Class 8 : Science – ( English ) : Lesson 8. Nature of Matter: Elements, Compounds, and Mixtures
EXPLANATION AND ANALYSIS
🧭 Everything around us is made of matter, but matter does not exist in just one form.
Matter is present as elements 🧪, compounds ⚗️, and mixtures 🧫. Understanding these forms helps us know what substances are made of and how they behave in daily life and science.
🧠 From air 🌬️ and water 💧 to metals 🔩 and food 🍲, all materials belong to one of these categories.
🔍 Studying the nature of matter builds the foundation of chemistry.
🧪 An element is a pure substance made of only one kind of particle.
It cannot be broken into simpler substances by ordinary methods.
🧠 Elements are the basic building blocks of matter.
🧭 Examples of elements are found everywhere.
Iron 🔩
Oxygen 🌬️
Carbon 🧱
Copper 🪙
🧠 Each element has its own properties and uses.
🧬 Elements are classified into different types.
Metals ⚙️
Non-metals 🧪
Metalloids ⚖️
🧠 This classification helps us understand their behavior.
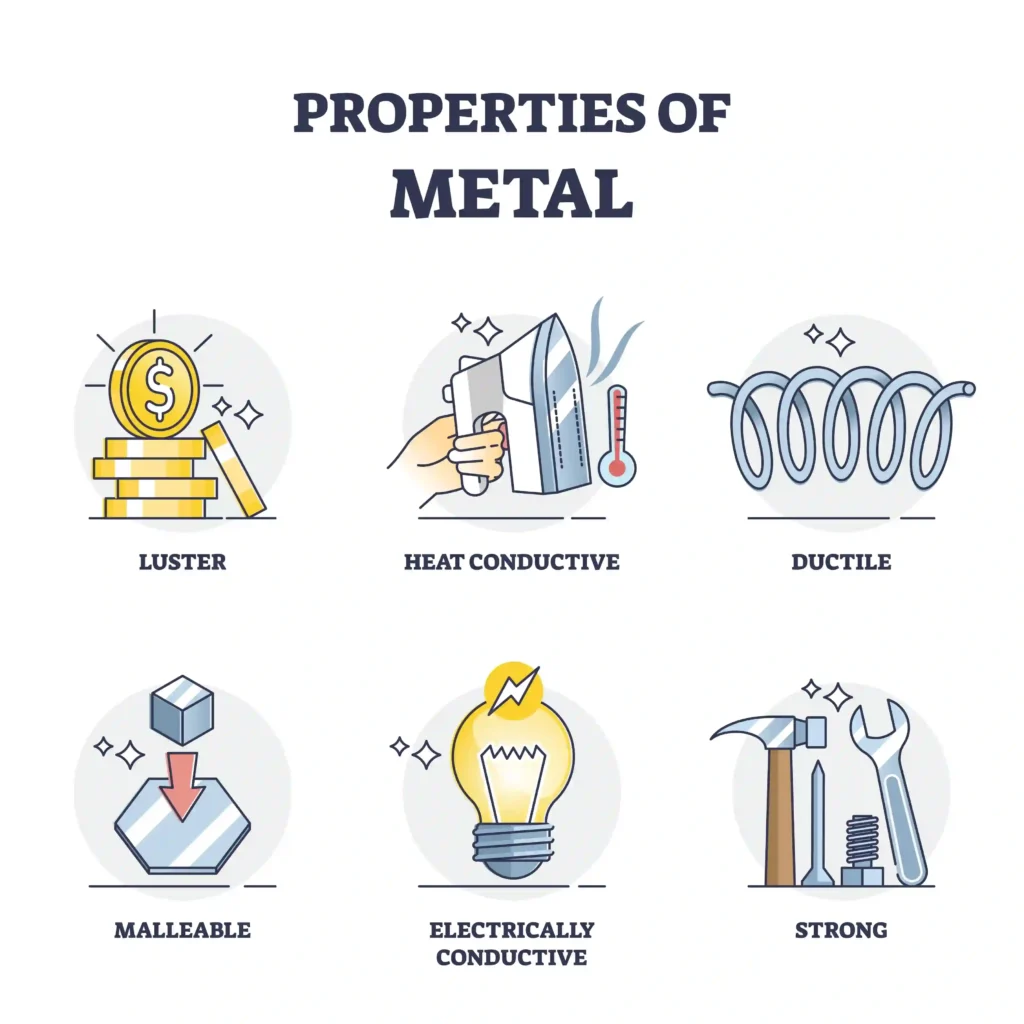
⚙️ Metals are generally hard, shiny, and strong.
