Class 7 : Science – ( English ) : Lesson 4. The World of Metals and Non-metals
EXPLANATION AND ANALYSIS
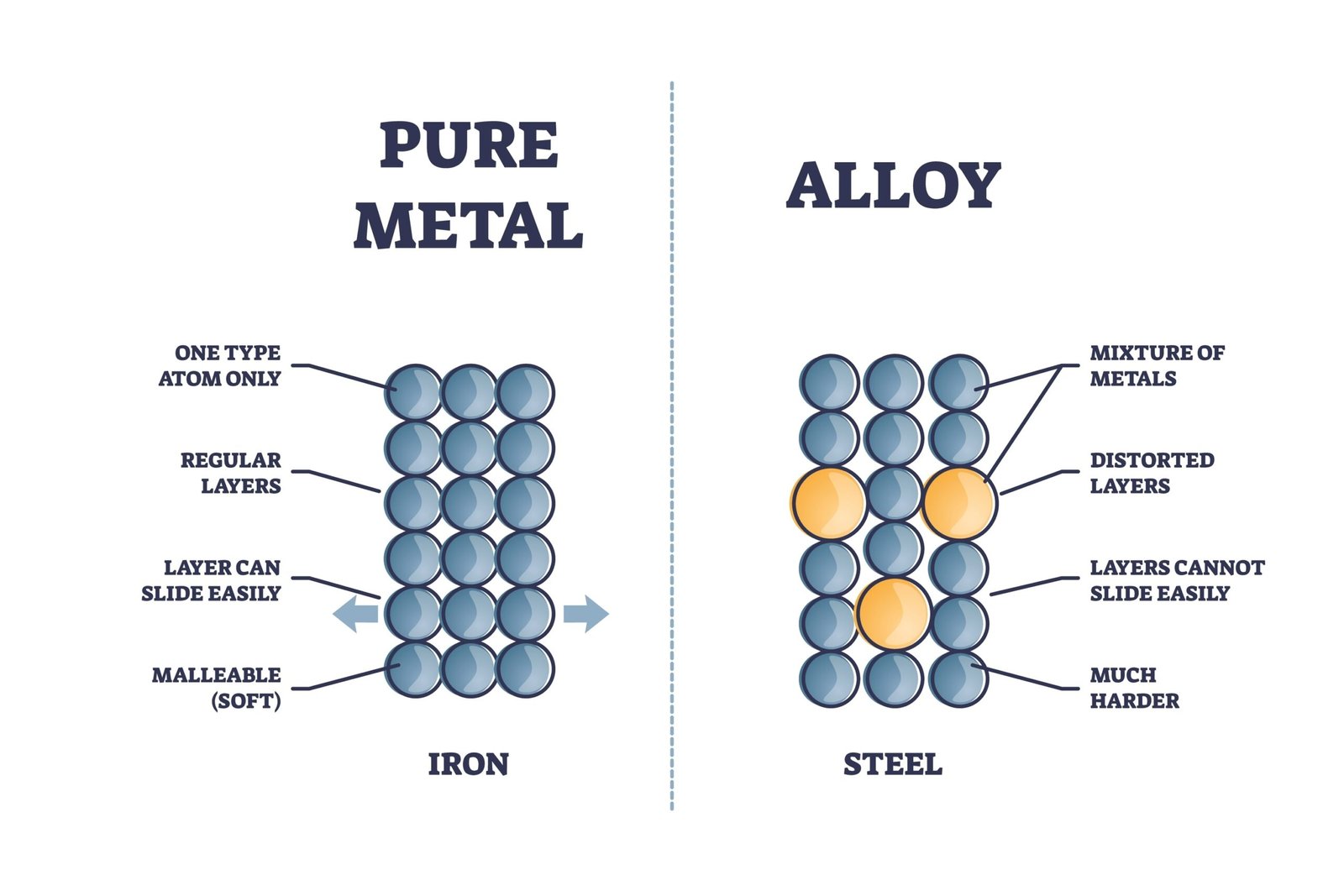
🧭 Materials around us are not the same.
Some are hard and shiny ✨, some are dull 🌫️, some bend easily, while others break. These differences help us classify materials into metals and non-metals, which form an important part of science and daily life.
