Class 8 : Science – ( English ) : Lesson 7. Particulate Nature of Matter
EXPLANATION AND ANALYSIS
🧭 Everything around us is made of matter.
Air 🌬️, water 💧, soil 🌱, food 🍎, and even our own body 🫀 are forms of matter. Matter occupies space and has mass, but its structure is not continuous—it is made up of tiny particles.
🧠 These particles are so small that we cannot see them with our naked eyes.
Yet, their behavior explains many everyday observations.
🔍 Studying the particulate nature of matter helps us understand how substances behave, change, and interact.
🧪 Matter is made up of extremely small particles called particles of matter 🧬.
These particles are present in solids 🧱, liquids 💧, and gases 🌬️.
🧠 Even a tiny grain of sugar 🍬 contains millions of particles.
🧭 One important property of particles is that they have spaces between them.
The amount of space differs in different states of matter.
🧠 In solids, particles are closely packed.
In liquids, particles have more space.
In gases, particles are far apart.
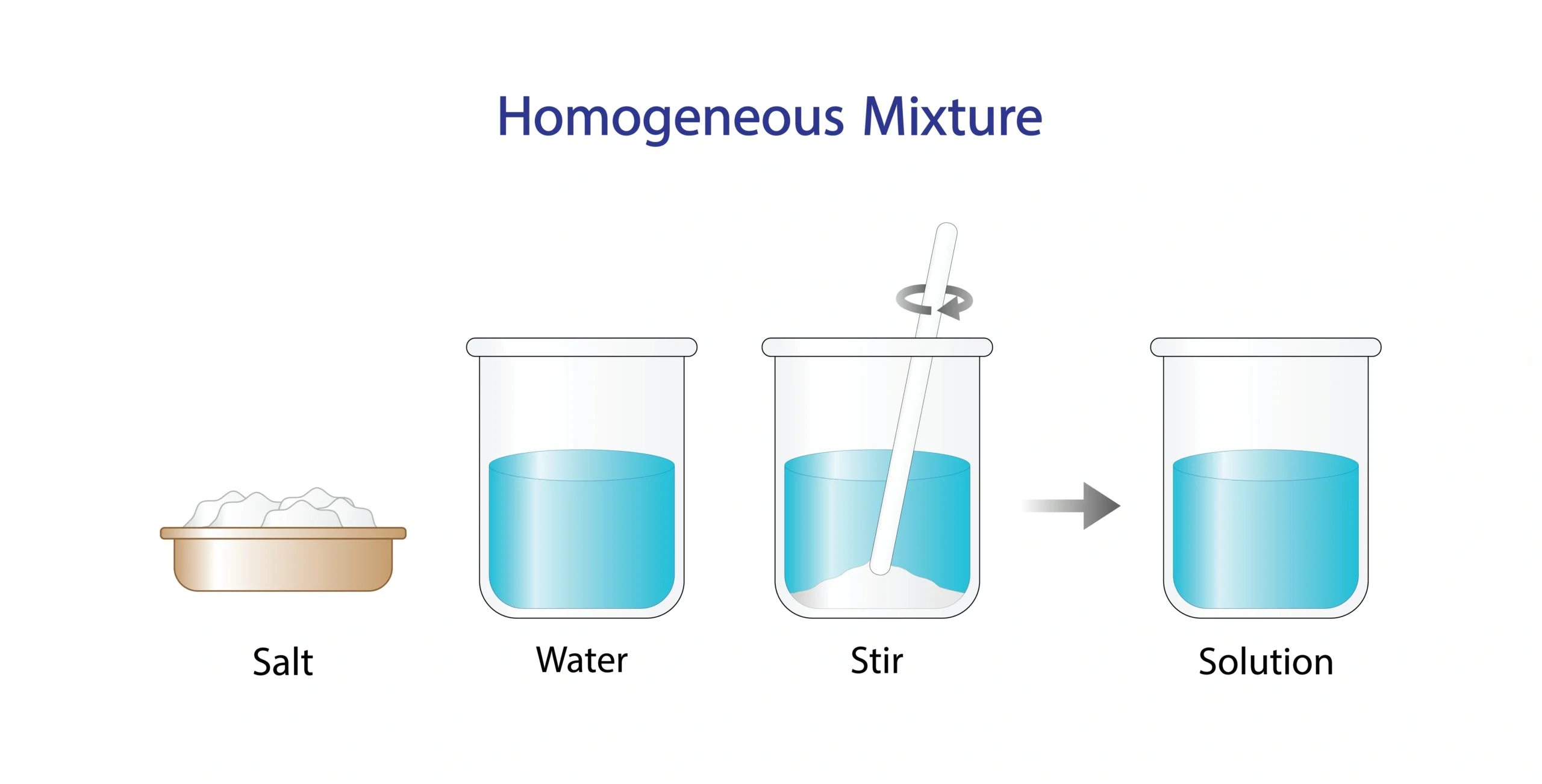
🧪 The presence of space between particles explains why substances can mix.
