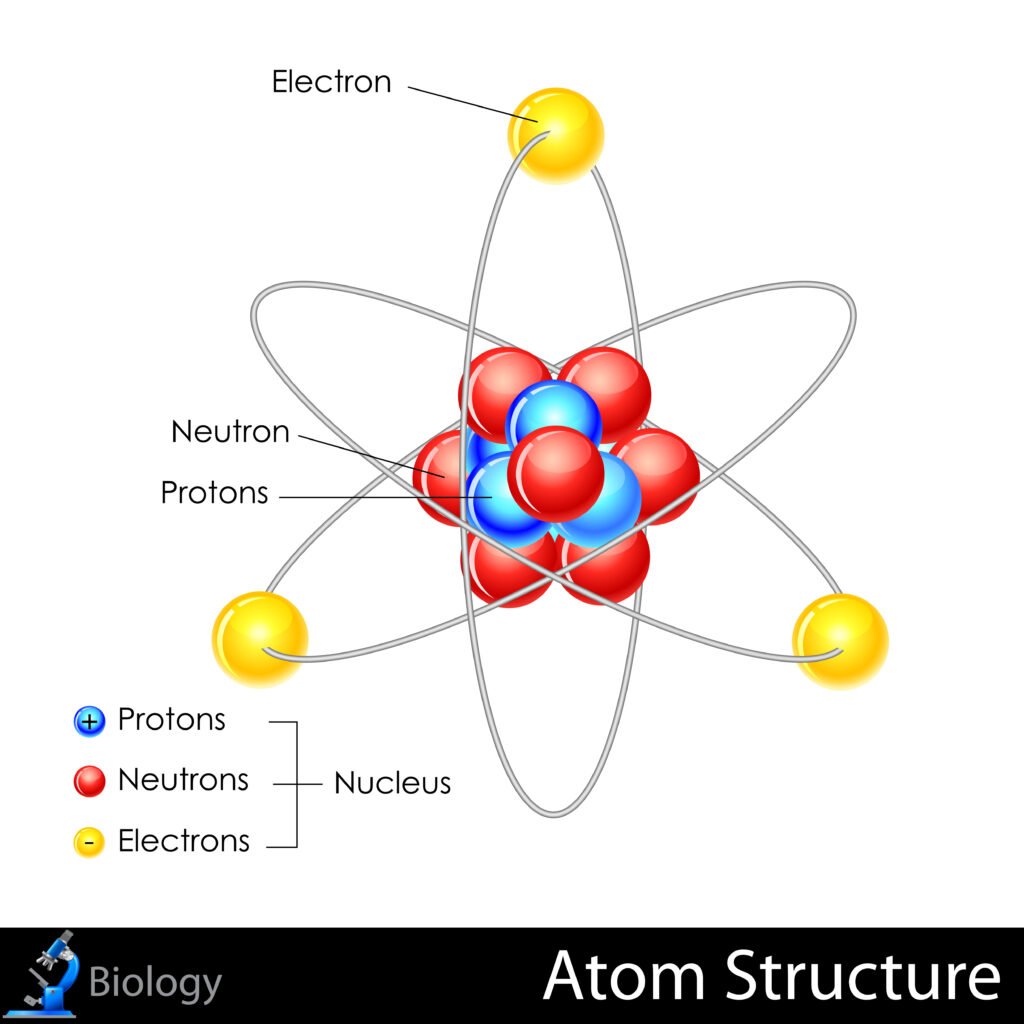
📌 Atoms consist of electrons (negative) revolving outside, and protons + neutrons (positive + neutral) in a dense nucleus.
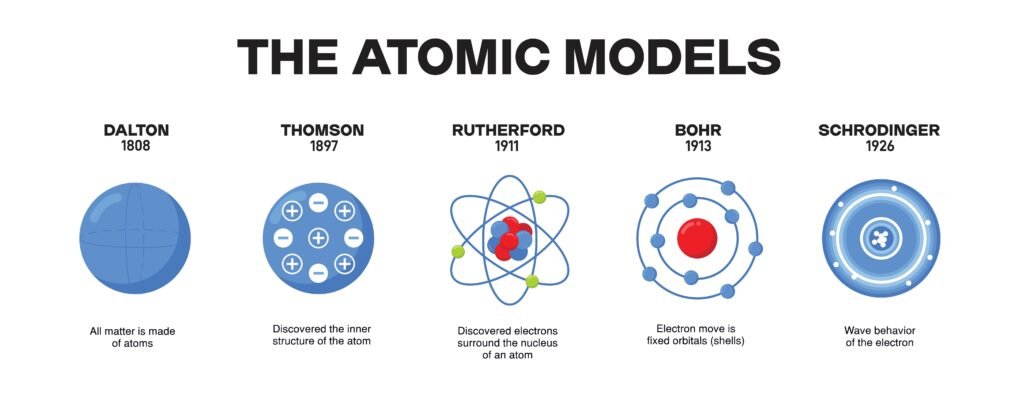
🔵 Early Models of the Atom
1️⃣ Thomson’s Model (Plum Pudding Model, 1898)
Atom imagined as a sphere of positive charge ☀ in which electrons were embedded like seeds in a watermelon or plums in pudding.
Failed because it could not explain experimental results like Rutherford’s scattering experiment.
2️⃣ Rutherford’s Nuclear Model (1911)
Based on alpha particle scattering experiment.
Rutherford bombarded thin gold foil with α-particles (helium nuclei).
Findings:
Most α-particles passed undeflected → atom is mostly empty space.
Few deflected at small angles → presence of positive charge.
Very few deflected at large angles → tiny dense positively charged nucleus.
Model: atom consists of nucleus (protons + neutrons) at center, electrons revolve around like planets around sun 🌞.
❌ Limitation: Electrons in orbit should lose energy and spiral into nucleus, but this doesn’t happen.
🟢 Bohr’s Model of Atom (1913)
Bohr improved Rutherford’s model using quantum theory:
✔ Electrons revolve around nucleus in fixed energy levels (orbits) without radiating energy.
✔ These levels are quantized, denoted as n = 1, 2, 3… (K, L, M, N shells).
✔ Energy is emitted/absorbed only when electron jumps between levels.
✔ Energy difference = hν, where h = Planck’s constant.
🌟 Bohr’s model explained stability of atom and hydrogen spectrum lines (Balmer, Lyman, Paschen series).
❌ Limitation: Couldn’t explain multi-electron atoms and fine details of spectra.
