Class 10 : Science (In English) – Lesson 3. Metals and Non-metals
EXPLANATION & SUMMARY
🔵 Introduction
➡️ Our Earth’s crust contains many elements called metals and non-metals.
➡️ These elements occur in nature in combined or uncombined states and are essential to human life.
💡 Metals — Elements that are usually hard, lustrous, malleable, ductile, and good conductors of heat and electricity.
💡 Non-metals — Elements that are generally soft (if solid), dull, brittle, and poor conductors of heat and electricity.
🔵 Physical Properties of Metals and Non-metals
🔵 Metals
✔️ Usually solid at room temperature (except mercury).
✔️ Have metallic lustre and high density.
✔️ Are malleable (can be beaten into thin sheets).
✔️ Are ductile (can be drawn into wires).
✔️ Are good conductors of heat and electricity.
✔️ Produce a sonorous sound when struck.
✔️ Have high melting and boiling points.
🟢 Non-metals
✔️ May be solid, liquid, or gas (e.g., carbon, bromine, oxygen).
✔️ Generally dull and brittle in solid state.
✔️ Poor conductors of heat and electricity (except graphite).
✔️ Have low density and low melting points.
✔️ Are non-sonorous.
✏️ Note: Diamond (a form of carbon) is the hardest known natural substance.
🔵 Chemical Properties of Metals
🔵 1. Reaction with Oxygen
➡️ Metals react with oxygen to form metal oxides.
➡️ Example: 4Na + O₂ → 2Na₂O
➡️ Metal oxides are basic in nature; they turn red litmus blue.
💡 Amphoteric oxides show both acidic and basic properties — e.g., Al₂O₃, ZnO.
🟢 2. Reaction with Water
➡️ Metals form metal hydroxides and hydrogen gas.
✔️ Na + H₂O → NaOH + H₂↑ (vigorous reaction)
✔️ Mg + H₂O → MgO + H₂ (slow reaction)
✏️ Metals like gold, silver, platinum do not react with water.
🔴 3. Reaction with Acids
➡️ Metals react with dilute acids to form hydrogen gas.
✔️ Zn + 2HCl → ZnCl₂ + H₂↑
💡 Hydrogen pop test: A lighted matchstick produces a “pop” sound when H₂ burns.
🟡 4. Reaction with Other Metal Salts (Displacement Reaction)
➡️ A more reactive metal displaces a less reactive one from its salt solution.
✔️ Fe + CuSO₄ → FeSO₄ + Cu
💡 Used to arrange metals in order of reactivity — the Reactivity Series.
🔵 Reactivity Series of Metals
➡️ Arranged from most reactive to least reactive:
K > Na > Ca > Mg > Al > Zn > Fe > Pb > H > Cu > Hg > Ag > Au
💡 Highly reactive metals (K, Na, Ca) react vigorously with air and water; hence stored under kerosene.
💡 Less reactive metals (Cu, Ag, Au) do not corrode easily and exist in free state.
🔬 Chemical Properties of Non-metals
🟢 1. Reaction with Oxygen
✔️ Non-metals form non-metallic oxides, which are acidic in nature.
➡️ S + O₂ → SO₂
➡️ SO₂ + H₂O → H₂SO₃ (acidic solution turns blue litmus red)
🔴 2. Reaction with Water
✔️ Non-metals do not react with water directly.
🔵 3. Reaction with Acids or Bases
✔️ Non-metals generally do not react with acids, as they are electron acceptors.
🟡 4. Reaction with Hydrogen
✔️ Forms covalent hydrides.
➡️ H₂ + Cl₂ → 2HCl
➡️ H₂ + S → H₂S
💡 Uses of Metals
✔️ Iron — for machinery, vehicles, tools.
✔️ Aluminium — for aircraft, utensils, wires.
✔️ Copper — for electrical wiring.
✔️ Zinc — for galvanising iron.
✔️ Gold and Silver — for jewellery and electrical contacts.
💡 Uses of Non-metals
✔️ Oxygen — respiration and combustion.
✔️ Nitrogen — fertilisers and inert atmosphere.
✔️ Chlorine — water purification.
✔️ Sulphur — medicines and matches.
✔️ Carbon — fuel and steel industry.
🔄 Corrosion of Metals
💡 Definition: The gradual deterioration of metals due to chemical reaction with moisture, air, or acids.
