Class 12 : Chemistry (English) – Chapter 9: Amines
EXPLANATION & SUMMARY
✨ Introduction
🔵 Amines are derivatives of ammonia (NH₃) in which one or more hydrogen atoms are replaced by alkyl or aryl groups.
🟢 They are considered as organic bases and are also important as nucleophiles.
🟠 Found in natural products (alkaloids, hormones, vitamins) and industrially useful (dyes, drugs, polymers).
🔴 Classified into aliphatic and aromatic amines.
🌿 Classification of Amines
By type of substitution:
Primary (1°): One R group, formula R–NH₂ (e.g., CH₃NH₂).
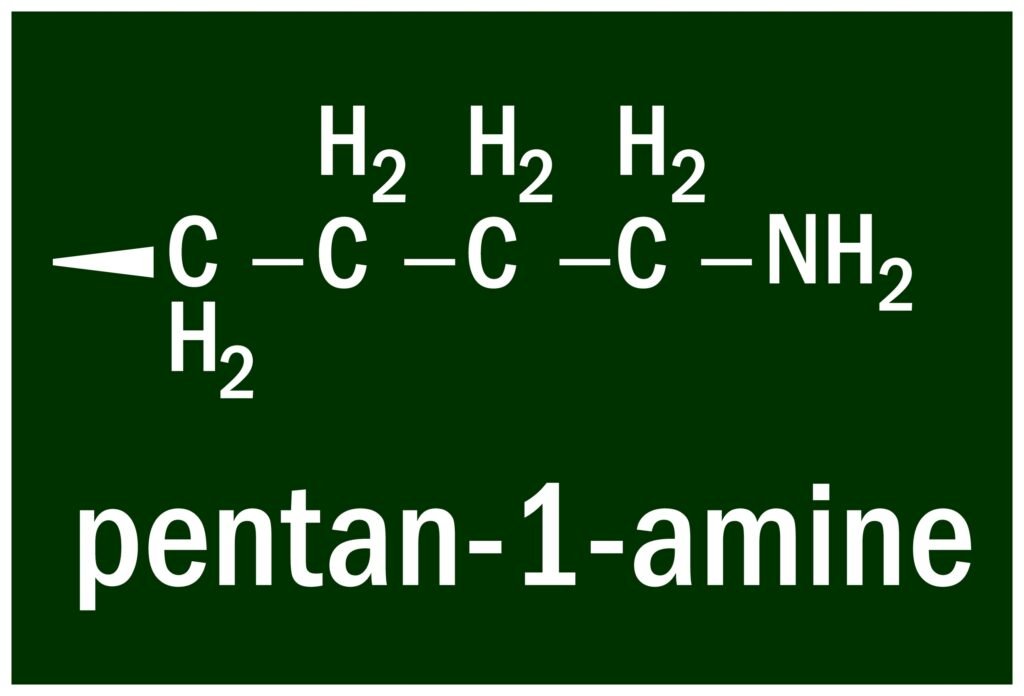
Secondary (2°): Two R groups, formula R₂NH (e.g., (CH₃)₂NH).
Tertiary (3°): Three R groups, formula R₃N (e.g., (CH₃)₃N).
By nature of group attached:
Aliphatic amines: R = alkyl.
Aromatic amines: R = aryl (e.g., aniline).
🧪 Nomenclature
Common system: Alkyl groups + amine (e.g., ethylamine).
IUPAC system: Replace “-e” of alkane with “-amine” (e.g., ethanamine).
For aromatic amines, name derived from parent aromatic amine (e.g., aniline, N-methylaniline).