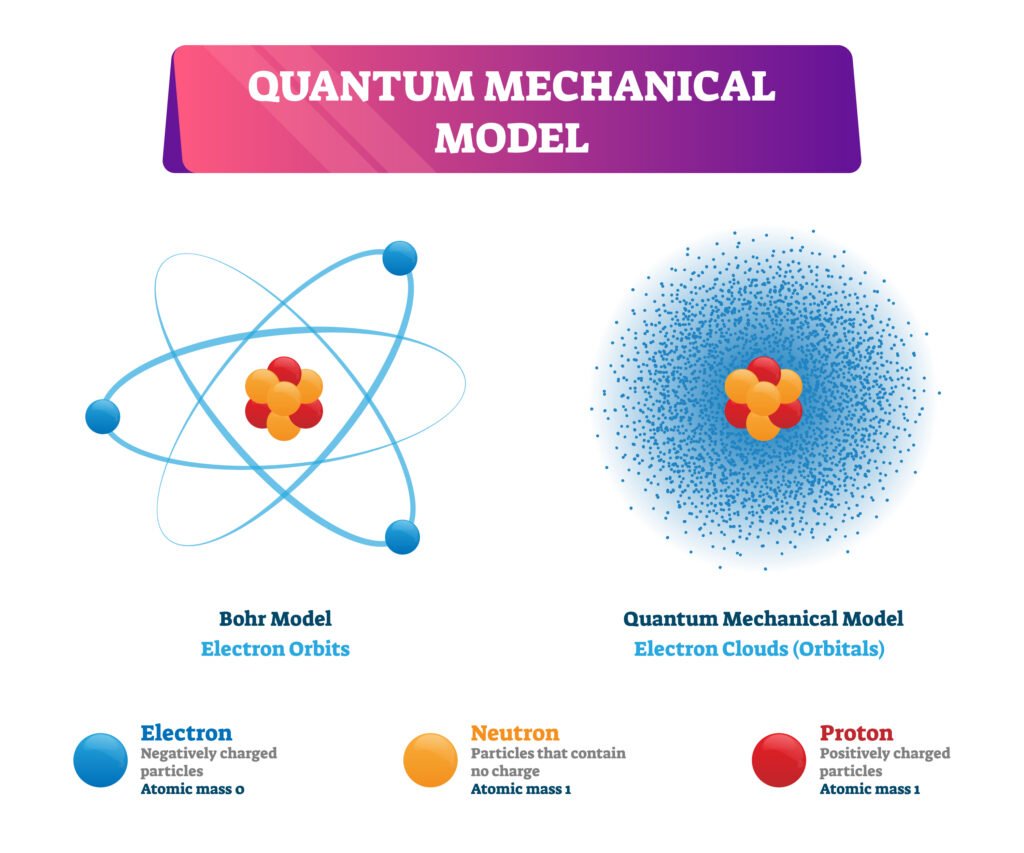
🔵 Quantum Mechanical Model of Atom
Modern understanding of atoms is based on Quantum Mechanics ⚡, developed by Schrödinger, Heisenberg, de Broglie, etc.
1️⃣ de Broglie’s Hypothesis (1924)
Electrons behave like waves 🌊 as well as particles (wave–particle duality).
Wavelength (λ) = h / mv.
2️⃣ Heisenberg’s Uncertainty Principle (1927)
Impossible to know both position and momentum of electron simultaneously with precision.
So, instead of fixed paths, electrons exist in regions called orbitals (probability zones).
3️⃣ Schrödinger Wave Equation (1926)
Gave mathematical description of electron as a wave.
Solutions gave atomic orbitals → 3D regions around nucleus with high probability of finding electrons.
🟢 Quantum Numbers
Each electron in atom is described by four quantum numbers:
Principal Quantum Number (n)
Denotes main energy level or shell (K=1, L=2, M=3…).
Values: 1, 2, 3…
Larger n → farther electron from nucleus, higher energy.
Azimuthal Quantum Number (l)
Denotes subshell (s, p, d, f).
Values: 0 to (n-1).
Shape of orbital:
l=0 → s (spherical ⚪)
l=1 → p (dumbbell 🎭)
l=2 → d (cloverleaf 🍀)
l=3 → f (complex shapes).
Magnetic Quantum Number (mₗ)
Orientation of orbital in space.
Values: −l to +l.
For p (l=1) → mₗ = −1, 0, +1 (3 orientations: px, py, pz).
Spin Quantum Number (ms)
Spin of electron: +½ or −½.
Two electrons in same orbital must have opposite spins (Pauli principle).
🔵 Electronic Configuration
Distribution of electrons into orbitals of atom is called electronic configuration.
Rules:
✔ Aufbau Principle → fill lower energy orbitals first (1s < 2s < 2p < 3s < 3p < 4s < 3d …).
✔ Pauli’s Exclusion Principle → no two electrons in atom can have same four quantum numbers.
✔ Hund’s Rule of Maximum Multiplicity → in degenerate orbitals (same energy), electrons occupy singly before pairing, with parallel spins.
Examples:
Hydrogen: 1s¹
Helium: 1s²
Oxygen: 1s² 2s² 2p⁴
Sodium: 1s² 2s² 2p⁶ 3s¹
🟢 Atomic Spectra
Atoms absorb or emit radiation when electrons jump between energy levels.
Hydrogen Spectrum: Lyman (UV), Balmer (Visible), Paschen (IR), Brackett, Pfund series.
Explanation by Bohr model was a milestone.
Spectrum lines show quantized nature of energy.
🔵 Important Concepts
Node: point/region where probability of finding electron is zero.
Isotopes: same Z, different mass (H-1, H-2, H-3).
Isobars: same mass, different Z (40Ar, 40Ca).
Isotones: same neutrons, different Z (14C, 15N).
Orbit vs Orbital: Orbit = fixed path (Bohr), Orbital = region of probability (Quantum theory).
Energy Order: s < p < d < f.
Maximum electrons formula: 2n² per shell.
🟢 Applications of Atomic Structure Concepts
✔ Explains stability of atoms and bonding.
✔ Foundation of periodic table arrangement.
✔ Basis of chemical reactivity, ionization energy, etc.
✔ Helps in spectroscopy, quantum chemistry, nuclear science.
✔ Important in lasers, semiconductors, medical imaging, etc.
✨ Conclusion
The journey from indivisible atom of Democritus to modern quantum mechanical atom shows how experiments and theories gradually built our present understanding. The atom is not a tiny solid sphere but a dynamic system of nucleus + electrons described by wave functions.
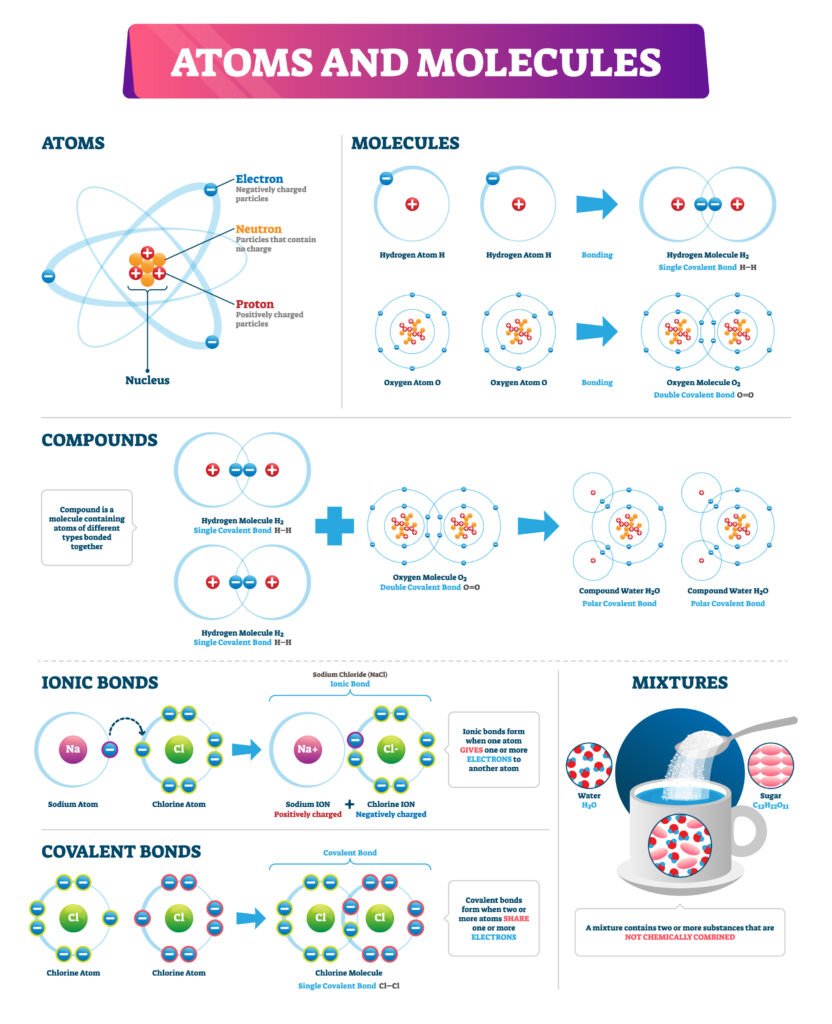
🔹 II. Lesson Summary
📌 Structure of Atom – Key Points
🔵 Atoms are fundamental particles of matter, composed of electrons, protons, and neutrons.