➡️ Example: Rusting of iron — Fe + O₂ + H₂O → Fe₂O₃·xH₂O
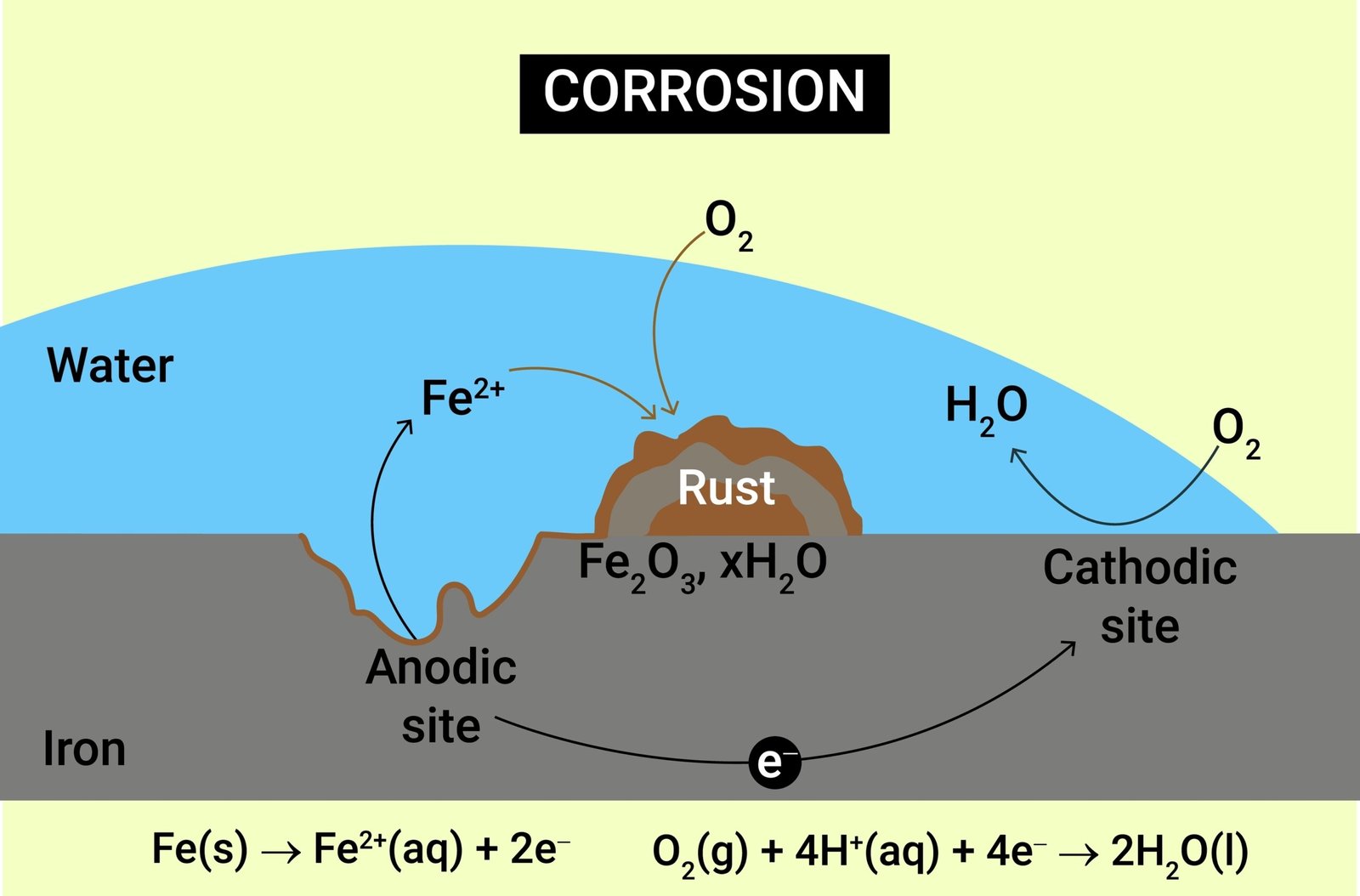
✏️ Prevention of Corrosion:
🔵 Painting
🟢 Oiling or greasing
🔴 Galvanisation (zinc coating)
🟡 Electroplating and alloying
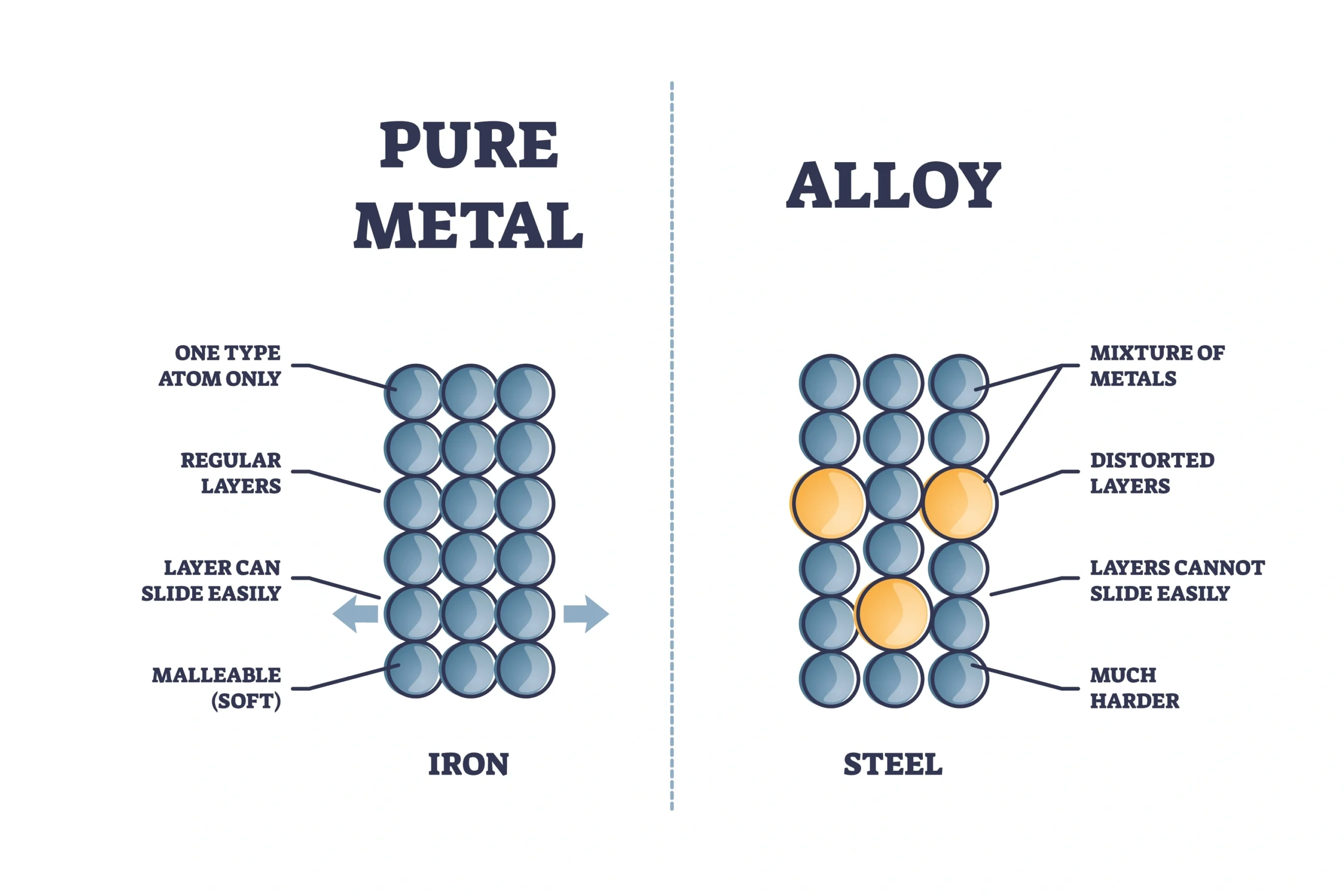
⚗️ Alloys
💡 Alloy — A homogeneous mixture of two or more metals, or a metal and a non-metal.
➡️ Examples:
✔️ Brass — Cu + Zn
✔️ Bronze — Cu + Sn
✔️ Solder — Pb + Sn
✔️ Steel — Fe + C
💡 Alloying improves hardness, strength, and corrosion resistance.