🧠 From cooking utensils 🍳 to electric wires 🔌, from coins 🪙 to buildings 🏗️, metals are everywhere.
At the same time, non-metals like oxygen 🌬️, carbon 🖤, and sulphur 🟡 are equally essential for life and nature.
🔍 Understanding metals and non-metals helps us choose materials wisely and use them safely.
🧪 Metals are materials that usually have special physical properties.
Most metals are hard, shiny, and strong.
✨ Metals have a shiny surface called lustre.
🧲 Metals are generally hard and strong.
🔔 Metals produce sound when struck.
🧠 Examples of metals include iron 🧲, copper 🟠, aluminium 🪙, and gold 🟡.
⚙️ One important property of metals is malleability.
Malleability means metals can be beaten into thin sheets without breaking.
🪙 Gold can be beaten into very thin sheets.
🏠 Aluminium sheets are used for roofs and packaging.
🧠 This property makes metals useful for making tools, utensils, and machines.
🔩 Metals are also ductile, which means they can be drawn into thin wires.
Copper wires 🔌 are used in electrical connections.
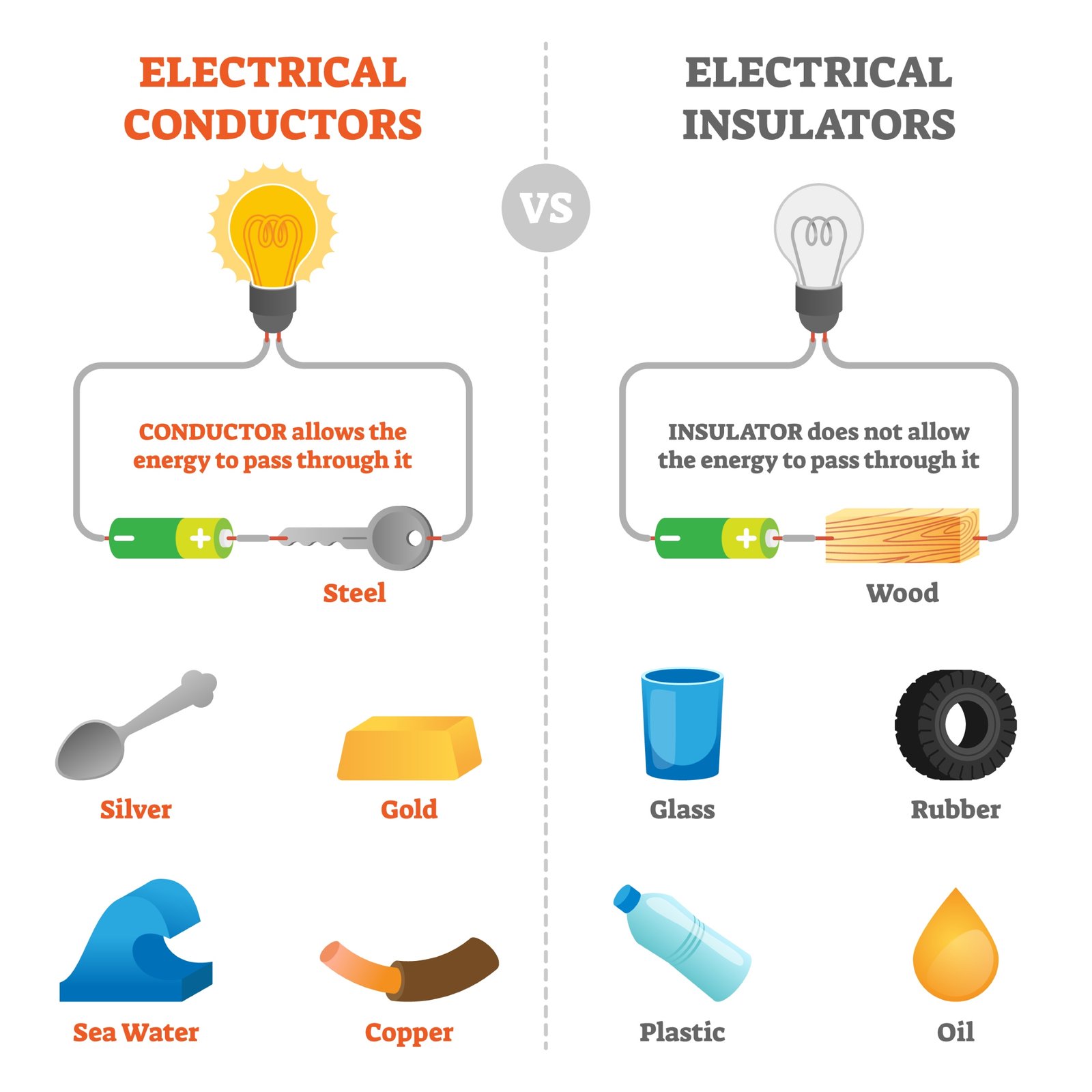
⚡ Electricity flows easily through metal wires because metals are good conductors.