🟢 Discovery:
Electrons by Thomson (cathode rays).
Protons by Goldstein (canal rays).
Neutrons by Chadwick.
🔴 Atomic Models:
Thomson: positive sphere with electrons.
Rutherford: nucleus with electrons revolving.
Bohr: quantized orbits, stability explained.
Modern Quantum Model: orbitals, probability distribution.
🟡 Quantum Concepts:
de Broglie: wave-particle duality.
Heisenberg: uncertainty principle.
Schrödinger: wave equation → orbitals.
💡 Quantum Numbers:
n (shell), l (subshell), mₗ (orientation), ms (spin).
Orbitals: s (sphere), p (dumbbell), d (clover), f (complex).
📚 Electronic Configuration:
Rules: Aufbau, Pauli, Hund.
Examples: H → 1s¹, Na → 1s²2s²2p⁶3s¹.
🌟 Spectra:
Hydrogen spectrum → Lyman, Balmer, Paschen, Brackett, Pfund.
Evidence of quantized energy levels.
📍 Special Terms:
Isotopes (same Z, diff A), Isobars (same A, diff Z), Isotones (same neutrons).
Nodes = zero probability regions.
Max electrons in shell = 2n².
✨ Applications:
Explains periodic table, bonding, reactivity.
Useful in spectroscopy, electronics, quantum mechanics, medicine.
🔹 III. Quick Recap
✔ Atom = nucleus (protons + neutrons) + electrons revolving.
✔ Discovery of e⁻, p⁺, n⁰ shaped early models (Thomson, Rutherford).
✔ Bohr introduced quantized orbits, hydrogen spectra explanation.
✔ Quantum mechanics gave modern view: orbitals, wave-particle duality, uncertainty principle.
✔ Quantum numbers (n, l, mₗ, ms) define each electron.
✔ Electronic configuration rules: Aufbau, Pauli, Hund.
✔ Isotopes, isobars, isotones → classification of atoms.
✔ Concept forms basis of chemical bonding, periodic table, reactivity, and modern applications.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
🔵 Question 2.1:
(i) Calculate the number of electrons which will together weigh one gram.
(ii) Calculate the mass and charge of one mole of electrons.
🟢 Answer:
✳ (i) To find number of electrons having total mass = 1 g
Mass of one electron = 9.109 × 10⁻³¹ kg
Convert into grams → 9.109 × 10⁻³¹ × 1000 = 9.109 × 10⁻²⁸ g
Number of electrons = Total mass / mass of one electron
➡ N = 1 / 9.109 × 10⁻²⁸ = 1.10 × 10²⁷ electrons ✔
✳ (ii) For one mole of electrons
1 mole = 6.022 × 10²³ electrons
Mass = 9.109 × 10⁻³¹ × 6.022 × 10²³ = 5.48 × 10⁻⁷ kg = 0.548 mg
Charge = 6.022 × 10²³ × 1.602 × 10⁻¹⁹ = 9.65 × 10⁴ C ✔
🔵 Question 2.2:
(i) Calculate the total number of electrons present in one mole of methane.
(ii) Find (a) the total number and (b) the total mass of neutrons in 7 g of ¹⁴C.
(iii) Find (a) the total number and (b) the total mass of protons in 34 mg of NH₃ at STP.
🟢 Answer:
✳ (i) Electrons in 1 mole CH₄
C atom = 6 electrons, 4 H atoms = 4 electrons
➡ Total = 10 electrons per molecule
1 mole = 6.022 × 10²³ molecules
➡ Electrons = 10 × 6.022 × 10²³ = 6.022 × 10²⁴ electrons ✔
✳ (ii) Neutrons in 7 g ¹⁴C
Neutrons per atom = 14 − 6 = 8
Moles in 7 g = 7 / 14 = 0.5 mol
Atoms = 0.5 × 6.022 × 10²³ = 3.011 × 10²³
Total neutrons = 8 × 3.011 × 10²³ = 2.41 × 10²⁴
Mass = 2.41 × 10²⁴ × 1.675 × 10⁻²⁷ = 4.04 × 10⁻³ kg = 4.04 g ✔
✳ (iii) Protons in 34 mg NH₃
Protons per molecule = 7 (N) + 3 (H) = 10
Mass = 34 mg = 0.034 g; molar mass = 17 g/mol
Moles = 0.034 / 17 = 0.002 mol
Molecules = 0.002 × 6.022 × 10²³ = 1.20 × 10²¹
Protons = 10 × 1.20 × 10²¹ = 1.20 × 10²²
Mass = 1.20 × 10²² × 1.673 × 10⁻²⁷ = 2.0 × 10⁻⁵ g ✔
🔵 Question 2.3:
How many neutrons and protons are there in the following nuclei:
¹³₆C, ¹⁶₈O, ²⁴₁₂Mg, ⁵⁶₂₆Fe, ⁸⁸₃₈Sr
🟢 Answer:
Use formula → n = A − Z, p = Z
Nucleus Protons (Z) Neutrons (A−Z)
¹³₆C 6 7
¹⁶₈O 8 8
²⁴₁₂Mg 12 12
⁵⁶₂₆Fe 26 30
⁸⁸₃₈Sr 38 50
🔵 Question 2.4:
Write the complete symbol for the atom with given atomic number and mass number.
(i) Z = 17, A = 35 (ii) Z = 92, A = 233 (iii) Z = 4, A = 9
🟢 Answer:
(i) ³⁵₁₇Cl (ii) ²³³₉₂U (iii) ⁹₄Be ✔
🔵 Question 2.5:
Yellow light from sodium lamp: λ = 580 nm. Find frequency and wavenumber.
🟢 Answer:
λ = 580 nm = 5.80 × 10⁻⁷ m
Frequency ν = c / λ = 3.0 × 10⁸ / 5.80 × 10⁻⁷ = 5.17 × 10¹⁴ s⁻¹
Wavenumber ṽ = 1 / λ = 1 / 5.80 × 10⁻⁵ cm = 1.72 × 10⁴ cm⁻¹ ✔
🔵 Question 2.6:
Find energy of photon which
(i) has frequency 3 × 10¹⁵ Hz
(ii) has wavelength 0.50 Å
🟢 Answer:
✳ (i) E = hν = 6.626 × 10⁻³⁴ × 3 × 10¹⁵ = 1.99 × 10⁻¹⁸ J
✳ (ii) λ = 0.50 Å = 5 × 10⁻¹¹ m
E = hc / λ = 6.626 × 10⁻³⁴ × 3 × 10⁸ / 5 × 10⁻¹¹ = 3.97 × 10⁻¹⁵ J ✔
🔵 Question 2.7:
Light wave with period T = 2.0 × 10⁻¹⁰ s. Find ν, λ, ṽ.
🟢 Answer:
ν = 1 / T = 1 / 2.0 × 10⁻¹⁰ = 5.0 × 10⁹ s⁻¹
λ = c / ν = 3.0 × 10⁸ / 5.0 × 10⁹ = 0.06 m
ṽ = 1 / λ = 1 / 0.06 = 16.7 m⁻¹ ✔
🔵 Question 2.8:
Number of photons with λ = 4000 pm that give total energy = 1 J.