✳️ Metals and Non-metals in Daily Life
➡️ Iron for tools, copper wires for electricity, aluminium utensils for cooking, and silver for ornaments.
➡️ Non-metals like oxygen, nitrogen, and carbon are essential for respiration, agriculture, and energy.
📖 (Part 2) Summary
🔵 Metals are lustrous, malleable, ductile, and good conductors of heat and electricity.
🟢 Non-metals are dull, brittle, and poor conductors.
🔴 Metals form basic oxides; non-metals form acidic oxides.
🟡 Reactive metals displace less reactive ones from solutions.
✏️ Corrosion affects metals, prevented by painting or galvanising.
💡 Alloys are mixtures improving metal properties.
📝 (Part 3) Quick Recap
1️⃣ Metals form basic oxides; non-metals form acidic ones.
2️⃣ Reactivity series ranks metals by their tendency to lose electrons.
3️⃣ Corrosion prevention — painting, oiling, galvanising.
4️⃣ Alloys enhance strength and durability.
5️⃣ Everyday life uses — copper, aluminium, oxygen, sulphur, etc.
————————————————————————————————————————————————————————————————————————–
QUESTIONS FROM TEXTBOOK
🔵 Question 1: Which of the following pairs will give displacement reactions?
(a) NaCl solution and copper metal
(b) MgCl₂ solution and aluminium metal
(c) FeSO₄ solution and silver metal
(d) AgNO₃ solution and copper metal
✔️ Answer: (d) AgNO₃ solution and copper metal
💡 Copper is more reactive than silver and displaces silver from its salt solution:
➡️ Cu + 2AgNO₃ → Cu(NO₃)₂ + 2Ag
🟢 Question 2: Which of the following methods is suitable for preventing an iron frying pan from rusting?
(a) Applying grease
(b) Applying paint
(c) Applying a coating of zinc
(d) All of the above
✔️ Answer: (d) All of the above
💡 All these methods prevent the contact of iron with air and moisture, thus preventing rusting.
🔴 Question 3: An element reacts with oxygen to give a compound with a high melting point. This compound is also soluble in water. The element is likely to be —
(a) calcium
(b) carbon
(c) silicon
(d) iron
✔️ Answer: (a) calcium
💡 Calcium reacts with oxygen to form calcium oxide (CaO), which has a high melting point and forms calcium hydroxide (Ca(OH)₂) when dissolved in water.
🟡 Question 4: Food cans are coated with tin and not with zinc because —
(a) zinc is costlier than tin
(b) zinc has a higher melting point than tin
(c) zinc is more reactive than tin
(d) zinc is less reactive than tin
✔️ Answer: (c) zinc is more reactive than tin
💡 Zinc being more reactive could corrode and contaminate food; tin, being less reactive, is safer for coating.
🔵 Question 5: You are given a hammer, a battery, a bulb, wires and a switch.
(a) How could you use them to distinguish between samples of metals and non-metals?
(b) Assess the usefulness of these tests in distinguishing between metals and non-metals.
✔️ Answer (a):
➡️ Use the hammer to check malleability — metals are malleable; non-metals are brittle.
➡️ Connect the sample in a simple circuit with the battery, bulb, and wires — metals conduct electricity and make the bulb glow; non-metals do not.
✔️ Answer (b):
💡 These tests are useful but limited — some exceptions exist (e.g., graphite, a non-metal, conducts electricity).
🟢 Question 6: What are amphoteric oxides? Give two examples of amphoteric oxides.
✔️ Answer:
💡 Amphoteric oxides are those which react with both acids and bases to form salts and water.
➡️ Examples: Aluminium oxide (Al₂O₃) and Zinc oxide (ZnO)
🔴 Question 7: Name two metals which will displace hydrogen from dilute acids, and two metals which will not.
✔️ Answer:
➡️ Metals that displace hydrogen: Zinc (Zn), Iron (Fe)
➡️ Metals that do not: Copper (Cu), Silver (Ag)
💡 Metals above hydrogen in the reactivity series can displace hydrogen from dilute acids, while those below cannot.
🔵 Question 8:
In the electrolytic refining of a metal M, what would you take as the anode, the cathode and the electrolyte?