🧠 This property makes metals essential for electrical devices.
🔥 Metals are good conductors of heat.
They quickly transfer heat from one place to another.
🍳 Cooking utensils are made of metals because they heat up quickly and evenly.
🧠 This helps save time and fuel.
🧪 Non-metals are materials that show properties opposite to metals.
Most non-metals are dull, soft, and weak.
🌫️ Non-metals usually do not shine.
🚫 They are poor conductors of heat and electricity.
🧠 Examples of non-metals include oxygen 🌬️, sulphur 🟡, carbon 🖤, and nitrogen 🌫️.
📦 Many non-metals are brittle.
They break easily when hit.
🧠 Sulphur breaks into powder when struck.
This makes non-metals unsuitable for making tools.
🌬️ Some non-metals exist as gases.
Oxygen 🌬️ is needed for breathing.
Nitrogen 🌫️ is important for plant growth.
🧠 Non-metals play a key role in supporting life.
🧪 Metals and non-metals also differ in their chemical behavior.
Metals react with oxygen to form metal oxides 🧪.
🔥 Iron reacts with oxygen to form rust 🟤.
Rust weakens iron objects.
🧠 This is why iron objects are painted or coated.
🌬️ Non-metals also react with oxygen.
They form non-metal oxides.
🧪 These oxides often dissolve in water and form acids.
🧠 This shows how non-metals are involved in chemical changes.
⚙️ Metals react with water differently.
Some metals react strongly, while others do not react at all.
🔥 Sodium reacts violently with water.
🧊 Iron reacts slowly with water and air.
🧠 This knowledge helps prevent accidents.
🧪 Metals react with acids to produce hydrogen gas 💨.
This reaction helps identify metals.
🧠 Non-metals usually do not react with acids in this way.
🌍 Metals and non-metals are used widely in daily life.
Metals are used in construction 🏗️, transport 🚆, and machines ⚙️.
📱 Non-metals are used in fertilizers 🌱, medicines 💊, and fuels 🔥.
🧠 Both are essential for human progress.
🧠 Recycling metals helps protect the environment.
♻️ Metals can be reused many times without losing quality.
🌱 This reduces mining and saves natural resources.
🧭 Learning about metals and non-metals builds scientific thinking.
It teaches comparison 🧩.
It teaches observation 👀.
It teaches safe handling ⚠️.
🧠 This knowledge helps us respect materials and nature.
📜 Metals and non-metals together support modern life.
They form the foundation of science, technology, and industry.
🚀 Understanding their properties prepares students for advanced science.
📝 Summary of the Lesson
Materials are classified into metals and non-metals based on their properties. Metals are generally shiny, hard, malleable, ductile, and good conductors of heat and electricity. They are used in making tools, machines, utensils, and electrical wires. Non-metals are usually dull, soft, brittle, and poor conductors. They play important roles in life processes, agriculture, and industry. Metals and non-metals also differ in their chemical behavior, such as reactions with oxygen, water, and acids. Both types of materials are essential for daily life and scientific development. Understanding their properties helps us use them safely and wisely.
⚡ Quick Recap
⭐ Metals are shiny and strong
⭐ Metals conduct heat and electricity
⭐ Non-metals are dull and brittle
⭐ Non-metals support life processes
⭐ Metals react with acids
⭐ Properties decide uses
⭐ Both are essential for life
——————————————————————————————————————————————————————————————————————————–
TEXTBOOK QUESTIONS
🔒 ❓ Question 1.