🟢 Answer:
λ = 4000 pm = 4 × 10⁻⁹ m
E (one photon) = hc / λ = 6.626 × 10⁻³⁴ × 3 × 10⁸ / 4 × 10⁻⁹ = 4.97 × 10⁻¹⁷ J
Number = Total energy / energy per photon
➡ N = 1 / 4.97 × 10⁻¹⁷ = 2.01 × 10¹⁶ photons ✔
🔵 Question 2.9:
Photon λ = 4 × 10⁻⁷ m hits metal (work function = 2.13 eV). Find:
(i) energy of photon (ii) KE of e⁻ (iii) velocity of e⁻
🟢 Answer:
E = hc / λ = 6.626 × 10⁻³⁴ × 3 × 10⁸ / 4 × 10⁻⁷ = 4.97 × 10⁻¹⁹ J = 3.10 eV
KE = 3.10 − 2.13 = 0.97 eV = 1.55 × 10⁻¹⁹ J
v = √(2KE / m) = √(2 × 1.55 × 10⁻¹⁹ / 9.11 × 10⁻³¹) = 5.85 × 10⁵ m/s ✔
🔵 Question 2.10:
Radiation λ = 242 nm ionises sodium. Find ionisation energy (kJ mol⁻¹).
🟢 Answer:
λ = 242 nm = 2.42 × 10⁻⁷ m
E = hc / λ = 6.626 × 10⁻³⁴ × 3 × 10⁸ / 2.42 × 10⁻⁷ = 8.21 × 10⁻¹⁹ J
Per mole = 8.21 × 10⁻¹⁹ × 6.022 × 10²³ / 1000 = 494 kJ mol⁻¹ ✔
🔵 Question 2.11:
A 25 W bulb emits light λ = 0.57 μm. Find number of photons per second.
🟢 Answer:
λ = 0.57 μm = 5.7 × 10⁻⁷ m
E = hc / λ = 6.626 × 10⁻³⁴ × 3 × 10⁸ / 5.7 × 10⁻⁷ = 3.49 × 10⁻¹⁹ J
Power = 25 J/s
N = Power / E = 25 / 3.49 × 10⁻¹⁹ = 7.16 × 10¹⁹ photons/s ✔
🔵 Question 2.12:
Two spectral lines have frequencies 2.47 × 10¹⁵ Hz and 5.46 × 10¹⁴ Hz. Find energy difference.
🟢 Answer:
Δν = 2.47 × 10¹⁵ − 5.46 × 10¹⁴ = 1.924 × 10¹⁵ Hz
ΔE = h × Δν = 6.626 × 10⁻³⁴ × 1.924 × 10¹⁵ = 1.28 × 10⁻¹⁸ J ✔
🔵 Question 2.13:
Calculate the frequency and wavelength of radiation emitted when an electron in a hydrogen atom jumps from n = 3 to n = 2 level.
🟢 Answer:
✳ Step 1: Use formula for energy difference
E = 2.18 × 10⁻¹⁸ (1/n₁² − 1/n₂²)
= 2.18 × 10⁻¹⁸ (1/2² − 1/3²)
= 2.18 × 10⁻¹⁸ (1/4 − 1/9)
= 2.18 × 10⁻¹⁸ × (5/36)
➡ E = 3.03 × 10⁻¹⁹ J
✳ Step 2: Frequency
ν = E / h = 3.03 × 10⁻¹⁹ / 6.626 × 10⁻³⁴ = 4.57 × 10¹⁴ Hz
✳ Step 3: Wavelength
λ = c / ν = 3 × 10⁸ / 4.57 × 10¹⁴ = 6.56 × 10⁻⁷ m = 656 nm ✔
🔵 Question 2.14:
What is the energy (in joules) of a photon of wavelength 4 × 10⁻⁷ m?
🟢 Answer:
✳ E = hc / λ
= 6.626 × 10⁻³⁴ × 3 × 10⁸ / 4 × 10⁻⁷
➡ E = 4.97 × 10⁻¹⁹ J ✔
🔵 Question 2.15:
Calculate the energy in eV of a photon of wavelength 310 nm.
🟢 Answer:
✳ Step 1: Convert λ = 310 nm = 3.10 × 10⁻⁷ m
✳ Step 2: E = hc / λ
= 6.626 × 10⁻³⁴ × 3 × 10⁸ / 3.10 × 10⁻⁷ = 6.41 × 10⁻¹⁹ J
✳ Step 3: Convert to eV
1 eV = 1.602 × 10⁻¹⁹ J
E = 6.41 × 10⁻¹⁹ / 1.602 × 10⁻¹⁹ = 4.00 eV ✔
🔵 Question 2.16:
Calculate the wavelength associated with an electron moving with velocity 2.05 × 10⁷ m/s. (mass of electron = 9.11 × 10⁻³¹ kg)
🟢 Answer:
✳ λ = h / (mv)
= 6.626 × 10⁻³⁴ / (9.11 × 10⁻³¹ × 2.05 × 10⁷)
➡ λ = 3.54 × 10⁻¹¹ m ✔
🔵 Question 2.17:
Find the kinetic energy of an electron moving with de Broglie wavelength of 1 Å.
🟢 Answer:
✳ λ = 1 Å = 1 × 10⁻¹⁰ m
✳ Formula: E = h² / (2mλ²)
= (6.626 × 10⁻³⁴)² / [2 × 9.11 × 10⁻³¹ × (1 × 10⁻¹⁰)²]
= 4.39 × 10⁻⁶⁷ / 1.82 × 10⁻⁵⁰ = 2.41 × 10⁻¹⁷ J
Convert to eV: 2.41 × 10⁻¹⁷ / 1.602 × 10⁻¹⁹ = 15.0 eV ✔
🔵 Question 2.18:
Calculate the wavelength associated with an electron accelerated through a potential difference of 100 V.
🟢 Answer:
✳ Energy E = eV = 1.602 × 10⁻¹⁹ × 100 = 1.602 × 10⁻¹⁷ J
✳ λ = h / √(2mE)
= 6.626 × 10⁻³⁴ / √(2 × 9.11 × 10⁻³¹ × 1.602 × 10⁻¹⁷)
= 6.626 × 10⁻³⁴ / 5.38 × 10⁻²⁴ = 1.23 × 10⁻¹⁰ m = 1.23 Å ✔
🔵 Question 2.19:
Calculate the wavenumber for the electron transition from n = 3 to n = 2 in hydrogen atom.
🟢 Answer:
✳ ṽ = R (1/n₁² − 1/n₂²)
= 1.097 × 10⁷ (1/2² − 1/3²)
= 1.097 × 10⁷ (1/4 − 1/9) = 1.097 × 10⁷ × 5/36
➡ ṽ = 1.52 × 10⁶ m⁻¹ ✔
🔵 Question 2.20:
Calculate the energy difference between the n = 2 and n = 3 states of hydrogen atom.
🟢 Answer:
✳ Eₙ = −2.18 × 10⁻¹⁸ / n²
E₂ = −2.18 × 10⁻¹⁸ / 4 = −5.45 × 10⁻¹⁹ J
E₃ = −2.18 × 10⁻¹⁸ / 9 = −2.42 × 10⁻¹⁹ J
✳ ΔE = E₃ − E₂ = (−2.42 × 10⁻¹⁹) − (−5.45 × 10⁻¹⁹) = 3.03 × 10⁻¹⁹ J ✔
🔵 Question 2.21:
Calculate the energy required to remove an electron from n = 2 orbit of He⁺ ion.