Salt dissolves in water 🧂➡️💧.
Sugar mixes uniformly in tea 🍵.
🧠 Dissolving shows particles moving into the spaces between water particles.
🧭 Another important property is that particles of matter are in constant motion 🔄.
They keep moving all the time.
🧠 This motion increases when temperature rises 🌡️.
🧪 The continuous movement of particles can be observed through diffusion.
Diffusion is the mixing of particles on their own.
🌸 Smell of perfume spreads in air.
🖊️ Ink spreads in water.
🧠 Diffusion proves that particles move constantly.
🧭 The speed of diffusion differs in solids, liquids, and gases.
Fastest in gases 🌬️
Slower in liquids 💧
Slowest in solids 🧱
🧠 This happens because particle movement differs.
🧪 Particles of matter also attract each other 🤝.
This attraction is called inter-particle force.
🧠 Strong attraction keeps particles tightly packed in solids.
Weak attraction allows particles to move freely in gases.
🧭 Inter-particle force determines the shape and volume of matter.
Solids have fixed shape 📦.
Liquids have fixed volume but no fixed shape 🥤.
Gases have neither fixed shape nor volume 🌫️.
🧪 Heating affects particle behavior.
When heat is supplied 🔥, particles gain energy ⚡.
🧠 They move faster and overcome attraction forces.
🧭 Due to heating, matter can change its state.
Solid ➡️ Liquid ➡️ Gas
🧠 Cooling reverses this process ❄️.
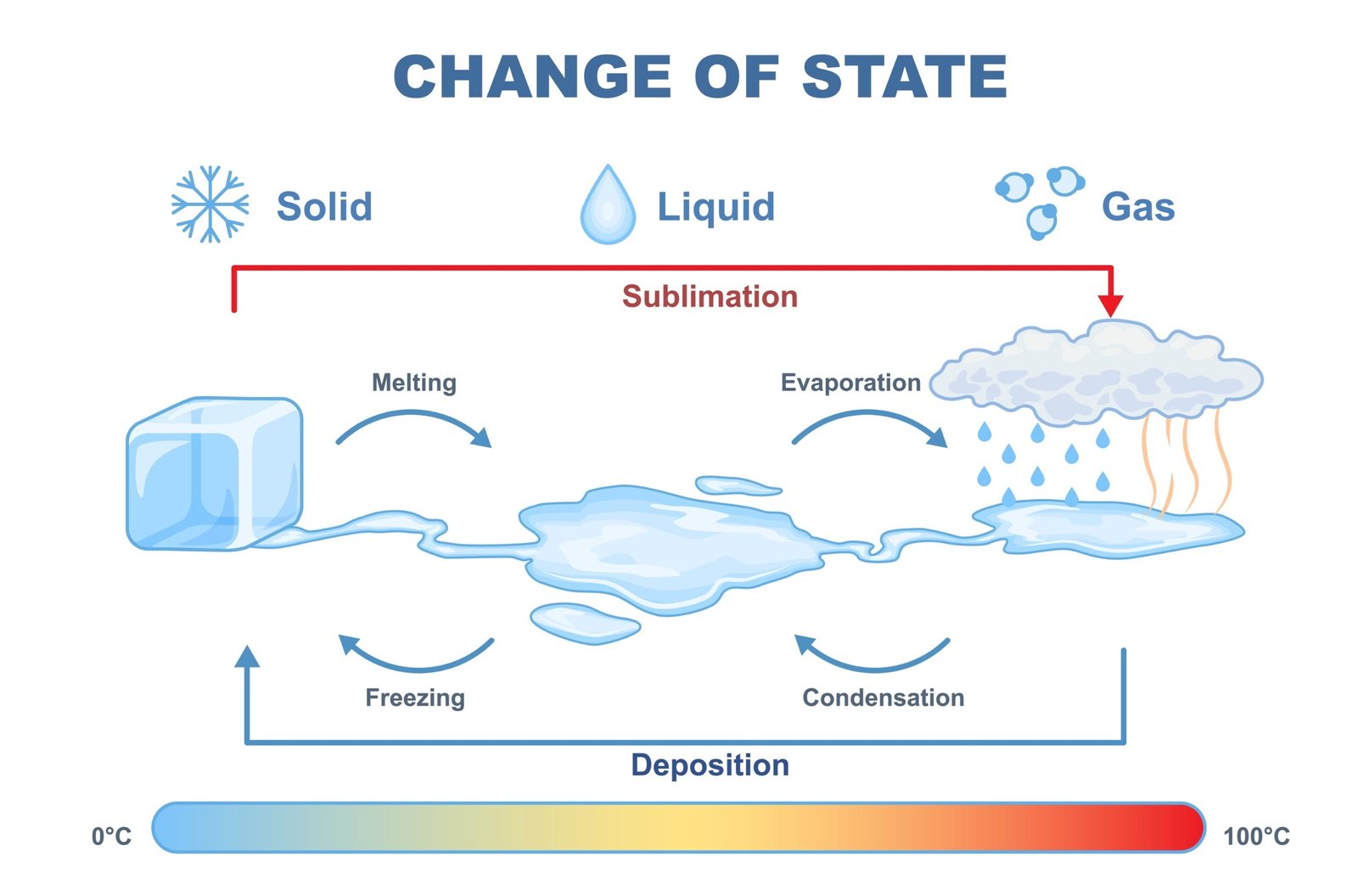
🧪 Melting of ice ❄️➡️💧 is caused by increased particle motion.
Freezing of water 💧➡️❄️ occurs when particles lose energy.
🧠 State change is a physical change.
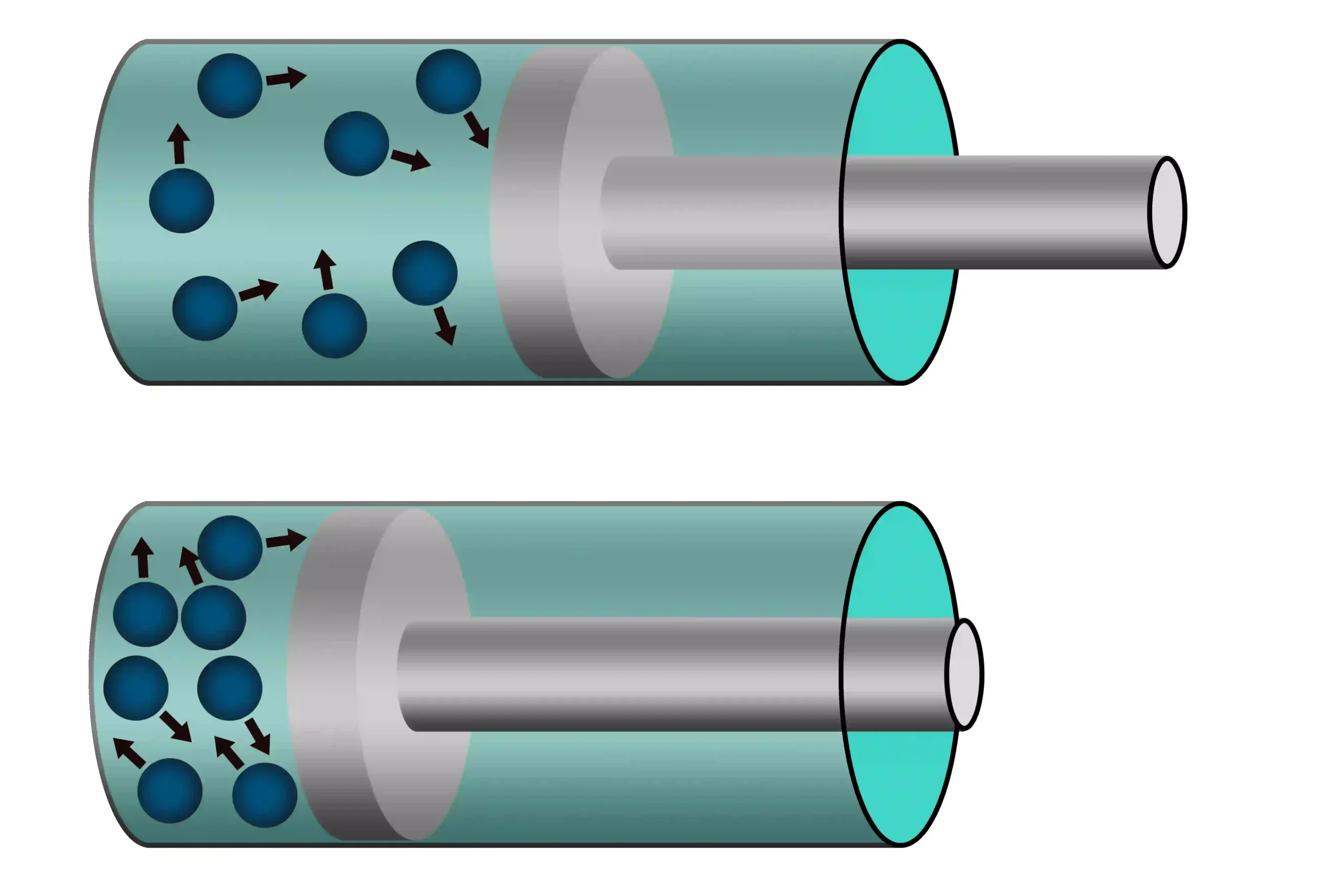
🧭 The particulate nature of matter explains compressibility.
Gases can be compressed easily 🧪.
Liquids are less compressible.
Solids are almost incompressible.
🧠 This is because of space between particles.
🧭 Matter shows expansion on heating.
Railway tracks 🚆 expand in summer.
Electric wires 🔌 sag when heated.
🧠 Expansion happens due to increased particle movement.
🧠 Understanding particles helps explain evaporation.
Some surface particles escape into air 🌬️.
🧠 Evaporation causes cooling ❄️.
🌍 The particulate nature of matter explains natural and daily phenomena.
Weather 🌦️
Cooking 🍳
Drying clothes 👕
Smell spreading 🌸
🧠 Microscopic particles control macroscopic behavior.
🧭 Studying particles builds scientific thinking.
Observation 👀
Reasoning 🧠
Cause–effect understanding 🧩
🧠 It forms the base of chemistry and physics.
📜 Matter may appear continuous, but it is made of tiny moving particles.
This idea changed scientific understanding.
🚀 Learning about particles prepares students for advanced science.
📝 Summary of the Lesson
Matter is made up of extremely small particles that are in constant motion and have spaces between them. These particles attract each other with varying strength. The arrangement, movement, and attraction of particles determine whether matter exists as a solid, liquid, or gas. Diffusion shows the movement of particles, and heating increases their motion. Changes of state such as melting and freezing occur due to changes in particle energy. The particulate nature of matter explains mixing, expansion, compressibility, evaporation, and many everyday phenomena.
