Class 7 : Science – ( English ) : Lesson 2. Exploring Substances: Acidic, Basic, and Neutral
EXPLANATION AND ANALYSIS
🧭 Substances around us show different behaviors when they come in contact with water, food, skin, or other materials.
Some substances taste sour 🍋, some taste bitter 🥬, while others feel soapy 🧼. These differences help us classify substances into acidic, basic, and neutral categories.
🧠 Understanding the nature of substances is important because it explains many everyday experiences.
Why does lemon juice taste sour?
Why does soap feel slippery?
Why is pure water tasteless?
🔍 These questions lead us to the study of acids, bases, and neutral substances.
🧪 Acids are substances that usually taste sour and can react with many materials.
Examples of acidic substances are lemon juice 🍋, vinegar 🧴, curd 🥛, and tamarind.
🧠 Acids are commonly found in food and natural sources.
Citrus fruits contain citric acid 🍊.
Curd contains lactic acid 🥣.
Vinegar contains acetic acid ⚗️.
⚠️ Acids should not be tasted directly in laboratories.
Some acids are very strong and harmful 🧪☠️.
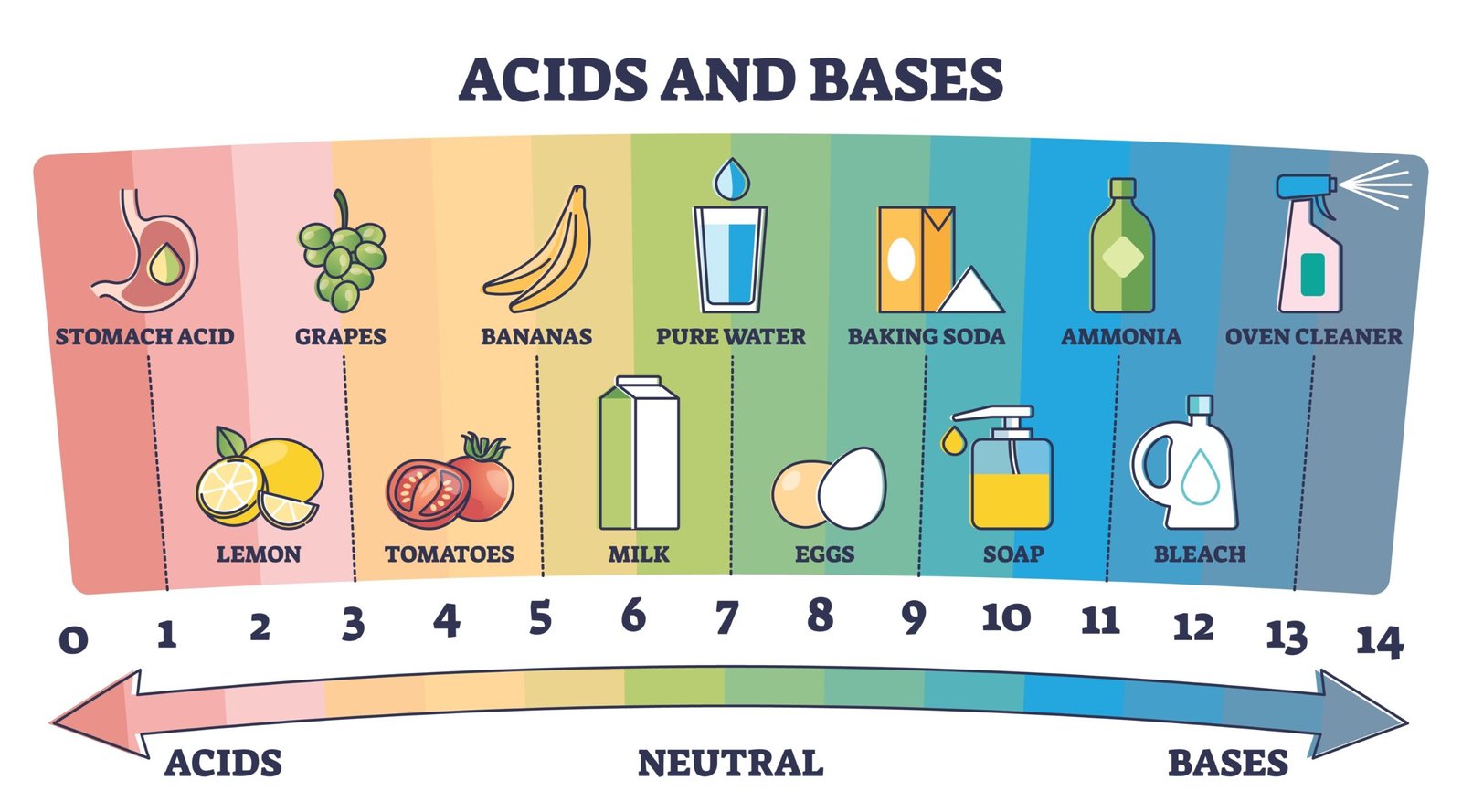
🧭 Acids show special properties when tested.