✔️ Answer:
➡️ In the electrolytic refining process:
- Anode: Impure metal (M)
- Cathode: Thin strip of pure metal (M)
- Electrolyte: Solution of a salt of the metal (e.g., CuSO₄ for copper)
💡 On passing current, pure metal from the anode dissolves into the electrolyte and gets deposited on the cathode.
🟢 Question 9:
Pratyush took sulphur powder on a spatula and heated it. He collected the gas evolved by inverting a test tube over it.
(a) What will be the action of gas on
(i) dry litmus paper?
(ii) moist litmus paper?
(b) Write a balanced chemical equation for the reaction taking place.
✔️ Answer:
(a)
(i) On dry litmus paper — No change.
(ii) On moist litmus paper — It turns blue litmus red because sulphur dioxide dissolves in water to form sulphurous acid.
(b) Balanced equation:
➡️ S + O₂ → SO₂
💡 Reaction shows that sulphur (a non-metal) forms an acidic oxide.
🔴 Question 10:
State two ways to prevent the rusting of iron.
✔️ Answer:
1️⃣ Applying oil, grease, or paint on the iron surface.
2️⃣ Galvanisation — coating iron with a thin layer of zinc metal.
💡 Both methods prevent air and moisture from coming in contact with iron.
🟡 Question 11:
What type of oxides are formed when non-metals combine with oxygen?
✔️ Answer:
➡️ Non-metals form acidic oxides when they combine with oxygen.
Example:
C + O₂ → CO₂ (carbon dioxide is acidic)
💡 Some non-metallic oxides can be neutral (e.g., CO, N₂O).
🔵 Question 12:
Give reasons:
(a) Platinum, gold and silver are used to make jewellery.
✔️ They are highly lustrous, malleable, and resistant to corrosion.
(b) Sodium, potassium and lithium are stored under oil.
✔️ They are highly reactive with air and water; oil prevents contact with moisture and oxygen.
(c) Aluminium is a highly reactive metal, yet it is used to make utensils for cooking.
✔️ It forms a protective oxide layer (Al₂O₃) that prevents further reaction and corrosion.
(d) Carbonate and sulphide ores are usually converted into oxides during the process of extraction.
✔️ Metal oxides are easier to reduce with carbon or other reducing agents than carbonates or sulphides.
🟢 Question 13:
You must have seen tarnished copper vessels being cleaned with lemon or tamarind juice. Explain why these sour substances are effective in cleaning the vessels.
✔️ Answer:
💡 Lemon or tamarind juice contains acids (citric or tartaric acid) that react with the basic copper carbonate layer formed on the vessel:
➡️ CuCO₃ + 2H⁺ → Cu²⁺ + CO₂ + H₂O
This removes the green coating, making the copper surface shine again.
🔴 Question 14:
Differentiate between metal and non-metal on the basis of their chemical properties.
✔️ Answer:
🔹 Metals
➡️ Form basic oxides.
➡️ Displace hydrogen from acids.
➡️ Form positive ions (electropositive).
🔹 Non-metals
➡️ Form acidic or neutral oxides.
➡️ Do not displace hydrogen from acids.
➡️ Form negative ions (electronegative).
🟡 Question 15:
A man went door to door posing as a goldsmith. He promised to bring back the glitter of old ornaments. An unsuspecting lady gave him a set of gold bangles to polish. He dipped the bangles in a particular solution and returned them shining like new. Later she found the weight reduced drastically. The lady was upset but after a futile argument, she had to accept the loss. Can you play the detective to find out the nature of the solution he had used?
✔️ Answer:
💡 The solution used was aqua regia, a mixture of concentrated hydrochloric acid (HCl) and nitric acid (HNO₃) in 3:1 ratio.
➡️ Aqua regia dissolves gold and silver, forming soluble chloroauric acid (HAuCl₄).
➡️ Hence, some part of gold was lost, causing reduction in weight.
🔵 Question 16:
Give reasons why copper is used to make hot water tanks and not steel (an alloy of iron).
✔️ Answer:
➡️ Copper does not react with water even at high temperatures, hence resists corrosion.
➡️ Steel (iron alloy) reacts with water and corrodes over time.
💡 Therefore, copper is more durable and suitable for hot water tanks.
——————————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTION PAPER)
ESPECIALLY MADE FROM THIS CHAPTER ONLY
⚙️ Section A: Q1–6 (1 Mark Each – Very Short/MCQ Type)
🔵 Question 1: Which of the following metals can displace hydrogen from dilute acids?