They conduct heat 🔥 and electricity ⚡.
🧠 Iron is used in buildings 🏗️.
Copper is used in wires 🔌.
🧪 Non-metals are usually soft and dull.
They do not conduct heat or electricity easily.
🧠 Oxygen helps in breathing 🫁.
Carbon forms fuels and living tissues 🌱.
🧭 Some elements combine to form compounds.
A compound is a pure substance made by chemical combination of two or more elements.
🧠 Compounds have fixed composition and definite properties.
⚗️ Water 💧 is a compound formed from hydrogen and oxygen.
Salt 🧂 is formed from sodium and chlorine.
🧠 Compounds are different from the elements that form them.
🧭 Compounds cannot be separated by physical methods.
They require chemical reactions to break them.
🧠 This shows strong bonding between particles.
🧪 A mixture is formed when two or more substances are mixed physically.
No chemical change occurs.
🧠 Each component keeps its own properties.
🧭 Mixtures are very common in daily life.
Air 🌬️
Soil 🌱
Salt water 🌊🧂
Tea 🍵
🧠 Mixtures can be separated easily.
🧪 Mixtures are of different types.
Homogeneous mixtures 🧫
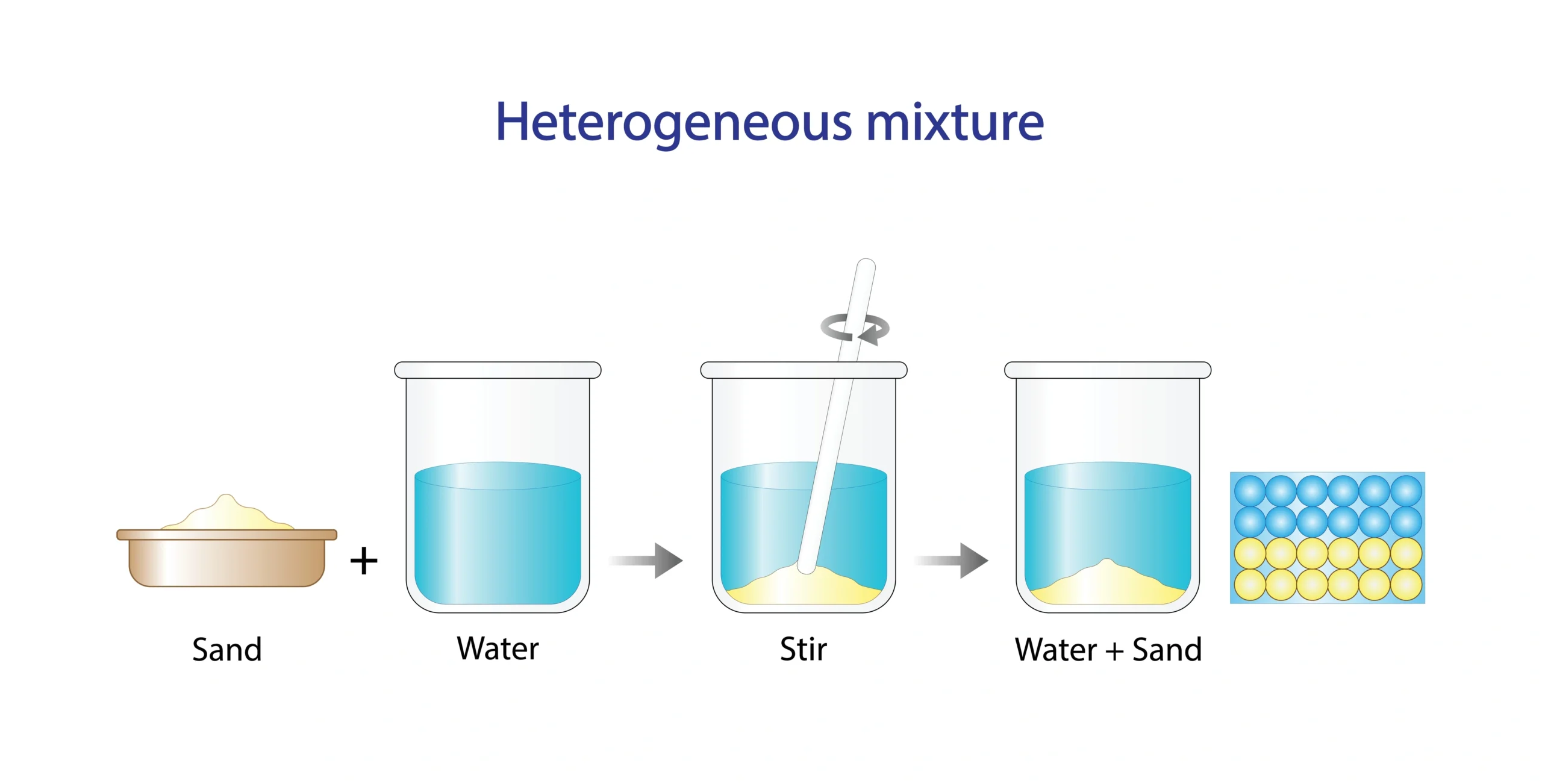
Heterogeneous mixtures 🧺
🧠 This depends on uniformity.