⚡ Quick Recap
⭐ Matter is made of tiny particles
⭐ Particles have spaces between them
⭐ Particles are always moving
⭐ Diffusion proves particle motion
⭐ Heating increases particle speed
⭐ Attraction differs in states of matter
⭐ Particle behavior explains changes
——————————————————————————————————————————————————————————————————————————–
TEXTBOOK QUESTIONS
🔒 ❓ Question 1. Choose the correct option
The primary difference between solids and liquids is that the constituent particles are:
📌 Answer:
🔵 (iv) closely packed in solids and move past each other in liquids
🟢 In solids, particles are very closely packed and can only vibrate about fixed positions.
🟣 In liquids, particles are still close but have enough freedom to slide past one another.
🟡 This ability to move past each other allows liquids to flow, unlike solids.
🔒 ❓ Question 2. Which of the following statements are true? Correct the false statements.
📌 Answer:
🔵 (i) True
Melting ice into water is a clear example of a solid changing into a liquid state.
🟢 (ii) True
During melting, interparticle attractions decrease, allowing particles to move more freely.
🟣 (iii) True
Solids have a fixed shape and a fixed volume due to strong interparticle forces.
🟡 (iv) True
Particles in solids have very strong interactions and extremely small interparticle spaces.
🔒 ❓ Question 3. Choose the correct answer with justification.
If we could remove all the constituent particles from a chair, what would happen?
📌 Answer:
🔵 (iii) Nothing of the chair will remain.
🟢 Matter is made up of particles.
🟣 Removing all particles means removing mass, shape, and volume.
🟡 Without particles, the chair cannot exist in any form.
🔒 ❓ Question 4. Why do gases mix easily, while solids do not?
📌 Answer:
🔵 Gas particles are far apart and move randomly at high speeds.
🟢 Large interparticle spaces allow gases to diffuse easily.
🟣 Solid particles are tightly packed with strong attractions.
🟡 Therefore, solids do not mix easily like gases.
🔒 ❓ Question 5.
When spilled on the table, milk in a glass tumbler flows and spreads out, but the glass tumbler stays in the same shape. Justify.
📌 Answer:
🔵 Milk is a liquid, so its particles can move freely.
🟢 Liquids have fixed volume but no fixed shape.
🟣 Glass is a solid with strong interparticle forces.
🟡 Hence, glass retains its shape while milk spreads.
🔒 ❓ Question 6. Represent diagrammatically the changes in the arrangement of particles as ice melts and transforms into water vapour.
📌 Answer (Textual Representation):
🔵 Ice (Solid): particles very closely packed in an orderly arrangement
🟢 Water (Liquid): particles slightly apart, less orderly, able to slide
🟣 Water vapour (Gas): particles far apart, random motion, maximum space
🔒 ❓ Question 7. Draw a picture representing particles present in the following:
(i) Aluminium foil
(ii) Glycerin
(iii) Methane gas
📌 Answer (Conceptual Description):
🔵 Aluminium foil (Solid):
Particles tightly packed in a fixed, regular pattern.
🟢 Glycerin (Liquid):
Particles close together but irregularly arranged, able to move past one another.
🟣 Methane gas (Gas):
Particles very far apart, moving randomly in all directions.
🔒 ❓ Question 8.
Observe Fig. 7.16a showing a candle just extinguished. Identify states of wax and match them with Fig. 7.16b.
📌 Answer:
🔵 Solid wax: present in the candle body (closely packed particles).
🟢 Liquid wax: molten wax near the flame (moderately spaced particles).
🟣 Wax vapour: wax fumes after extinguishing (widely spaced particles).
🟡 These match the solid, liquid, and gas arrangements in Fig. 7.16b.
🔒 ❓ Question 9. Why does the water in the ocean taste salty even though the salt is not visible?
📌 Answer:
🔵 Salt dissolves completely in water.
🟢 Salt particles break into very small ions.
🟣 These particles spread uniformly throughout water.
🟡 Though invisible, dissolved salt still affects taste.
🔒 ❓ Question 10.
Grains of rice and rice flour take the shape of the container when placed in different jars. Are they solids or liquids? Explain.
📌 Answer:
🔵 They are solids.
🟢 Each grain has a fixed shape and volume.
🟣 They appear to flow due to spaces between particles.
🟡 This behaviour is due to granularity, not liquidity.