🔵 (A) Copper
🟢 (B) Silver
🔴 (C) Zinc
🟡 (D) Gold
✔️ Answer: (C) Zinc
💡 Zinc is more reactive than hydrogen, so it displaces hydrogen gas from dilute acids.
🔵 Question 2: Which of the following is an amphoteric oxide?
🔵 (A) CO₂
🟢 (B) SO₂
🔴 (C) Al₂O₃
🟡 (D) CaO
✔️ Answer: (C) Al₂O₃
💡 Aluminium oxide reacts with both acids and bases to form salt and water.
🔵 Question 3: The property of metals by which they can be drawn into wires is called —
🔵 (A) Malleability
🟢 (B) Ductility
🔴 (C) Sonority
🟡 (D) Conductivity
✔️ Answer: (B) Ductility
💡 Ductility is the ability of metals to be stretched into thin wires.
🔵 Question 4: Which one of the following is the most reactive metal?
🔵 (A) Fe
🟢 (B) Zn
🔴 (C) Na
🟡 (D) Cu
✔️ Answer: (C) Na
💡 Sodium is highly reactive and placed at the top among these in the reactivity series.
🔵 Question 5: Rusting of iron takes place in the presence of —
🔵 (A) Oxygen only
🟢 (B) Moisture only
🔴 (C) Both oxygen and moisture
🟡 (D) Carbon dioxide
✔️ Answer: (C) Both oxygen and moisture
💡 Rusting requires both air (oxygen) and water to form hydrated ferric oxide.
🔵 Question 6: Food cans are coated with tin because —
🔵 (A) Tin is more reactive than iron
🟢 (B) Tin is less reactive than iron
🔴 (C) Tin is cheaper than iron
🟡 (D) Tin gives flavour to food
✔️ Answer: (B) Tin is less reactive than iron
💡 Tin prevents food from reacting with the metal, protecting it from corrosion.
⚡ Section B: Q7–12 (2 Marks Each – Short Answers)
🔴 Question 7: Why are sodium and potassium stored under kerosene?
✔️ Answer: They are highly reactive metals which react vigorously with air and water.
➡️ Storing under kerosene prevents contact with oxygen and moisture.
🟡 Question 8: Define corrosion and rusting.
✔️ Answer:
➡️ Corrosion is the gradual destruction of metals by chemical reactions with air, moisture, or other substances.
➡️ Rusting is corrosion of iron forming hydrated ferric oxide (Fe₂O₃·xH₂O).
🔵 Question 9: Write the balanced equation for the reaction of iron with copper sulphate solution.
✔️ Answer: Fe + CuSO₄ → FeSO₄ + Cu
💡 Iron displaces copper as it is more reactive.
🟢 Question 10: Why are alloys made? Give two examples.
✔️ Answer:
💡 Alloys are made to improve properties like strength, hardness, and resistance to corrosion.
➡️ Examples: Brass (Cu + Zn), Steel (Fe + C).
🔴 Question 11: Name two metals that are found in the free state in nature. Why?
✔️ Answer: Gold and Platinum — because they are very unreactive and do not combine with other elements.
🟡 Question 12: Why are aluminium utensils used for cooking?
✔️ Answer: Aluminium is a good conductor of heat and forms a protective oxide layer that prevents further corrosion.
⚙️ Section C: Q13–22 (3 Marks Each – Short to Mid-Length)
🔵 Question 13: Explain the reactivity series of metals with suitable examples.
✔️ Answer:
➡️ The reactivity series arranges metals in order of decreasing reactivity.
➡️ Highly reactive metals like K, Na react violently with water.
➡️ Moderately reactive metals like Zn, Fe react slowly.
➡️ Least reactive metals like Ag, Au do not react with air or water.
🟢 Question 14: Describe an activity to show that copper cannot displace zinc from its salt solution.
✔️ Answer:
➡️ Take Cu strip in ZnSO₄ solution.
➡️ No colour change or deposition observed — no reaction occurs.
➡️ This shows copper is less reactive than zinc.
🔴 Question 15: What is the composition and use of alloy brass?
✔️ Answer:
➡️ Composition: Copper (Cu) + Zinc (Zn).