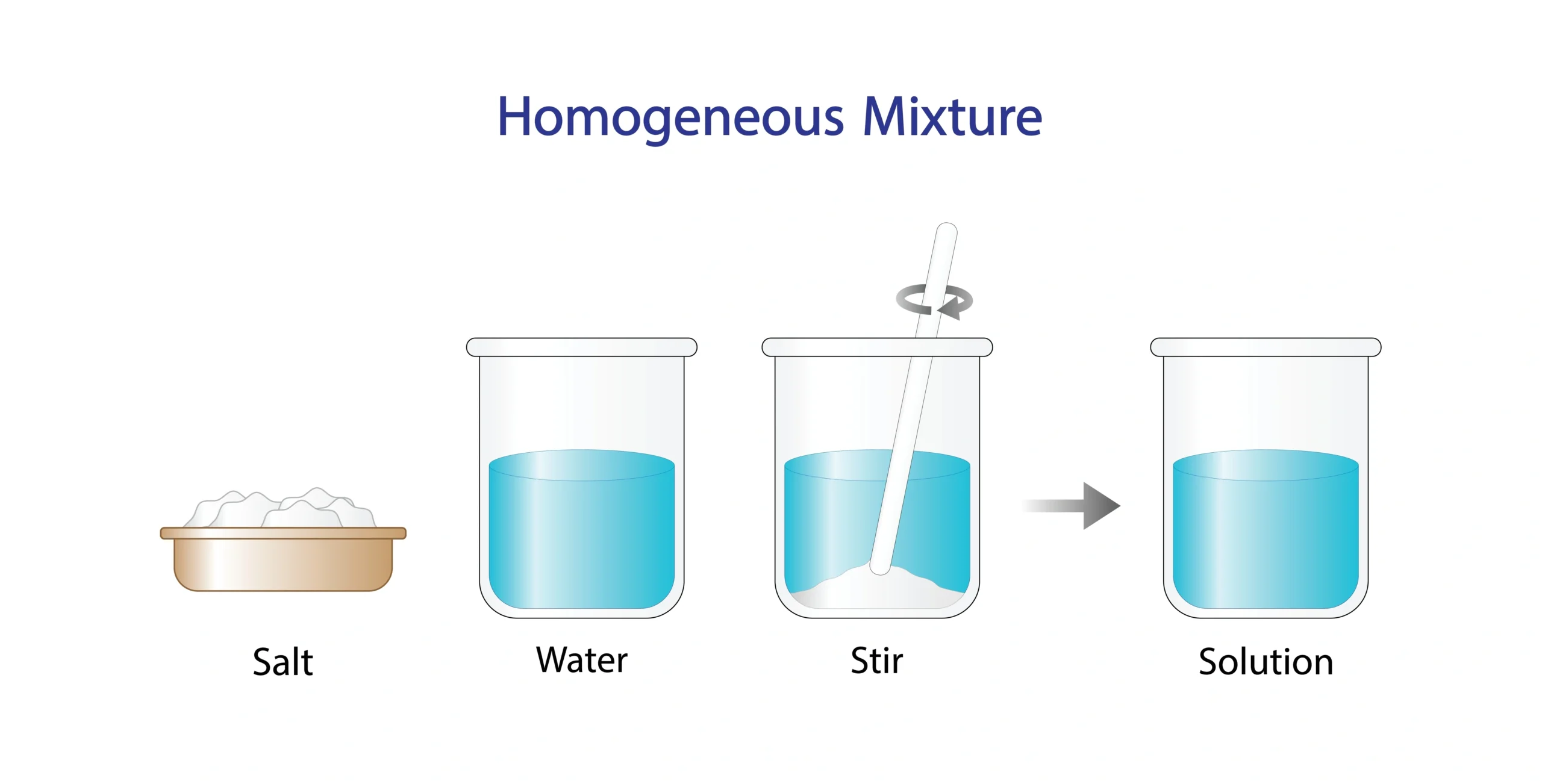
🧭 In a homogeneous mixture, components are evenly mixed.