They turn blue litmus paper red 🔵➡️🔴.
They react with metals to produce gas 💨.
They react with bases to form salt and water ⚗️.
🧠 These properties help scientists identify acidic substances safely using indicators.

🧪 Bases are substances that usually taste bitter and feel slippery or soapy.
Common examples of bases include soap 🧼, baking soda 🥄, lime water 🧪, and washing powder 🧺.
🧠 Bases are widely used in cleaning and daily life.
Soap removes grease 🧽.
Toothpaste cleans teeth 🦷.
Baking soda helps in cooking 🍰.
⚠️ Like acids, strong bases can also be harmful if touched directly.
🧭 Bases have their own identifying properties.
They turn red litmus paper blue 🔴➡️🔵.
They neutralize acids ⚖️.
They feel slippery due to their chemical nature.
🧠 These properties help us distinguish bases from acids.
🧪 Neutral substances are those that are neither acidic nor basic.
Pure water 💧 is the most common neutral substance.
🧠 Neutral substances do not change the color of litmus paper.
They usually have no sharp taste.
They are often safe and gentle.
🌍 Many substances around us are neutral and help balance acidic and basic effects.
🧭 To test whether a substance is acidic, basic, or neutral, indicators are used.
Indicators change color depending on the nature of the substance 🔍.
🧪 Litmus is a natural indicator obtained from lichens 🌿.
It is available as red and blue litmus paper.
🧠 Litmus helps in simple and quick identification of substances.
🧪 Other natural indicators are also used.
Turmeric 🟡
China rose 🌺
Red cabbage 🥬
🧠 These indicators change color differently in acids and bases, making learning more interesting and safe.
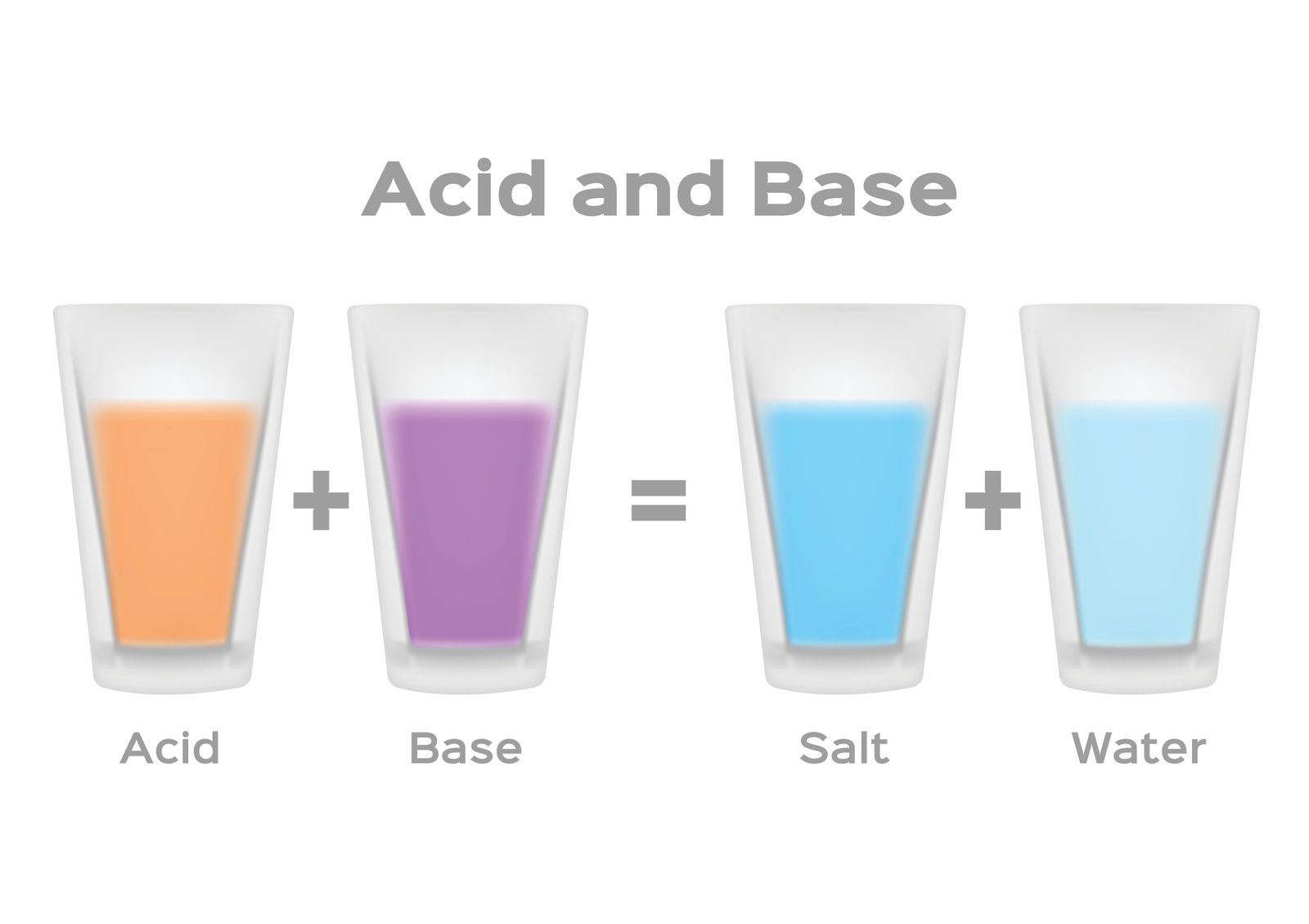
🧭 Neutralization is an important chemical process.