➡️ Use: For making electrical fittings, utensils, and musical instruments.
🟡 Question 16: Explain why iron articles are painted.
✔️ Answer:
💡 Paint forms a protective layer preventing air and moisture from reaching iron surface, thus preventing rust.
🔵 Question 17: What are the uses of non-metals in daily life?
✔️ Answer:
➡️ Oxygen — for respiration.
➡️ Nitrogen — for fertilisers.
➡️ Sulphur — for medicines.
➡️ Carbon — as fuel and reducing agent.
🟢 Question 18: Write differences between metals and non-metals based on physical properties.
✔️ Answer:
➡️ Metals — lustrous, malleable, ductile, good conductors.
➡️ Non-metals — dull, brittle, non-ductile, poor conductors.
🔴 Question 19: Explain electrolytic refining with the help of a labelled diagram.
✔️ Answer:
➡️ Impure metal is made the anode, pure metal the cathode, and a salt solution as electrolyte.
➡️ On passing electric current, pure metal deposits on cathode, impurities settle below anode as anode mud.
🟡 Question 20: Why does aluminium not corrode easily though it is a reactive metal?
✔️ Answer:
💡 It forms a thin protective oxide layer (Al₂O₃) that prevents further oxidation.
🔵 Question 21: Give any three uses of alloys.
✔️ Answer:
1️⃣ Brass — utensils and electrical parts.
2️⃣ Bronze — medals and statues.
3️⃣ Stainless steel — surgical instruments and kitchenware.
🟢 Question 22: Explain the reaction between magnesium and steam.
✔️ Answer:
➡️ Mg + H₂O (steam) → MgO + H₂
💡 Hydrogen gas is evolved and magnesium oxide forms as white solid.
🧠 Section D: Q23–30 (4 Marks Each – Long and Case-Based)
🔴 Question 23: Explain with equations how metals react with acids and bases.
✔️ Answer:
➡️ Metals react with acids to produce hydrogen gas and salt.
Example: Zn + 2HCl → ZnCl₂ + H₂↑
➡️ Amphoteric metals like Al react with bases too:
2Al + 2NaOH + 2H₂O → 2NaAlO₂ + 3H₂↑
💡 These show the dual reactivity of amphoteric metals.
🟡 Question 24: What is corrosion? How can it be prevented?
✔️ Answer:
➡️ Corrosion is the slow destruction of metals due to chemical reactions with the environment.
➡️ Example: Rusting of iron (Fe₂O₃·xH₂O).
➡️ Prevention:
1️⃣ Painting or greasing.
2️⃣ Galvanisation.
3️⃣ Electroplating.
4️⃣ Alloying with chromium/nickel (stainless steel).
🔵 Question 25: Compare the properties of ionic and covalent compounds.
✔️ Answer:
➡️ Ionic compounds — formed by transfer of electrons, high melting point, soluble in water, conduct electricity in molten state.
➡️ Covalent compounds — formed by sharing of electrons, low melting point, poor conductors, usually insoluble in water.
🟢 Question 26: Describe an experiment to show that hydrogen gas is evolved when metals react with acids.
✔️ Answer:
➡️ Set up test tube with Zn granules and dilute HCl.
➡️ Observe gas bubbles and test by bringing burning matchstick — it burns with a pop sound.
➡️ Reaction: Zn + 2HCl → ZnCl₂ + H₂↑
💡 Pop sound confirms presence of hydrogen gas.
🔴 Question 27: Explain the meaning of the term “reactivity series.” How does it help in predicting the products of displacement reactions?
✔️ Answer:
➡️ The reactivity series lists metals in decreasing order of reactivity.
➡️ A metal higher in the series displaces another lower in the series from its compound.
➡️ Example: Fe + CuSO₄ → FeSO₄ + Cu.
💡 It helps identify which metals can displace others in chemical reactions.
🟡 Question 28: A case-based study:
A student observed that aluminium foil does not corrode in air, while an iron nail rusts easily. Explain why.
✔️ Answer:
➡️ Aluminium forms a tough oxide layer (Al₂O₃) that protects it.
➡️ Iron reacts with oxygen and water to form Fe₂O₃·xH₂O, which flakes off exposing more metal.
💡 Thus, iron rusts but aluminium resists corrosion.
————————————————————————————————————————————————————————————————————————————