Solution of sugar in water 🍬➡️💧.
🧠 The mixture looks the same everywhere.
🧭 In a heterogeneous mixture, components are not evenly mixed.
Sand and water 🏖️💧.
🧠 Different parts can be seen clearly.
🧪 Solutions are special types of mixtures.
Solute 🧂
Solvent 💧
🧠 The solute dissolves in the solvent.
🧭 Solubility depends on temperature 🌡️ and nature of substances.
🧠 Heating often increases solubility.
🧭 Matter around us constantly changes form.
Elements combine to form compounds ⚗️.
Compounds mix to form mixtures 🧫.
🧠 These changes explain many natural and industrial processes.
🌍 Understanding elements, compounds, and mixtures helps in daily life.
Cooking 🍳
Cleaning 🧼
Medicine 💊
Industry 🏭
🧠 Chemistry improves quality of life.
🧭 Separation of mixtures is very useful.
Filtration 🧻
Evaporation 🌬️
Sedimentation ⬇️
🧠 These methods help obtain useful substances.
🧠 Knowing the nature of matter builds scientific thinking.
Classification 🧩
Observation 👀
Reasoning 🧠
🧠 It prepares students for advanced chemistry.
📜 Matter may look simple, but its forms are diverse.
Elements, compounds, and mixtures together form the material world.
🚀 Learning their nature helps understand science deeply.
📝 Summary of the Lesson
Matter exists mainly as elements, compounds, and mixtures. Elements are pure substances made of only one type of particle and cannot be broken down easily. Compounds are formed when elements combine chemically in fixed proportions and have properties different from their elements. Mixtures are formed by physical mixing of substances, and their components retain their individual properties. Mixtures can be homogeneous or heterogeneous and can be separated by physical methods. Understanding these forms of matter helps explain everyday materials and chemical processes.
⚡ Quick Recap
⭐ Elements are pure substances
⭐ Compounds form by chemical combination
⭐ Mixtures form by physical mixing
⭐ Elements have unique properties
⭐ Compounds have fixed composition
⭐ Mixtures can be separated
⭐ Matter exists in different forms
——————————————————————————————————————————————————————————————————————————–
TEXTBOOK QUESTIONS
🔒 ❓ Question 1
Consider the following reaction where two substances, A and B, combine to form a product C:
A + B → C
Assume that A and B cannot be broken down into simpler substances by chemical reactions. Which of the following statements is correct?
📌 Answer:
(iv) A and B are elements, C is a compound, and has a fixed composition.
🔵 Substances that cannot be broken down by chemical reactions are called elements.
🔴 When two or more elements chemically combine, they form a compound.
🟣 A compound always has a fixed composition because elements combine in a definite ratio.
🟢 Therefore, A and B must be elements, and C must be a compound with fixed composition.
🔒 ❓ Question 2
Assertion: Air is a mixture.
Reason: A mixture is formed when two or more substances are mixed, without undergoing any chemical change.
📌 Answer:
(i) Both Assertion and Reason are true, and Reason is the correct explanation for Assertion.
🔵 Air contains gases like nitrogen, oxygen, carbon dioxide, and water vapour mixed together.
🔴 These gases are not chemically combined and retain their individual properties.
🟣 This matches the definition of a mixture, where no chemical change occurs.
🟢 Hence, the reason correctly explains why air is a mixture.