When an acid reacts with a base, both lose their strong nature ⚖️.
🧪 Acid + Base → Salt + Water
This reaction helps reduce harmful effects.
🧠 Neutralization is used in daily life and nature.
🌍 In the human body 🫀, excess acid in the stomach causes indigestion.
Antacids 💊, which are basic, neutralize excess acid and give relief.
🐝 When an ant or bee stings, it injects acid into the skin.
Applying a mild base like baking soda 🧂 helps reduce pain.
🌱 Soil treatment in farming also uses neutralization.
Acidic soil is treated with quicklime 🧪.
Basic soil is treated with organic matter 🍂.

🧭 Acids, bases, and neutral substances play a major role in science and daily life.
They help in digestion, cleaning, agriculture, medicine, and industry ⚙️.
🧠 Understanding their properties helps us handle substances safely and wisely.
🧭 Learning about substances builds scientific thinking.
It teaches observation 👀.
It teaches comparison 🧩.
It teaches safe experimentation 🔬.
🧠 This knowledge connects chemistry to everyday experiences.
📝 Summary of the Lesson
Substances around us can be classified as acidic, basic, or neutral based on their properties. Acids taste sour and turn blue litmus paper red, while bases taste bitter, feel slippery, and turn red litmus paper blue. Neutral substances, such as pure water, do not show acidic or basic behavior. Indicators like litmus, turmeric, and china rose help identify the nature of substances safely. Neutralization occurs when an acid and a base react to form salt and water, reducing harmful effects. This process is useful in daily life, such as treating indigestion, relieving insect stings, and improving soil quality. Understanding acids, bases, and neutral substances helps us use materials safely and understand chemical changes around us.