🟢 Answer:
✳ Formula: E = 2.18 × 10⁻¹⁸ × Z² / n²
Here, Z = 2, n = 2
E = 2.18 × 10⁻¹⁸ × 4 / 4 = 2.18 × 10⁻¹⁸ J per atom
✳ Per mole = 2.18 × 10⁻¹⁸ × 6.022 × 10²³ / 1000 = 1313 kJ mol⁻¹ ✔
🔵 Question 2.22:
Calculate the wavelength of an electron in the third orbit of hydrogen atom.
🟢 Answer:
✳ Step 1: Radius of third orbit
rₙ = a₀ × n² = 0.529 × 3² = 4.76 Å
✳ Step 2: Velocity
vₙ = 2.19 × 10⁶ / n = 2.19 × 10⁶ / 3 = 7.3 × 10⁵ m/s
✳ Step 3: λ = h / mv
= 6.626 × 10⁻³⁴ / (9.11 × 10⁻³¹ × 7.3 × 10⁵) = 9.96 × 10⁻¹⁰ m = 0.996 nm ✔
🔵 Question 2.23:
The energy associated with the first orbit in hydrogen atom is −2.18 × 10⁻¹⁸ J/atom.
What is the energy associated with the second orbit?
What is the difference in energies between the two orbits?
🟢 Answer:
✳ Formula:
Eₙ = −2.18 × 10⁻¹⁸ / n²
➡ For n = 1: E₁ = −2.18 × 10⁻¹⁸ J (given)
➡ For n = 2: E₂ = −2.18 × 10⁻¹⁸ / 4 = −5.45 × 10⁻¹⁹ J
✳ Energy difference:
ΔE = E₂ − E₁ = (−5.45 × 10⁻¹⁹) − (−2.18 × 10⁻¹⁸)
➡ ΔE = 1.64 × 10⁻¹⁸ J ✔
🔵 Question 2.24:
Calculate the frequency and wavelength of light emitted when electron in hydrogen atom undergoes transition from energy level n = 4 to n = 1.
🟢 Answer:
✳ Energy difference:
E = 2.18 × 10⁻¹⁸ (1/1² − 1/4²)
= 2.18 × 10⁻¹⁸ (1 − 1/16)
= 2.18 × 10⁻¹⁸ × 15/16 = 2.04 × 10⁻¹⁸ J
✳ Frequency:
ν = E / h = 2.04 × 10⁻¹⁸ / 6.626 × 10⁻³⁴ = 3.08 × 10¹⁵ Hz
✳ Wavelength:
λ = c / ν = 3 × 10⁸ / 3.08 × 10¹⁵ = 9.74 × 10⁻⁸ m = 97.4 nm ✔
🔵 Question 2.25:
Find the frequency and wavelength of light emitted during transition of electron from n = 5 to n = 2 state in the hydrogen atom.
🟢 Answer:
✳ Energy difference:
E = 2.18 × 10⁻¹⁸ (1/2² − 1/5²)
= 2.18 × 10⁻¹⁸ (1/4 − 1/25)
= 2.18 × 10⁻¹⁸ × (21/100)
= 4.58 × 10⁻¹⁹ J
✳ Frequency:
ν = E / h = 4.58 × 10⁻¹⁹ / 6.626 × 10⁻³⁴ = 6.91 × 10¹⁴ Hz
✳ Wavelength:
λ = c / ν = 3 × 10⁸ / 6.91 × 10¹⁴ = 4.34 × 10⁻⁷ m = 434 nm ✔
🔵 Question 2.26:
Calculate the wavenumber for the longest wavelength transition in the Balmer series of atomic hydrogen.
🟢 Answer:
✳ Longest wavelength = smallest energy difference = n₂ = 3 to n₁ = 2
ṽ = R (1/2² − 1/3²) = 1.097 × 10⁷ × (1/4 − 1/9)
= 1.097 × 10⁷ × 5/36 = 1.52 × 10⁶ m⁻¹ ✔
🔵 Question 2.27:
Calculate the wavelength for the shortest wavelength transition in Balmer series of hydrogen spectrum.
🟢 Answer:
✳ Shortest wavelength = largest energy difference = n₂ = ∞ to n₁ = 2
ṽ = R (1/2² − 1/∞²) = R × 1/4 = 1.097 × 10⁷ × 0.25 = 2.74 × 10⁶ m⁻¹
λ = 1 / ṽ = 1 / 2.74 × 10⁶ = 3.65 × 10⁻⁷ m = 365 nm ✔
🔵 Question 2.28:
Calculate the ionization energy of hydrogen atom in kJ mol⁻¹.
🟢 Answer:
✳ Energy required to remove electron from n = 1:
E = 2.18 × 10⁻¹⁸ J per atom
Per mole: 2.18 × 10⁻¹⁸ × 6.022 × 10²³ / 1000 = 1312 kJ mol⁻¹ ✔
🔵 Question 2.29:
Calculate the radius of the third orbit of hydrogen atom.
🟢 Answer:
✳ Formula: rₙ = a₀ × n²
a₀ = 0.529 Å, n = 3
r₃ = 0.529 × 9 = 4.76 Å ✔
🔵 Question 2.30:
Calculate the speed of electron in the second orbit of hydrogen atom.
🟢 Answer:
✳ Formula: vₙ = 2.19 × 10⁶ / n
For n = 2 → v₂ = 2.19 × 10⁶ / 2 = 1.095 × 10⁶ m/s ✔
🔵 Question 2.31:
Calculate the total energy of an electron in the first Bohr orbit of hydrogen atom.
🟢 Answer:
✳ Formula: E₁ = −2.18 × 10⁻¹⁸ / 1² = −2.18 × 10⁻¹⁸ J per atom
Per mole: −2.18 × 10⁻¹⁸ × 6.022 × 10²³ / 1000 = −1312 kJ mol⁻¹ ✔
🔵 Question 2.32:
Calculate the total energy for the electron in the third orbit of hydrogen atom.
🟢 Answer:
E₃ = −2.18 × 10⁻¹⁸ / 3² = −2.18 × 10⁻¹⁸ / 9 = −2.42 × 10⁻¹⁹ J per atom
Per mole = −2.42 × 10⁻¹⁹ × 6.022 × 10²³ / 1000 = −145.7 kJ mol⁻¹ ✔
🔵 Question 2.33:
What transition in the hydrogen spectrum would have the same wavelength as the Balmer transition n = 4 to n = 2 of He⁺ spectrum?
🟢 Answer
For He⁺:
ṽ = RZ²(1/2² − 1/4²) = 4R(3/16) = (3/4)R
For H, we need (1/n₁² − 1/n₂²) = 3/4
This is satisfied by n₁ = 1, n₂ = 2
✔ Equivalent transition = 2 → 1 (Lyman series)
🔵 Question 2.34:
Calculate the velocity and wavelength of an electron ejected from the surface of a metal by radiation of wavelength 200 nm. The work function of the metal is 4.2 eV.
🟢 Answer:
✳ Step 1: Find energy of incident photon
E = hc / λ
= 6.626 × 10⁻³⁴ × 3 × 10⁸ / 2 × 10⁻⁷ = 9.94 × 10⁻¹⁹ J
Convert to eV → 9.94 × 10⁻¹⁹ / 1.602 × 10⁻¹⁹ = 6.2 eV
✳ Step 2: Kinetic energy of emitted electron
KE = E − work function = 6.2 − 4.2 = 2.0 eV
In joules → 2.0 × 1.602 × 10⁻¹⁹ = 3.20 × 10⁻¹⁹ J
✳ Step 3: Velocity
v = √(2KE / m) = √(2 × 3.20 × 10⁻¹⁹ / 9.11 × 10⁻³¹) = 8.39 × 10⁵ m/s
✳ Step 4: de Broglie wavelength
λ = h / (mv) = 6.626 × 10⁻³⁴ / (9.11 × 10⁻³¹ × 8.39 × 10⁵) = 8.67 × 10⁻¹⁰ m = 0.867 nm ✔
🔵 Question 2.35:
Calculate the uncertainty in the position of an electron if the uncertainty in its velocity is 5.0 × 10³ m/s.