🔒 ❓ Question 3
Water, a compound, has different properties compared to those of the elements oxygen and hydrogen from which it is formed. Justify this statement.
📌 Answer:
🔵 Hydrogen is a combustible gas and oxygen supports burning, but water extinguishes fire.
🔴 Hydrogen and oxygen are gases, whereas water is a liquid at room temperature.
🟣 Water has properties entirely different from its constituent elements.
🟢 This happens because chemical combination produces a new substance with new properties.
🔒 ❓ Question 4
In which of the following cases are all the examples correctly matched? Give reasons.
(i) Elements — water, nitrogen, iron, air
(ii) Uniform mixtures — minerals, seawater, bronze, air
(iii) Pure substances — carbon dioxide, iron, oxygen, sugar
(iv) Non-uniform mixtures — air, sand, brass, muddy water
📌 Answer:
(iii) Pure substances — carbon dioxide, iron, oxygen, sugar
🔵 Carbon dioxide and sugar are compounds with fixed composition.
🔴 Iron and oxygen are elements.
🟣 All listed substances have uniform composition throughout.
🟢 Other options include incorrect classifications like air (a mixture) or brass (an alloy).
🔒 ❓ Question 5
Iron reacts with moist air to form iron oxide, and magnesium burns in oxygen to form magnesium oxide. Classify all the substances involved as elements, compounds, or mixtures, with justification.
📌 Answer:
🔵 Iron – Element (cannot be broken into simpler substances)
🔴 Moist air – Mixture (air + water vapour)
🟣 Iron oxide – Compound (formed by chemical combination of iron and oxygen)
🟢 Magnesium – Element
🟡 Oxygen – Element
🔵 Magnesium oxide – Compound (new substance with fixed composition)
🔒 ❓ Question 6
Classify the following as elements, compounds, or mixtures:
Carbon dioxide, sand, seawater, magnesium oxide, muddy water, aluminium, gold, oxygen, rust, iron sulfide, glucose, air, water, fruit juice, nitrogen, sodium chloride, sulfur, hydrogen, baking soda.
📌 Answer:
🔵 Elements: aluminium, gold, oxygen, nitrogen, sulfur, hydrogen
🔴 Compounds: carbon dioxide, magnesium oxide, rust, iron sulfide, glucose, water, sodium chloride, baking soda
🟣 Mixtures: sand, seawater, muddy water, air, fruit juice
🟢 Pure substances: all elements and compounds listed above
🔒 ❓ Question 7
What new substance is formed when a mixture of iron filings and sulfur powder is heated? How is it different from the original mixture? Also, write the word equation.
📌 Answer:
🔵 The new substance formed is iron sulfide.
🔴 Iron sulfide is a compound with properties different from iron and sulfur.
🟣 It cannot be separated by physical methods like a magnet.
🟢 The original mixture could be separated physically.
Word equation:
Iron + Sulfur → Iron sulfide
🔒 ❓ Question 8
Is it possible for a substance to be classified as both an element and a compound? Explain why or why not.
📌 Answer:
🔵 No substance can be both an element and a compound.
🔴 An element contains only one kind of atom.
🟣 A compound contains two or more elements chemically combined.
🟢 Their definitions are fundamentally different.
🔒 ❓ Question 9
How would our daily lives be changed if water were not a compound but a mixture of hydrogen and oxygen?
📌 Answer:
🔵 The composition of water would not be fixed.
🔴 Water might show properties of hydrogen and oxygen, making it dangerous.
🟣 It would not be stable or safe for drinking and daily use.
🟢 Life as we know it would not be possible.
🔒 ❓ Question 10
Analyse Fig. 8.24. Identify Gas A. Also, write the word equation of the chemical reaction.
📌 Answer:
🔵 Gas A is hydrogen gas.
🔴 Hydrogen is released when iron reacts with dilute hydrochloric acid.
🟣 This is a displacement reaction.
Word equation:
Iron + Hydrochloric acid → Iron chloride + Hydrogen
🔒 ❓ Question 11
Write the names of any two compounds made only from non-metals, and also mention two uses of each of them.
📌 Answer:
🔵 Carbon dioxide
🟢 Used in fire extinguishers
🟣 Used in carbonated drinks
🔴 Water
🟡 Used for drinking
🔵 Used for washing and cooking
🔒 ❓ Question 12
How can gold be classified as both a mineral and a metal?