Which metal is commonly used to make food packaging materials as it is cheaper and its thin sheets can be folded easily into any shape?
📌 Answer:
The metal used is aluminium.
Aluminium is light in weight, inexpensive, and highly malleable, which means it can be beaten into very thin sheets. These thin sheets can be easily folded into different shapes and are widely used for food packaging because aluminium does not react with food and protects it from moisture and air.
🔒 ❓ Question 2.
Which of the following metal catches fire when it comes in contact with water?
(i) Copper (ii) Aluminium (iii) Zinc (iv) Sodium
📌 Answer:
The correct answer is (iv) Sodium.
Sodium is a highly reactive metal. When it comes in contact with water, it reacts violently, producing heat and hydrogen gas, which can catch fire immediately.
🔒 ❓ Question 3.
State with reasons whether the following statements are True (T) or False (F).
📌 Answer:
(i) Aluminium and copper are examples of non-metals used for making utensils and statues.
False. Aluminium and copper are metals, not non-metals, and they are used because they are strong and good conductors of heat.
(ii) Metals form oxides when combined with oxygen, the solution of which turns blue litmus paper to red.
False. Metal oxides are generally basic, and their solutions turn red litmus blue, not blue to red.
(iii) Oxygen is a non-metal essential for respiration.
True. Oxygen is a non-metal and is required by living organisms for respiration.
(iv) Copper vessels are used for boiling water because they are good conductors of electricity.
False. Copper vessels are used because copper is a good conductor of heat, not electricity.
🔒 ❓ Question 4.
Why are only a few metals suitable for making jewellery?
📌 Answer:
Only a few metals like gold, silver, and platinum are suitable for making jewellery because they are lustrous, do not corrode easily, and are malleable. Most metals react with air and moisture, lose their shine, or rust, which makes them unsuitable for jewellery.
🔒 ❓ Question 5.
Match the uses of metals and non-metals given in Column I with the jumbled names in Column II.
📌 Answer:
(i) Used in electrical wiring → Copper (PEPORC)
(ii) Most malleable and ductile → Gold (OGDL)
(iii) Living organisms cannot survive without it → Oxygen (ENXYGO)
(iv) Plants grow healthy when fertilisers containing it are added → Nitrogen (TENGOINR)
(v) Used in water purification → Chlorine (NECOHIRL)
🔒 ❓ Question 6.
What happens when oxygen reacts with magnesium and sulfur? What are the main differences in the nature of products formed?
📌 Answer:
When magnesium reacts with oxygen, it forms magnesium oxide, which is a basic oxide.
When sulfur reacts with oxygen, it forms sulfur dioxide, which is an acidic oxide.
The main difference is that magnesium oxide shows basic nature, while sulfur dioxide shows acidic nature.
🔒 ❓ Question 7.
Complete the following flow chart.
📌 Answer:
Metal + Air + Heat → Ash
Ash + Water → Solution
When tested with litmus:
Blue litmus → No change
Red litmus → Turns blue
This shows that the solution formed is basic in nature.
🔒 ❓ Question 8.
You are provided with the following materials. Which material would you choose to make a pan suitable for boiling water and why?
(Iron, copper, sulfur, coal, plastic, wood, cardboard)
📌 Answer:
Copper would be the best choice.
Copper is a good conductor of heat, strong, and does not melt easily. It allows uniform heating, which makes it ideal for boiling water. Other materials either do not conduct heat well or may burn or melt.
🔒 ❓ Question 9.
You are provided with three iron nails, each dipped in oil, water, and vinegar. Which iron nail will not rust, and why?
📌 Answer:
The iron nail dipped in oil will not rust.
Oil prevents air and moisture from reaching the iron surface. Since rusting requires both air and water, the nail in oil remains protected from rust.
🔒 ❓ Question 10.
How do the different properties of metals and non-metals determine their uses in everyday life?
📌 Answer:
Metals are hard, shiny, and good conductors of heat and electricity, so they are used in utensils, wires, and machines.
Non-metals are usually poor conductors and may be soft or brittle, so they are used in making fertilizers, medicines, and fuels. Their properties decide where and how they are used.
🔒 ❓ Question 11.
One method of protecting iron from rusting is coating it with zinc. Since sulfur does not react with water, can it be used for this purpose? Give reason.