——————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS
SECTION 1 — MCQs (5 Questions)
🔒 ❓ Q1. Which statement best supports the particulate nature of matter?
🟢 1️⃣ Matter occupies space
🔵 2️⃣ Matter is continuous
🟡 3️⃣ Matter is made of tiny particles
🟣 4️⃣ Matter is heavy
✔️ Answer: 🟡 3️⃣ Matter is made of tiny particles
🔒 ❓ Q2. Which property shows that particles of matter have spaces between them?
🟢 1️⃣ Diffusion
🔵 2️⃣ Shape
🟡 3️⃣ Colour
🟣 4️⃣ Density
✔️ Answer: 🟢 1️⃣ Diffusion
🔒 ❓ Q3. Which state of matter has the least attraction between particles?
🟢 1️⃣ Solid
🔵 2️⃣ Liquid
🟡 3️⃣ Gas
🟣 4️⃣ Plasma
✔️ Answer: 🟡 3️⃣ Gas
🔒 ❓ Q4. What happens to particle motion when temperature increases?
🟢 1️⃣ Stops
🔵 2️⃣ Decreases
🟡 3️⃣ Increases
🟣 4️⃣ Remains same
✔️ Answer: 🟡 3️⃣ Increases
🔒 ❓ Q5. Which process shows random movement of particles?
🟢 1️⃣ Evaporation
🔵 2️⃣ Diffusion
🟡 3️⃣ Condensation
🟣 4️⃣ Freezing
✔️ Answer: 🔵 2️⃣ Diffusion
SECTION 2 — Very Short Answer (5 Questions)
🔒 ❓ Q6. What is matter made of?
📌 ✅ Answer: Particles
🔒 ❓ Q7. Name the movement of particles from high to low concentration.
📌 ✅ Answer: Diffusion
🔒 ❓ Q8. Which state of matter is highly compressible?
📌 ✅ Answer: Gas
🔒 ❓ Q9. What happens to particles on heating?
📌 ✅ Answer: Move faster
🔒 ❓ Q10. Name the force between particles.
📌 ✅ Answer: Attraction
SECTION 3 — Short Answer (3 Questions)
🔒 ❓ Q11. How does diffusion prove that matter is made of particles?
📌 ✅ Answer:
🔹 Diffusion shows particles move continuously.
🔸 Particles mix on their own due to spaces between them.
🔹 This behaviour confirms matter consists of tiny moving particles.
🔒 ❓ Q12. Why are gases easily compressible?
📌 ✅ Answer:
🔹 Gas particles have large spaces between them.
🔸 Attractive forces are very weak.
🔹 Hence gases can be compressed easily.
🔒 ❓ Q13. How does temperature affect particle motion?
📌 ✅ Answer:
🔹 Heating increases particle kinetic energy.
🔸 Particles move faster and spread out.
🔹 Cooling slows particle motion.
SECTION 4 — Long Answer (1 Question)
🔒 ❓ Q14. Explain the main characteristics of particles of matter.
📌 ✅ Answer:
🔹 Particles of matter are extremely small and have spaces between them.
🔸 They are in constant motion and show diffusion.
🔹 Particles attract each other with varying strength.
🔸 Increase in temperature increases particle motion.
🔹 These properties explain different states and behaviour of matter.
——————————————————————————————————————————————————————————————————————————–
ADVANCE KNOWLEDGE
🌍 Matter: Not Continuous, but Secretly Fragmented
To our eyes, matter looks continuous and solid. A stone seems compact, water seems smooth, air seems empty. But deep inside, matter is not continuous at all. It is made of unimaginably tiny particles, always moving, colliding, and interacting.
🧠 Big idea:
Matter is not still, not solid inside, and not quiet — it is constantly active at the microscopic level.
⚛️ What Does “Particulate” Really Mean?
Particulate nature means:
Matter is made of tiny discrete particles
These particles are separate, not joined as one block
⭐ These particles are so small that millions fit in a pinhead.
🧠 What we call “solid” is actually mostly empty space between particles.