⚡ Quick Recap
⭐ Substances can be acidic, basic, or neutral
⭐ Acids taste sour and change blue litmus to red
⭐ Bases taste bitter and feel slippery
⭐ Neutral substances show no change
⭐ Indicators help identify substances
⭐ Neutralization reduces harmful effects
⭐ Chemistry is linked to daily life
——————————————————————————————————————————————————————————————————————————–
TEXTBOOK QUESTIONS
🔒 ❓ Question 1.
A solution turns the red litmus paper blue. Excess addition of which of the following solution would reverse the change?
(i) Lime water
(ii) Baking soda
(iii) Vinegar
(iv) Common salt solution
📌 Answer:
The solution has turned red litmus blue, so it is basic in nature. To reverse this change, an acidic solution must be added.
✔️ Correct option: (iii) Vinegar
Vinegar is acidic and neutralises the base, bringing the solution back towards neutral and reversing the colour change.
🔒 ❓ Question 2.
Three unknown solutions A, B and C are tested using indicators. Based on the observations, identify their nature.
📌 Answer:
Solution A turns red litmus blue → Basic
Solution B turns turmeric red → Basic
Solution C turns red rose extract green → Basic
✔️ Correct sequence: (iv) Basic, basic and basic
🔒 ❓ Question 3.
Observe the figures showing red rose extract paper strips. Label the nature of solutions in each container.
📌 Answer:
If the strip turns green, the solution is basic.
If there is no change, the solution is neutral.
If the strip turns dark pink/red, the solution is acidic.
Thus, each container can be labelled based on the colour change observed using the red rose indicator.
🔒 ❓ Question 4.
A liquid sample was tested with indicators. Identify its nature.
📌 Answer:
Red litmus: No change
Blue litmus: Turns red
Turmeric: No change
Blue litmus turning red indicates an acidic substance.
✔️ Therefore, the liquid is acidic in nature.
🔒 ❓ Question 5.
Manya is blindfolded and given two unknown solutions. Which indicator should she use and why?
📌 Answer:
Manya should use olfactory indicators like onion or vanilla essence.
These indicators do not require vision. The smell changes in acidic or basic solutions, making them suitable for a blindfolded person.
🔒 ❓ Question 6.
Suggest materials for writing a secret message and what could be in the spray bottle.
📌 Answer:
Writing materials:
Lemon juice
Onion juice
Vinegar
Spray bottle contents (indicators):
Turmeric solution
Soap solution
Baking soda solution
The colour change after spraying reveals the hidden message due to acid–base reactions.
🔒 ❓ Question 7.
Grape juice mixed with red rose extract turns red. What happens if baking soda is added?
📌 Answer:
Baking soda is basic. When added, it neutralises the acidic grape juice.
The red colour will change to green, indicating a basic solution.
🔒 ❓ Question 8.
Keerthi wrote a secret message using orange juice. How can it be revealed?
📌 Answer:
Orange juice is acidic.
Spraying a basic indicator like turmeric solution will reveal the message as a colour change on the paper.
🔒 ❓ Question 9.
How can natural indicators be prepared? Give an example.
📌 Answer:
Natural indicators are prepared by extracting colour from natural substances.
Example:
Boil red rose petals in water
Cool and filter the solution
This extract changes colour in acidic and basic solutions and acts as a natural indicator.
🔒 ❓ Question 10.
Three liquids—vinegar, baking soda solution and sugar solution—are given. Can turmeric paper identify them?
📌 Answer:
Vinegar: No colour change → Acidic
Baking soda: Turmeric turns red → Basic
Sugar solution: No change → Neutral
Thus, turmeric paper can be used to identify all three.
🔒 ❓ Question 11.
Red rose extract turns liquid X green. What is its nature? What happens if excess amla juice is added?
📌 Answer:
Green colour indicates the liquid is basic.