📌 Answer:
🔵 Gold occurs naturally in the Earth’s crust, so it is a mineral.
🔴 Gold shows properties like malleability, ductility, and conductivity.
🟣 These properties classify it as a metal.
🟢 Hence, gold can be both a mineral and a metal.
——————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS
SECTION 1 — MCQs (5 Questions)
🔒 ❓ Q1. Which substance contains only one kind of atom?
🟢 1️⃣ Compound
🔵 2️⃣ Mixture
🟡 3️⃣ Element
🟣 4️⃣ Solution
✔️ Answer: 🟡 3️⃣ Element
🔒 ❓ Q2. Which of the following is a compound?
🟢 1️⃣ Air
🔵 2️⃣ Salt
🟡 3️⃣ Soil
🟣 4️⃣ Brass
✔️ Answer: 🔵 2️⃣ Salt
🔒 ❓ Q3. Which property helps separate iron from sand?
🟢 1️⃣ Solubility
🔵 2️⃣ Magnetism
🟡 3️⃣ Density
🟣 4️⃣ Colour
✔️ Answer: 🔵 2️⃣ Magnetism
🔒 ❓ Q4. In a mixture, the components are:
🟢 1️⃣ Chemically combined
🔵 2️⃣ Fixed in ratio
🟡 3️⃣ Physically mixed
🟣 4️⃣ Always solids
✔️ Answer: 🟡 3️⃣ Physically mixed
🔒 ❓ Q5. Which method is used to separate salt from salt solution?
🟢 1️⃣ Filtration
🔵 2️⃣ Evaporation
🟡 3️⃣ Sedimentation
🟣 4️⃣ Decantation
✔️ Answer: 🔵 2️⃣ Evaporation
SECTION 2 — Very Short Answer (5 Questions)
🔒 ❓ Q6. Name a pure substance.
📌 ✅ Answer: Element
🔒 ❓ Q7. What is a mixture of metals called?
📌 ✅ Answer: Alloy
🔒 ❓ Q8. Name one homogeneous mixture.
📌 ✅ Answer: Air
🔒 ❓ Q9. Which substance is formed by chemical combination?
📌 ✅ Answer: Compound
🔒 ❓ Q10. Name the process of separating insoluble solids from liquids.
📌 ✅ Answer: Filtration
SECTION 3 — Short Answer (3 Questions)
🔒 ❓ Q11. What is an element?
📌 ✅ Answer:
🔹 An element is a pure substance made of one kind of atom.
🔸 It cannot be broken into simpler substances by chemical methods.
🔹 Elements form the basic building blocks of matter.
🔒 ❓ Q12. How is a compound different from a mixture?
📌 ✅ Answer:
🔹 In a compound, elements combine chemically in fixed ratio.
🔸 In a mixture, substances mix physically in any ratio.
🔹 Components of mixtures can be separated by physical methods.
🔒 ❓ Q13. Why are mixtures easy to separate?
📌 ✅ Answer:
🔹 Components of mixtures retain their properties.
🔸 They are not chemically combined.
🔹 Simple physical methods can separate them.
SECTION 4 — Long Answer (1 Question)
🔒 ❓ Q14. Explain elements, compounds and mixtures with examples.
📌 ✅ Answer:
🔹 Elements are pure substances made of one type of atom, like iron.
🔸 Compounds are formed by chemical combination of elements, like water.
🔹 Mixtures are physical combinations of substances, like air.
🔸 Mixtures do not have fixed composition.
🔹 Understanding these helps classify matter correctly.
——————————————————————————————————————————————————————————————————————————–
ADVANCE KNOWLEDGE
🌍 From One World to Many Substances
Look around and everything seems different—air, water, soil, steel, sugar, medicines. Yet, all of them are made from a small set of basic building blocks. Science discovered that matter organizes itself in three powerful ways: elements, compounds, and mixtures. This idea transformed chemistry from guesswork into a logical science of matter.
🧠 Big idea:
Complex matter is created by simple units combining in different ways.
⚛️ Elements: The Pure Foundations of Matter
An element is a substance made of only one kind of atom.
🧠 Key truth:
Elements cannot be broken into simpler substances by chemical means
⭐ Every element has a unique atomic identity.