⚠️ Misconception vs Reality
⚠️ Misconception: Matter is continuous like a solid block
✅ Reality: Matter has spaces between particles
⚠️ Misconception: Particles in solids do not move
✅ Reality: Particles in solids vibrate continuously
🧪 Evidence of Particles: Mixing Without Stirring
Some observations prove particle nature.
🧪 Examples:
Smell of perfume spreading in a room
Ink spreading in water
Sugar dissolving completely
🧠 These happen because particles move on their own.
⭐ Diffusion is silent proof that matter is particulate.
🔄 Motion of Particles: Always Active
Particles of matter are never at rest.
🧠 Particle motion depends on:
Temperature
State of matter
⭐ Higher temperature → faster particle motion.
🧠 This is why heating causes expansion.
🌡️ Temperature: Speed Controller of Particles
Temperature controls how fast particles move.
🧠 Effects of heating:
Solids expand
Liquids flow faster
Gases spread rapidly
⭐ Cooling slows particles and brings them closer.
🧠 Temperature is a measure of particle motion, not heat quantity.
🧱 States of Matter Through Particles
Different states are different arrangements of particles.
🧱 Solids
Particles tightly packed
Strong attraction
Only vibration
💧 Liquids
Particles close but free to slide
Moderate attraction
🌫️ Gases
Particles far apart
Weak attraction
Random motion
⭐ Same particles — different arrangements.
🧠 Inter-Particle Space and Attraction
Two invisible factors decide behaviour.
🧠 Inter-particle space:
Small in solids
Larger in liquids
Largest in gases
🧠 Inter-particle attraction:
Strong in solids
Weak in gases
⭐ Balance between space and attraction defines state.
🔥 Change of State: Rearranging Particles
Melting, boiling, freezing are particle rearrangements.
🧠 Heating:
Increases particle energy
Breaks attraction
🧠 Cooling:
Reduces energy
Strengthens attraction
⭐ No particles are destroyed — only rearranged.
🌍 Diffusion: Matter Mixing Itself
Diffusion is movement of particles from high concentration to low concentration.
🧠 Fastest in gases
🧠 Slowest in solids
⭐ Diffusion explains:
Smell spreading
Gas mixing
Oxygen reaching lungs
🧠 Life depends on diffusion.
🌬️ Brownian Motion: Random Dance
Particles show zig-zag random motion.
⏳ Discovered by:
Robert Brown
🧠 This proved:
Particles are constantly moving
Motion is real, not imagined
⭐ Brownian motion is visible proof of invisible particles.
🧠 Matter at the Atomic Scale
Particles themselves are made of atoms.
🧠 Atoms:
Have nucleus and electrons
Mostly empty space
⭐ Touching objects never truly touch at atomic level.
🧠 All solidity is due to electrical forces, not contact.
🚀 Modern Science: Beyond Simple Particles
🚀 Scientists now study:
Nanoparticles
Quantum behaviour
Atomic manipulation
🧠 Particle control allows:
New materials
Medical treatments
Faster electronics
⭐ Nanotechnology works because matter is particulate.
🌍 Particulate Matter and Pollution
Tiny particles in air affect health.
⚠️ PM2.5 and PM10:
Enter lungs
Damage cells
🧠 Understanding particle size helps fight pollution.
🌌 Particles Beyond Earth
Matter behaves differently in space.
🌌 Microgravity:
Alters particle arrangement
Changes diffusion
⭐ Space experiments confirm particle theory universally.
⭐ Amazing Facts
⭐ A drop of water contains trillions of particles
⭐ Gases diffuse thousands of times faster than solids
⭐ Atoms are mostly empty space
⭐ Heating increases particle chaos
🧠 Why Curious Minds Must Understand Particles
This topic teaches:
Scientific imagination
Microscopic reasoning
Connection between invisible and visible
🧠 It trains the mind to think beyond appearances.
🌟 Final Thought
Matter may look calm and solid,
but inside it is alive with motion.
🧠 To understand the particulate nature of matter is to see the hidden universe beneath everyday reality.
——————————————————————————————————————————————————————————————————————————–