(h = 6.626 × 10⁻³⁴ J s, m = 9.11 × 10⁻³¹ kg)
🟢 Answer:
✳ Heisenberg uncertainty principle:
Δx × Δv ≥ h / (4πm)
Δx ≥ 6.626 × 10⁻³⁴ / (4 × 3.14 × 9.11 × 10⁻³¹ × 5.0 × 10³)
Denominator = 5.72 × 10⁻²⁶
Δx ≥ 1.16 × 10⁻⁸ m ✔
🔵 Question 2.36:
What is the maximum number of electrons that can be associated with the following sets of quantum numbers?
(a) n = 3, l = 2 (b) n = 4, l = 0
🟢 Answer:
✳ (a) For n = 3, l = 2 → d-subshell
Number of orbitals = 2l + 1 = 5
Each orbital holds 2 electrons
➡ Maximum electrons = 5 × 2 = 10
✳ (b) For n = 4, l = 0 → s-subshell
Number of orbitals = 1 → 2 electrons
➡ 2 electrons ✔
🔵 Question 2.37:
What is the total number of orbitals associated with the principal quantum number n = 3?
🟢 Answer:
Total orbitals = n² = 3² = 9 orbitals ✔
🔵 Question 2.38:
What is the total number of orbitals in the second energy level of an atom?
🟢 Answer:
n = 2 → total orbitals = n² = 2² = 4 orbitals ✔
🔵 Question 2.39:
How many electrons in an atom may have the following quantum numbers:
n = 4, l = 1, mₗ = 0?
🟢 Answer:
For n = 4, l = 1 (4p subshell)
mₗ = 0 represents one orbital
Each orbital can hold 2 electrons
➡ 2 electrons ✔
🔵 Question 2.40:
Write the set of four quantum numbers for the valence electron of sodium atom (Z = 11).
🟢 Answer:
Sodium configuration: 1s² 2s² 2p⁶ 3s¹
Valence electron in 3s¹:
n = 3, l = 0, mₗ = 0, mₛ = +½ ✔
🔵 Question 2.41:
Which of the following sets of quantum numbers are not possible? Why?
(a) n = 1, l = 0, mₗ = 0, mₛ = +½
(b) n = 1, l = 1, mₗ = 0, mₛ = +½
(c) n = 2, l = 0, mₗ = 0, mₛ = +½
🟢 Answer:
(a) ✅ Possible
(b) ❌ Not possible because for n = 1, l can be 0 only
(c) ✅ Possible ✔
🔵 Question 2.42:
What are the possible values of n and l for an electron in 3d orbital?
🟢 Answer:
For 3d orbital:
n = 3, l = 2 ✔
🔵 Question 2.43:
What is the maximum number of electrons in a subshell with l = 3?
🟢 Answer:
l = 3 → f-subshell
Number of orbitals = 2l + 1 = 7
Electrons = 7 × 2 = 14 electrons ✔
🔵 Question 2.44:
How many subshells are associated with n = 4?
🟢 Answer:
l = 0, 1, 2, 3 → 4 subshells (4s, 4p, 4d, 4f) ✔
🔵 Question 2.45:
Arrange the following radiations in increasing order of frequency:
(a) radiation from microwave oven
(b) amber light from traffic signal
(c) radiation from FM radio
(d) cosmic rays from outer space
(e) X-rays
🟢 Answer
Increasing frequency order:
(c) FM radio < (a) microwave oven < (b) amber light < (e) X-rays < (d) cosmic rays ✔
🔵 Question 2.46:
What will be the energy of the electron in the n = 4 orbit of hydrogen atom?
🟢 Answer:
✳ Formula: Eₙ = −2.18 × 10⁻¹⁸ / n²
➡ E₄ = −2.18 × 10⁻¹⁸ / 16 = −1.36 × 10⁻¹⁹ J per atom
Per mole: (−1.36 × 10⁻¹⁹) × 6.022 × 10²³ / 1000 = −82.0 kJ mol⁻¹ ✔
🔵 Question 2.47:
Calculate the wavelength and frequency of the spectral line emitted when electron jumps from n = 5 to n = 4 level in hydrogen atom.
🟢 Answer:
✳ Energy difference:
E = 2.18 × 10⁻¹⁸ (1/4² − 1/5²)
= 2.18 × 10⁻¹⁸ (1/16 − 1/25)
= 2.18 × 10⁻¹⁸ × (9/400)
➡ E = 4.90 × 10⁻²⁰ J
✳ Frequency:
ν = E / h = 4.90 × 10⁻²⁰ / 6.626 × 10⁻³⁴ = 7.39 × 10¹³ Hz
✳ Wavelength:
λ = c / ν = 3 × 10⁸ / 7.39 × 10¹³ = 4.06 × 10⁻⁶ m = 4060 nm ✔
🔵 Question 2.48:
Calculate the energy of a photon required to remove an electron from the n = 2 orbit of hydrogen atom.
🟢 Answer:
✳ Ionisation from n = 2 → ∞
E = 2.18 × 10⁻¹⁸ × (1/2²) = 2.18 × 10⁻¹⁸ × 0.25 = 5.45 × 10⁻¹⁹ J
Per mole: 5.45 × 10⁻¹⁹ × 6.022 × 10²³ / 1000 = 328.6 kJ mol⁻¹ ✔
🔵 Question 2.49:
Calculate the energy of the first line in the Lyman series of hydrogen spectrum.
🟢 Answer:
First line → n₂ = 2 → n₁ = 1
✳ E = 2.18 × 10⁻¹⁸ (1/1² − 1/2²)
= 2.18 × 10⁻¹⁸ (1 − 1/4)
= 2.18 × 10⁻¹⁸ × 3/4 = 1.64 × 10⁻¹⁸ J
Frequency = E / h = 1.64 × 10⁻¹⁸ / 6.626 × 10⁻³⁴ = 2.47 × 10¹⁵ Hz
Wavelength = 3 × 10⁸ / 2.47 × 10¹⁵ = 1.22 × 10⁻⁷ m = 122 nm ✔
🔵 Question 2.50:
What is the energy required to excite an electron in a hydrogen atom from n = 1 to n = 3 orbit?
🟢 Answer:
E = 2.18 × 10⁻¹⁸ (1/1² − 1/3²)
= 2.18 × 10⁻¹⁸ (1 − 1/9)
= 2.18 × 10⁻¹⁸ × 8/9 = 1.94 × 10⁻¹⁸ J
Per mole: 1.94 × 10⁻¹⁸ × 6.022 × 10²³ / 1000 = 1170 kJ mol⁻¹ ✔
🔵 Question 2.51:
Calculate the velocity of an electron in the first orbit of hydrogen atom.
🟢 Answer:
Formula: vₙ = 2.19 × 10⁶ / n
➡ v₁ = 2.19 × 10⁶ m/s ✔
🔵 Question 2.52:
Calculate the uncertainty in velocity of a wagon of mass 3000 kg if the uncertainty in its position is 5.5 × 10⁻⁵ m. (h = 6.626 × 10⁻³⁴ J s)
🟢 Answer (Corrected):
✳ Heisenberg: Δx × Δv ≥ h / (4πm)
Δv ≥ 6.626 × 10⁻³⁴ / (4 × 3.14 × 3000 × 5.5 × 10⁻⁵)
Denominator ≈ 2.07
➡ Δv ≥ 3.2 × 10⁻³⁴ m/s ✔ (negligible)
🔵 Question 2.53:
Calculate the number of possible orbitals for principal quantum number n = 4.