📌 Answer:
No, sulfur cannot be used for this purpose.
Zinc protects iron by forming a protective layer and reacting first with air and moisture. Sulfur does not form such a protective coating and cannot prevent iron from coming in contact with air and water.
🔒 ❓ Question 12.
An ironsmith heats iron before making tools. Why is heating necessary in this process?
📌 Answer:
Heating makes iron soft and malleable.
When iron is heated, it becomes easier to shape, bend, and hammer into tools without breaking. This helps the ironsmith to give the desired shape efficiently.
——————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS
SECTION 1 — MCQs (5 Questions)
🔒 ❓ Q1. Which property is commonly shown by metals?
🟢 1️⃣ Dull appearance
🔵 2️⃣ Brittle nature
🟡 3️⃣ Lustre
🟣 4️⃣ Solubility
✔️ Answer: 🟡 3️⃣ Lustre
🔒 ❓ Q2. Which metal is liquid at room temperature?
🟢 1️⃣ Iron
🔵 2️⃣ Copper
🟡 3️⃣ Mercury
🟣 4️⃣ Aluminium
✔️ Answer: 🟡 3️⃣ Mercury
🔒 ❓ Q3. Which of the following is a non-metal?
🟢 1️⃣ Iron
🔵 2️⃣ Copper
🟡 3️⃣ Sulphur
🟣 4️⃣ Aluminium
✔️ Answer: 🟡 3️⃣ Sulphur
🔒 ❓ Q4. Which property allows metals to be beaten into sheets?
🟢 1️⃣ Ductility
🔵 2️⃣ Malleability
🟡 3️⃣ Conductivity
🟣 4️⃣ Hardness
✔️ Answer: 🔵 2️⃣ Malleability
🔒 ❓ Q5. Which material is used for making electric wires?
🟢 1️⃣ Plastic
🔵 2️⃣ Rubber
🟡 3️⃣ Copper
🟣 4️⃣ Wood
✔️ Answer: 🟡 3️⃣ Copper
SECTION 2 — Very Short Answer (5 Questions)
🔒 ❓ Q6. Name one metal.
📌 ✅ Answer: Iron
🔒 ❓ Q7. Name one non-metal.
📌 ✅ Answer: Sulphur
🔒 ❓ Q8. Which metal is used in thermometers?
📌 ✅ Answer: Mercury
🔒 ❓ Q9. What property allows metals to conduct electricity?
📌 ✅ Answer: Conductivity
🔒 ❓ Q10. Which non-metal is essential for breathing?
📌 ✅ Answer: Oxygen
SECTION 3 — Short Answer (3 Questions)
🔒 ❓ Q11. Why are metals generally good conductors of electricity?
📌 ✅ Answer:
🔹 Metals allow electric current to pass through easily.
🔸 Their free electrons move freely inside the metal.
🔹 This movement of electrons helps in conducting electricity.
🔒 ❓ Q12. How are metals different from non-metals in terms of physical properties?
📌 ✅ Answer:
🔹 Metals are shiny, hard and malleable.
🔸 Non-metals are generally dull and brittle.
🔹 Metals conduct heat and electricity, while non-metals usually do not.
🔒 ❓ Q13. Why is copper widely used for making electrical wires?
📌 ✅ Answer:
🔹 Copper is a very good conductor of electricity.
🔸 It is flexible and can be drawn into thin wires.
🔹 Hence, copper is suitable for electrical wiring.
SECTION 4 — Long Answer (1 Question)
🔒 ❓ Q14. Explain the properties and uses of metals and non-metals.
📌 ✅ Answer:
🔹 Metals are shiny, hard and good conductors of heat and electricity.
🔸 They are used to make wires, utensils and tools.
🔹 Non-metals are usually dull and poor conductors.
🔸 They are used in making fertilizers, medicines and fuels.
——————————————————————————————————————————————————————————————————————————–
ADVANCE KNOWLEDGE
🌍 A World Built from Two Invisible Teams
Look around you—buildings, wires, vehicles, utensils, medicines, even your own body. Behind everything stand two great groups of substances: metals and non-metals. They may look ordinary, but together they have shaped human civilization from the Stone Age to the Space Age.