When excess amla juice (acidic) is added, the solution becomes neutral or acidic, and the colour changes back towards red.
🔒 ❓ Question 12.
Observe the flowchart and complete the information.
📌 Answer:
The soil can be acidic in nature.
The soil can be basic in nature.
Indicator used: pH paper / universal indicator
Acidic soil treated with quick lime or slaked lime
Basic soil treated with organic matter or compost
🔹 Dive Deeper
🔒 ❓ Question 13.
Why do bubbles appear when vinegar is added to eggshell or marble but not with soap solution?
📌 Answer:
Eggshell and marble contain calcium carbonate. Vinegar is acidic and reacts with calcium carbonate to release carbon dioxide gas, forming bubbles.
Soap solution is basic and does not react with calcium carbonate, so no bubbles are produced.
——————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS
SECTION 1 — MCQs (5 Questions)
🔒 ❓ Q1. Which substance turns blue litmus red?
🟢 1️⃣ Acid
🔵 2️⃣ Base
🟡 3️⃣ Neutral substance
🟣 4️⃣ Salt
✔️ Answer: 🟢 1️⃣ Acid
🔒 ❓ Q2. Which of the following is basic in nature?
🟢 1️⃣ Lemon juice
🔵 2️⃣ Vinegar
🟡 3️⃣ Soap solution
🟣 4️⃣ Curd
✔️ Answer: 🟡 3️⃣ Soap solution
🔒 ❓ Q3. Which substance shows no change in litmus paper?
🟢 1️⃣ Acid
🔵 2️⃣ Base
🟡 3️⃣ Neutral substance
🟣 4️⃣ Alkali
✔️ Answer: 🟡 3️⃣ Neutral substance
🔒 ❓ Q4. Which indicator is obtained from plants?
🟢 1️⃣ Litmus
🔵 2️⃣ Phenolphthalein
🟡 3️⃣ Methyl orange
🟣 4️⃣ Universal indicator
✔️ Answer: 🟢 1️⃣ Litmus
🔒 ❓ Q5. What happens when an acid reacts with a base?
🟢 1️⃣ Heat only
🔵 2️⃣ Neutralisation
🟡 3️⃣ Evaporation
🟣 4️⃣ Condensation
✔️ Answer: 🔵 2️⃣ Neutralisation
SECTION 2 — Very Short Answer (5 Questions)
🔒 ❓ Q6. Name one acidic substance.
📌 ✅ Answer: Lemon juice
🔒 ❓ Q7. Name one basic substance.
📌 ✅ Answer: Soap
🔒 ❓ Q8. Which indicator is used to test acids and bases?
📌 ✅ Answer: Litmus
🔒 ❓ Q9. What is the nature of pure water?
📌 ✅ Answer: Neutral
🔒 ❓ Q10. Name the reaction between an acid and a base.
📌 ✅ Answer: Neutralisation
SECTION 3 — Short Answer (3 Questions)
🔒 ❓ Q11. How can you identify an acidic substance using litmus paper?
📌 ✅ Answer:
🔹 An acidic substance turns blue litmus paper red.
🔸 It shows this change because acids release hydrogen ions.
🔹 This property helps in identifying acids easily.
🔒 ❓ Q12. Why is soap solution considered basic?
📌 ✅ Answer:
🔹 Soap solution turns red litmus paper blue.
🔸 This indicates the presence of basic substances.
🔹 Hence, soap solution is basic in nature.
🔒 ❓ Q13. What is neutralisation?
📌 ✅ Answer:
🔹 Neutralisation is a reaction between an acid and a base.
🔸 It produces salt and water.
🔹 This reaction reduces harmful effects of acids and bases.
SECTION 4 — Long Answer (1 Question)
🔒 ❓ Q14. Explain acids, bases and neutral substances with examples.
📌 ✅ Answer:
🔹 Acids are substances that turn blue litmus red, like lemon juice.
🔸 Bases turn red litmus blue, such as soap solution.
🔹 Neutral substances show no change in litmus paper, like pure water.
🔸 These properties help in identifying substances easily.