🟢 Answer:
Number of orbitals = n² = 4² = 16 orbitals ✔
🔵 Question 2.54:
What are the possible values of l and mₗ for an electron in the n = 4 energy level?
🟢 Answer:
For n = 4:
l = 0, 1, 2, 3
For each l:
l = 0 → mₗ = 0
l = 1 → mₗ = −1, 0, +1
l = 2 → mₗ = −2, −1, 0, +1, +2
l = 3 → mₗ = −3, −2, −1, 0, +1, +2, +3 ✔
🔵 Question 2.55:
How many subshells are present in the shell with n = 3?
🟢 Answer:
For n = 3 → l = 0, 1, 2 → 3 subshells (3s, 3p, 3d) ✔
🔵 Question 2.56:
What is the maximum number of electrons that can have the following quantum numbers?
(a) n = 4 (b) n = 3, l = 1 (c) n = 3, l = 0 (d) n = 4, l = 2
🟢 Answer:
✳ (a) n = 4 → Total electrons = 2n² = 2 × 4² = 32 electrons
✳ (b) n = 3, l = 1 → p-subshell has 3 orbitals × 2 = 6 electrons
✳ (c) n = 3, l = 0 → s-subshell = 1 orbital × 2 = 2 electrons
✳ (d) n = 4, l = 2 → d-subshell = 5 orbitals × 2 = 10 electrons ✔
🔵 Question 2.57:
Which of the following will not show a photoelectric effect?
(a) Blue light (b) Green light (c) Yellow light (d) Red light
🟢 Answer:
✳ Photoelectric effect requires photon energy ≥ work function.
Red light has lowest frequency, hence lowest energy.
➡ Red light (d) will not show the photoelectric effect ✔
🔵 Question 2.58:
What are the possible values of mₗ for l = 3?
🟢 Answer:
✳ For l = 3 → f-subshell
mₗ = −3, −2, −1, 0, +1, +2, +3 ✔
🔵 Question 2.59:
What is the shape of the orbital having l = 2?
🟢 Answer:
✳ l = 0 → s (spherical)
✳ l = 1 → p (dumb-bell)
✳ l = 2 → d-orbital
➡ Shape: Double dumb-bell (clover-leaf) ✔
🔵 Question 2.60:
How many orbitals are possible for n = 3?
🟢 Answer:
Number of orbitals = n² = 3² = 9 orbitals ✔
🔵 Question 2.61:
State the Heisenberg uncertainty principle.
🟢 Answer:
✳ It is impossible to determine simultaneously the exact position and exact momentum of a microscopic particle such as an electron.
Mathematically:
➡ Δx × Δp ≥ h / 4π
where Δx = uncertainty in position, Δp = uncertainty in momentum ✔
🔵 Question 2.62:
Which of the following statements are correct?
(a) All orbits in an atom are circular.
(b) Orbitals have definite shape and orientation.
(c) In a given atom, no two electrons can have the same set of four quantum numbers.
(d) The maximum number of electrons in a subshell is given by 2(2l + 1).
🟢 Answer (Corrected):
✔ Correct statements: (b), (c), (d)
❌ (a) is incorrect (modern atomic theory uses orbitals, not circular orbits)
🔵 Question 2.63:
Write the electronic configuration of Fe³⁺ ion. (Atomic number 26)
🟢 Answer:
Fe: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁶
Fe³⁺: remove 3 electrons → 2 from 4s and 1 from 3d
➡ 1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁵ ✔
🔵 Question 2.64:
How many unpaired electrons are present in Fe³⁺ ion?
🟢 Answer:
Fe³⁺ configuration: 3d⁵
Each d orbital singly filled → 5 unpaired electrons ✔
🔵 Question 2.65:
What are the possible values of n, l, and mₗ for the 19th electron of potassium (Z = 19)?
🟢 Answer:
K: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹
19th electron → 4s¹
➡ n = 4, l = 0, mₗ = 0 ✔
🔵 Question 2.66:
What is the azimuthal quantum number for an electron in 4d orbital?
🟢 Answer:
4d orbital → n = 4, l = 2 ✔
🔵 Question 2.67:
Name the principle which restricts the number of electrons in an orbital to two.
🟢 Answer:
✳ Pauli Exclusion Principle:
No two electrons in an atom can have the same set of four quantum numbers.
Hence, maximum two electrons per orbital ✔
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
✳ Section A (Q1–Q16) – MCQs (1 mark each, 16 × 1 = 16 marks)
Options:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Question 1. Who discovered the electron?
Rutherford
J.J. Thomson
Millikan
Chadwick
Answer: 2
Question 2. Which particle was discovered by Goldstein?
Electron
Neutron
Proton
Positron
Answer: 3
Question 3. Which experiment confirmed the charge of electron?
Rutherford’s scattering
Oil drop experiment
Cathode ray experiment
Canal ray experiment
Answer: 2
Question 4. The mass of neutron is approximately equal to:
Mass of proton
Mass of electron
Double of proton
One tenth of proton
Answer: 1
Question 5. Radius of first Bohr orbit of hydrogen atom is:
0.529 Å
1.058 Å
13.6 Å
5.29 Å
Answer: 1
Question 6. The energy of an electron in the second orbit of hydrogen is:
–3.4 eV
–13.6 eV
–6.8 eV
–1.51 eV
Answer: 1
Question 7. In hydrogen atom, transition from n = 3 to n = 2 gives:
Lyman series line
Balmer series line
Paschen series line
Brackett series line
Answer: 2
Question 8. The number of orbitals in third shell (n = 3) is:
3
6
9
18
Answer: 3
Question 9. For l = 2, the number of possible mₗ values is:
2
3
4
5
Answer: 4
Question 10. Which orbital has spherical shape?
s
p
d
f
Answer: 1
Question 11. The uncertainty principle was proposed by:
Bohr
Schrödinger
Heisenberg
Planck
Answer: 3
Question 12. Which principle states that no two electrons can have same set of four quantum numbers?
Hund’s rule
Pauli’s exclusion principle
Aufbau principle
Bohr’s postulate
Answer: 2
Question 13. The correct electronic configuration of oxygen atom (Z = 8) is:
1s² 2s² 2p⁶
1s² 2s² 2p⁴
1s² 2s² 2p³
1s² 2s² 2p⁵
Answer: 2
Question 14. The value of Planck’s constant is:
6.626 × 10⁻²³ J·s
6.626 × 10⁻³⁴ J·s
3 × 10⁸ m·s⁻¹
9.1 × 10⁻³¹ kg
Answer: 2
Question 15. (Assertion–Reason)
Assertion (A): The path of an electron around nucleus is well defined.
Reason (R): According to uncertainty principle, position and momentum of electron cannot be determined simultaneously.
Answer: 3
Question 16. (Assertion–Reason)
Assertion (A): Lyman series lies in the ultraviolet region.
Reason (R): The electronic transition takes place to n = 1 level.
Answer: 1
⚡ Section B (Q17–Q21) – Very Short Answer (2 marks each, 5 × 2 = 10 marks)
Q17. State two postulates of Dalton’s atomic theory.
🟦 Atoms are indivisible particles of matter which cannot be created or destroyed.
🟩 All atoms of an element are identical in mass and properties.
Q18. Write two limitations of Rutherford’s model.
🟪 Electrons revolving in nucleus should lose energy and collapse, but atoms are stable.
🟨 Model could not explain atomic spectra.
Q19. Write values of all four quantum numbers for the last electron of chlorine (Z = 17).