🧠 Big idea:
Metals and non-metals are not just materials—they are behaviour patterns of matter.
⏳ A Short Journey Through History
⏳ Human history itself is named after metals.
⏳ Ages of civilization:
Stone Age
Bronze Age
Iron Age
🧠 Discovery of metals changed:
Tools
Agriculture
Warfare
Trade
⭐ Without metals, modern science and technology could not exist.
⚙️ What Makes a Metal a Metal?
Metals share a special set of properties.
🧠 Key metallic behaviours:
✨ Shiny appearance
🔨 Can be hammered into sheets
🔌 Allow electricity to flow
🔥 Can withstand high temperatures
🧪 Deep reason:
Metal atoms allow their electrons to move freely—this creates strength, shine, and conductivity.
⚠️ Misconception vs Reality
⚠️ Misconception: All metals are hard
✅ Reality: Sodium and potassium are soft enough to cut with a knife
⚠️ Misconception: All metals sink in water
✅ Reality: Some metals float depending on density
⚠️ Misconception: Metals never break
✅ Reality: Metals can fatigue and fracture over time
🌌 Non-metals: The Quiet Controllers
Non-metals may look less impressive, but life depends on them.
🌱 Important non-metals:
Oxygen
Carbon
Nitrogen
Sulphur
🧠 Without non-metals:
No breathing
No food
No DNA
No life
⭐ Carbon alone forms millions of compounds—more than all other elements combined.
🧪 Why Non-metals Behave Differently
🧠 Non-metals hold their electrons tightly.
This causes them to:
Be poor conductors
Break easily if solid
Exist as gases, liquids, or brittle solids
⭐ Their strength lies in chemical bonding, not physical toughness.
🔥 Metals, Non-metals, and Energy
Metals handle heat and electricity well.
🔌 Copper → electric wires
🔥 Iron → engines and tools
Non-metals often control reactions.
🧪 Oxygen → burning
🌱 Nitrogen → plant growth
🧠 Together they manage Earth’s energy systems.
⚗️ Chemical Behaviour: Reactivity Secrets
Not all metals react equally.
⚡ Highly reactive:
Sodium
Potassium
🧱 Less reactive:
Gold
Silver
🧠 Reactivity decides:
Extraction method
Usage
Storage safety
⭐ Gold stays shiny because it refuses to react.
🧲 Metals and Non-metals in the Human Body
Your body is a living chemistry lab.
🧠 Metals in the body:
Iron → blood oxygen transport
Calcium → bones and teeth
🧠 Non-metals in the body:
Oxygen → respiration
Carbon → life molecules
⭐ Life is impossible without both groups working together.
🌍 Metals from the Earth: A Hidden Cost
⛏️ Metals are extracted from ores.
⚠️ Mining problems:
Deforestation
Pollution
Energy use
🧠 Metals take millions of years to form—once used up, they cannot be replaced.
♻️ Recycling: Modern Science’s Responsibility
🚀 Recycling metals:
Saves energy
Reduces pollution
Protects nature
⭐ Aluminium recycling saves up to 95% energy compared to fresh extraction.
🧠 Recycling is not just waste management—it is resource science.
🚀 Modern Science and Advanced Uses
🚀 Today’s science designs:
Lightweight alloys
Super-strong metals
Smart non-metal materials
🧠 Used in:
Spacecraft
Medical implants
Renewable energy systems
⭐ Silicon (a non-metal) runs the digital world.
⚠️ Safety and Care
Some metals and non-metals are dangerous.
⚠️ Mercury → toxic
⚠️ Lead → harmful to brain
⚠️ Chlorine → poisonous gas
🧠 Science teaches controlled use, not fear.
⭐ Amazing Facts
⭐ Gold does not rust
⭐ Iron in blood and iron in nails are related
⭐ Diamond and coal are both carbon
⭐ Non-metals control most chemical reactions
🧠 Why Curious Minds Must Look Deeper
Metals and non-metals explain:
Material science
Chemistry
Technology
Environmental balance
🧠 Understanding them helps young minds:
Think logically
Design responsibly
Respect Earth’s limits
🌟 Final Thought
Metals build strength.
Non-metals build life.
🧠 Civilization exists because these two opposite worlds work together in balance.
——————————————————————————————————————————————————————————————————————————–