——————————————————————————————————————————————————————————————————————————–
ADVANCE KNOWLEDGE
🌍 Substances Around Us: A Hidden Chemical World
Every substance around us—lemon juice, soap, milk, rainwater, soil—quietly carries a chemical nature. Some substances bite with sourness, some feel slippery, and some seem calm and harmless. These differences are not accidents; they arise from how substances interact at a microscopic level.
🧠 Big idea:
Acidic, basic, and neutral are not labels—they are behaviours of substances in water.
🧪 What Makes a Substance Acidic or Basic?
A substance shows its true nature only when dissolved in water.
🧪 Acids
Release special particles in water
Taste sour
Can be strong or weak
🧪 Bases
Release a different kind of particle
Taste bitter
Feel slippery
🧠 Neutral substances
Release neither of the above
Remain chemically balanced
⭐ Water is the silent stage where this chemical drama happens.
⚠️ Misconception vs Reality
⚠️ Misconception: All acids are dangerous
✅ Reality: Many acids are essential for life (like stomach acid)
⚠️ Misconception: Bases are always harmless
✅ Reality: Strong bases can burn skin badly
⚠️ Misconception: Neutral means useless
✅ Reality: Neutral substances are vital for balance in nature
⏳ A Short History of Acids and Bases
⏳ Ancient civilizations:
Used vinegar for preservation
Used lime for construction
🧠 The word acid comes from a Latin word meaning sour.
⏳ Scientists slowly realized:
Sour taste, corrosive nature, and reactivity were connected
Bases could neutralize acids
⭐ This understanding changed medicine, industry, and agriculture.
🧠 Indicators: Nature’s Chemical Testers
Indicators are substances that change colour depending on chemical nature.
🌿 Natural indicators
Litmus from lichens
Turmeric from plants
🧪 Synthetic indicators
Phenolphthalein
Methyl orange
⭐ Plants became chemists long before humans did.
🌈 pH Scale: Measuring Chemical Strength
Not all acids or bases are equal.
🧠 Scientists created the pH scale:
Ranges from 0 to 14
Middle (7) is neutral
🌡️ Lower pH → stronger acid
🌡️ Higher pH → stronger base
⭐ Small pH changes mean big chemical differences.
🌍 Acids and Bases in Living Organisms
Life depends on chemical balance.
🧠 Examples:
Stomach acid helps digestion
Blood pH must remain stable
Soil pH affects crop growth
⚠️ Even small imbalance can cause illness or crop failure.
🌧️ Acid Rain: A Modern Warning
🚀 Industrial gases react with water in air to form acids.
🌧️ Acid rain:
Damages monuments
Harms plants
Affects water bodies
🧠 This shows how human activity can change natural chemistry.
🌱 Bases in Agriculture and Daily Life
🧪 Farmers use basic substances to:
Reduce soil acidity
Improve crop yield
🧠 Soap and detergents work because of their basic nature—they break grease molecules.
🚀 Modern Uses of Acids and Bases
🚀 Acids are used in:
Batteries
Food preservation
Medicines
🚀 Bases are used in:
Paper making
Cleaning agents
Water treatment
⭐ Civilization runs silently on acid–base chemistry.
⚠️ Neutralisation: Chemistry’s Peace Process
When acids and bases meet, they cancel each other.
🧠 This process:
Reduces harmful effects
Produces salt and water
⭐ Antacids work using this simple idea.
🧠 Acids, Bases, and the Future
Scientists now design:
Safer chemicals
Environment-friendly cleaners
Better medicines
🧠 Understanding acids and bases helps protect:
Human health
Soil
Water
Environment
⭐ Amazing Chemical Facts
⭐ Rainwater is slightly acidic
⭐ Pure water rarely exists in nature
⭐ Bees sense flower pH
⭐ Ocean acidity affects coral reefs
🌟 Final Thought
Acids and bases are not enemies or heroes.
They are tools of balance, shaping life, industry, and nature.
🧠 To understand substances is to understand the chemistry of the world itself.
——————————————————————————————————————————————————————————————————————————–