🟦 Principal quantum number (n) = 3
🟩 Azimuthal quantum number (l) = 1 (p orbital)
🟨 Magnetic quantum number (mₗ) = –1, 0, +1 → last electron in +1
🟪 Spin quantum number (mₛ) = –½
Q20. Calculate the energy of an electron in first orbit of hydrogen atom.
➤ Formula: Eₙ = –13.6 Z²/n² eV
➤ Substitution: n = 1, Z = 1 → E₁ = –13.6 (1²)/(1²) eV
✅ Final Answer: –13.6 eV
Q21. Mention two differences between orbit and orbital.
🟦 Orbit is a circular path (Bohr’s model), orbital is a 3D probability region (Quantum model).
🟩 Orbit has fixed radius, orbital has different shapes (spherical, dumbbell, etc.).
🧪 Section C (Q22–Q28) – Short Answer (3 marks each, 7 × 3 = 21 marks)
Q22. State three main observations of Rutherford’s α-scattering experiment.
🔷 Most α-particles passed undeviated → atom mostly empty.
🔶 Few deflected at small angles → presence of positive charge.
🧪 Very few bounced back → dense nucleus at centre.
Q23. Write three postulates of Bohr’s model of hydrogen atom.
🟦 Electrons revolve in fixed circular orbits called stationary states.
🟩 Energy of electron is quantized.
🟨 Energy is absorbed/emitted when electron jumps between orbits (ΔE = hν).
Q24. Derive expression for radius of nth orbit of hydrogen atom.
➤ Centripetal force = Electrostatic force → mv²/r = (1/4πε₀)(Ze²/r²)
➤ Angular momentum quantization: mvr = nh/2π
➤ Solving → rₙ = (ε₀h²n²)/(πme²Z)
✅ Radius of nth orbit: rₙ = 0.529 n² Å for hydrogen.
Q25. State three points of difference between Thomson’s and Rutherford’s model.
🟦 Thomson: positive sphere, electrons embedded; Rutherford: dense nucleus with electrons revolving.
🟨 Thomson: did not explain scattering; Rutherford: explained scattering results.
🟩 Thomson: no nucleus; Rutherford: discovered nucleus.
Q26. Explain de Broglie hypothesis of matter waves. Give relation with Bohr’s quantization.
🟦 de Broglie: moving particles show wave nature, λ = h/mv.
🟩 Standing waves fit on orbits → 2πr = nλ.
🧮 Hence mvr = nh/2π, same as Bohr’s condition.
Q27. Write differences between Lyman and Balmer series of hydrogen spectrum.
🟦 Lyman series → transitions to n₁ = 1, lies in ultraviolet region.
🟨 Balmer series → transitions to n₁ = 2, lies in visible region.
🧪 Both explained by Bohr’s model.
Q28. State Hund’s Rule. Illustrate with nitrogen atom (Z = 7).
🟦 Hund’s rule: electrons occupy degenerate orbitals singly with parallel spins before pairing.
🟨 Nitrogen (1s² 2s² 2p³): three electrons fill 2p orbitals separately.
🧪 Orbital diagram: ↑ ↑ ↑ (2p orbitals).
🧭 Section D (Q29–Q30) – Case-Based Questions (4 marks each, 2 × 4 = 8 marks)
Q29. Read the following passage and answer the questions:
Rutherford’s gold foil experiment led to the discovery of nucleus. When α-particles were bombarded on thin gold foil, most passed straight, some deflected, and very few bounced back.
(a) What conclusion was drawn from most α-particles passing undeviated? (1 mark)
(b) Why did few α-particles deflect at small angles? (1 mark)
(c) What structure of atom was proposed from this experiment? (2 marks)
🧪 Answer:
(a) Atom is mostly empty space.
(b) Small deflections occur due to repulsion from positive charge in atom.
(c) Rutherford proposed that atom has a small, dense, positively charged nucleus at the center with electrons revolving around it.
Q30. Read the following passage and answer the questions:
According to Bohr’s model, electrons revolve in fixed circular orbits. Energy is quantized and photons are emitted when electrons jump from higher to lower energy levels.
(a) State Bohr’s quantization condition. (1 mark)
(b) Write expression for energy of nth orbit. (1 mark)
(c) Explain how Bohr’s model accounts for line spectrum of hydrogen. (2 marks)
🧪 Answer:
(a) mvr = nh/2π.
(b) Eₙ = –13.6 Z²/n² eV.
(c) When electron jumps between energy levels, energy difference appears as photons of definite frequency. This gives line spectrum of hydrogen.
⚡ Section E (Q31–Q33) – Long Answer (5 marks each, 3 × 5 = 15 marks)
Q31. (a) Derive the expression for energy of electron in nth orbit of hydrogen atom.
OR
(b) Calculate the wavelength of light emitted when electron in hydrogen atom falls from n = 3 to n = 2. (R = 1.097 × 10⁷ m⁻¹).
🧪 Answer (a):
➤ Force balance: mv²/r = (1/4πε₀)(Ze²/r²).
➤ Quantization: mvr = nh/2π.
➤ Radius: rₙ = (ε₀h²n²)/(πme²Z).
➤ Energy: Eₙ = –(2π²me⁴Z²)/(h²(4πε₀)²n²).
✅ For hydrogen: Eₙ = –13.6/n² eV.
Answer (b):
➤ Formula: 1/λ = R(1/n₁² – 1/n₂²).
➤ Substitution: n₁ = 2, n₂ = 3 → 1/λ = 1.097 × 10⁷ (1/2² – 1/3²).
➤ Calculation: 1/λ = 1.097 × 10⁷ (1/4 – 1/9) = 1.097 × 10⁷ (5/36).
➤ 1/λ = 1.52 × 10⁶ m⁻¹ → λ = 6.56 × 10⁻⁷ m.
✅ Final Answer: λ = 656 nm (Balmer series, red line).
Q32. (a) State and explain Heisenberg’s Uncertainty Principle. Show its significance for motion of electron.
OR
(b) Explain quantum numbers with their significance. Write values of quantum numbers for 2p⁴ electron.
🧪 Answer (a):
🟦 Principle: Δx·Δp ≥ h/4π.
🟨 It is impossible to determine simultaneously the exact position and momentum of electron.
🟩 Significance: Electrons cannot follow fixed paths; concept of orbit replaced by orbital.
Answer (b):
🟦 Principal (n): energy level (n = 2).
🟩 Azimuthal (l): shape of orbital (l = 1 → p).
🟨 Magnetic (mₗ): orientation (–1, 0, +1 → electron may occupy any).
🟪 Spin (mₛ): spin +½ or –½.
✅ For 2p⁴ electron of oxygen: (n = 2, l = 1, mₗ = –1 or 0 or +1, mₛ = +½ or –½).
Q33. (a) Discuss Aufbau principle, Hund’s rule, and Pauli’s exclusion principle. Apply these to write configuration of Fe (Z = 26).
OR
(b) Differentiate between orbit and orbital. Draw shapes of s, p orbitals and explain their significance.
🧪 Answer (a):
🔷 Aufbau Principle – Orbitals filled in order of increasing energy (n + l rule).
🔶 Pauli’s Principle – No two electrons can have same 4 quantum numbers; max 2 electrons per orbital with opposite spins.
🧪 Hund’s Rule – Degenerate orbitals filled singly with parallel spins before pairing.
🎯 Fe (Z = 26): 1s² 2s² 2p⁶ 3s² 3p⁶ 3d⁶ 4s².
Answer (b):
🟦 Orbit – circular path in Bohr’s model.
🟨 Orbital – 3D region of probability (Quantum model).
🧪 s orbital – spherical; p orbitals – dumbbell-shaped (3 orientations).
🎯 Significance: Defines electron distribution and chemical properties.
————————————————————————————————————————————————————————————————————————————