🧪 Examples:
Oxygen
Iron
Carbon
Gold
🧠 Atoms of different elements differ in structure and behaviour—this is why substances act differently.
⏳ A Short History of Elements
⏳ Ancient thinkers believed:
Matter was made of earth, water, fire, and air
🧠 Modern science revealed:
Over 100 distinct elements exist
⭐ This discovery reshaped medicine, industry, and technology.
🧠 The periodic table is not a chart—it is a map of matter.
⚠️ Misconception vs Reality
⚠️ Misconception: Elements are always simple and useless
✅ Reality: Elements form the backbone of all materials
⚠️ Misconception: Elements exist only in laboratories
✅ Reality: Elements exist naturally in air, water, soil, and living bodies
🧬 Compounds: When Elements Unite
A compound forms when two or more elements combine chemically in a fixed ratio.
🧠 In compounds:
Atoms bond strongly
New properties appear
⭐ The compound behaves completely differently from its elements.
🧪 Example:
Hydrogen + Oxygen → Water
🧠 Hydrogen burns, oxygen supports burning—but water puts out fire.
🔗 Chemical Bonds: Invisible Glue
Compounds exist because of chemical bonds.
🧠 Bonds form when:
Atoms share or transfer electrons
⭐ Bonding lowers energy and increases stability.
🧠 Bond strength controls:
Hardness
Melting point
Reactivity
🧪 Properties: More Than the Sum of Parts
Compounds show emergent properties.
🧠 This means:
New characteristics appear
Old properties disappear
⭐ Salt tastes salty, not like sodium metal or chlorine gas.
🧠 Nature creates surprises through bonding.
⚗️ Mixtures: Freedom Without Bonding
A mixture is a physical combination of substances.
🧠 In mixtures:
No chemical bonds form
Components keep their properties
Proportions can vary
⭐ Mixing does not create a new substance—it creates coexistence.
🧠 Types of Mixtures
Mixtures exist in many forms.
🧠 Homogeneous mixtures:
Uniform composition
Example: salt solution
🧠 Heterogeneous mixtures:
Non-uniform
Example: sand and iron filings
⭐ Nature prefers mixtures—air, soil, seawater are all mixtures.
⚠️ Misconception vs Reality
⚠️ Misconception: Mixtures are less important than compounds
✅ Reality: Life and environment depend mainly on mixtures
⚠️ Misconception: Mixtures cannot be separated
✅ Reality: Mixtures can be separated by physical methods
🧪 Separation: Reading Matter’s Structure
Separation techniques reveal matter’s nature.
🧠 Examples:
Filtration
Evaporation
Distillation
Chromatography
⭐ Separation methods prove whether matter is a compound or a mixture.
🌍 Elements, Compounds, and Life
Living organisms are chemical systems.
🧠 Elements in the body:
Carbon
Hydrogen
Oxygen
Nitrogen
🧠 Compounds in the body:
Water
Proteins
Sugars
🧠 Mixtures in the body:
Blood
Cytoplasm
⭐ Life is organized chemistry.
🌬️ Air and Water: Everyday Chemistry
Air is not an element.
🧠 Air is a mixture of:
Nitrogen
Oxygen
Carbon dioxide
🧠 Water is not a mixture—it is a compound.
⭐ This distinction explains why:
Air components can be separated
Water cannot be separated without chemical change
🚀 Modern Science: Advanced Material Design
🚀 Scientists design materials by controlling combinations.
🧠 Examples:
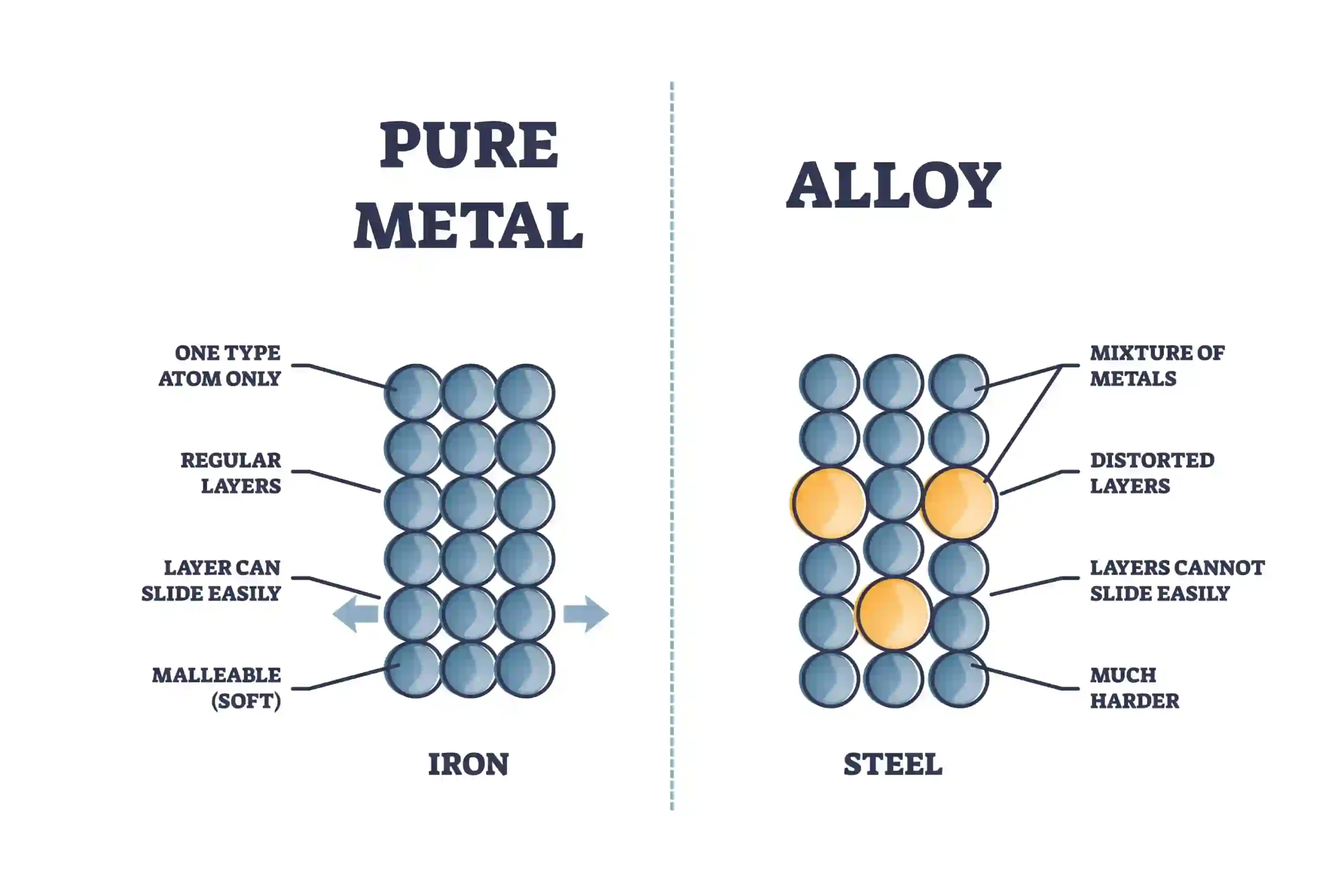
Alloys (metal mixtures)
Polymers (giant compounds)
Nanocomposites
⭐ Material science is applied chemistry.
🌍 Environmental Perspective
Understanding matter helps protect Earth.
🧠 Pollution involves:
Harmful mixtures
Toxic compounds
⭐ Cleaning pollution requires knowing what type of matter is present.
🧠 Matter Beyond Earth
Different planets have different compositions.
🌌 Examples:
Hydrogen-rich stars
Iron-rich planets
🧠 Studying composition helps:
Understand planet formation
Search for life elsewhere
⭐ Amazing Facts
⭐ Gold is an element that does not rust
⭐ Most medicines are carefully designed compounds
⭐ Seawater contains dozens of dissolved elements
⭐ Separation techniques built modern industries
🧠 Why Curious Minds Must Understand Matter Types
This topic trains:
Classification thinking
Logical separation
Microscopic reasoning
🧠 It forms the base of chemistry, biology, and material science.
🌟 Final Thought
Elements are nature’s letters.
Compounds are nature’s words.
Mixtures are nature’s conversations.
🧠 To understand matter is to understand how simplicity creates infinite complexity.
——————————————————————————————————————————————————————————————————————————–