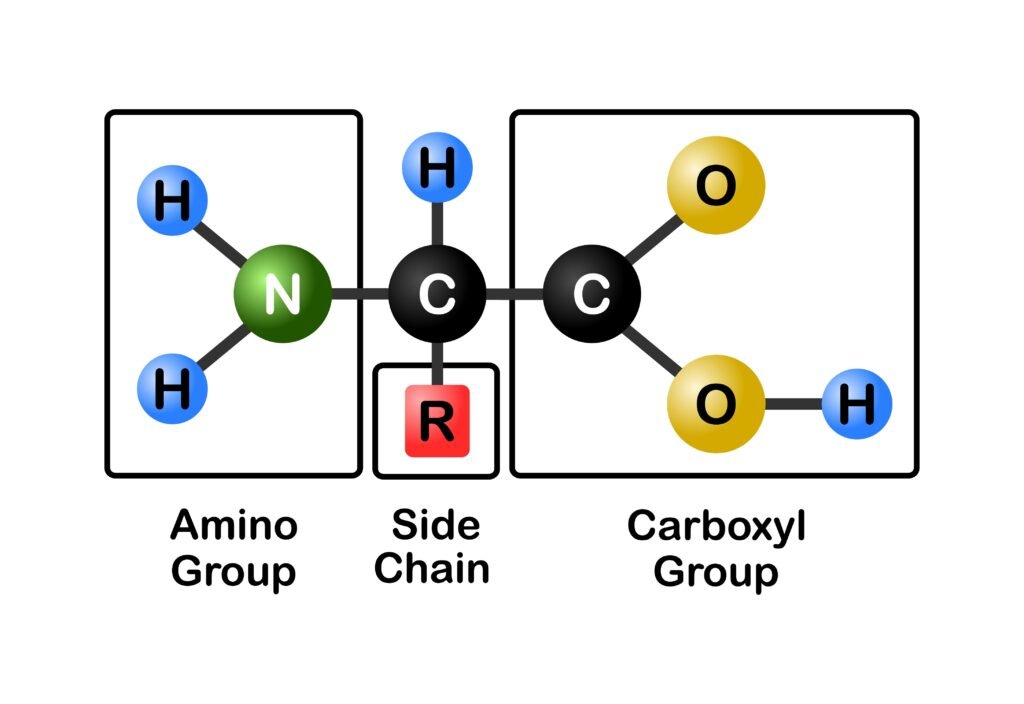
⚡ Structure of Amines
🔵 The nitrogen atom is sp³ hybridised.
🟢 Geometry: Trigonal pyramidal.
🟠 Bond angle ~ 107°, slightly less than tetrahedral angle due to lone pair repulsion.
🔬 Preparation of Amines
Reduction Methods
Nitro compounds → Amines (Sn/HCl, Fe/HCl, catalytic hydrogenation).
Nitriles (RCN) → Amines (LiAlH₄ or H₂/Ni).
Amides → Amines (LiAlH₄ or NaBH₄).
Ammonolysis of alkyl halides
R–X + NH₃ → R–NH₂ + HX (further reaction gives secondary/tertiary amines).
Gabriel phthalimide synthesis
K-phthalimide + R–X → R–NH₂ (primary amine only).
Hoffmann bromamide degradation
RCONH₂ + Br₂ + NaOH → R–NH₂ (1° amine, one carbon less).
🌍 Physical Properties
🔵 Lower aliphatic amines: gases with fishy odour.
🟢 Higher amines: liquids/solids.
🟠 Solubility: Lower amines soluble in water due to H-bonding; solubility decreases with size.
🔴 Boiling points: Primary > Secondary > Tertiary (due to H-bonding differences).
💡 Chemical Properties
Basic character
Amines act as bases due to lone pair on N.
Measured by Kb and pKb values.
Order in gas phase: 3° > 2° > 1° > NH₃.
In aqueous solution: 2° > 1° > 3° (hydration effect).
Aromatic amines less basic due to delocalisation of lone pair into ring.
Reaction with acids
R–NH₂ + HCl → R–NH₃⁺Cl⁻
Reaction with acyl halides
R–NH₂ + R′COCl → R′CONHR + HCl (acylation).
Reaction with nitrous acid
1° aliphatic amines → alcohols + N₂.
1° aromatic amines → diazonium salts.
2° amines → nitrosoamines.
3° amines → ammonium salts.
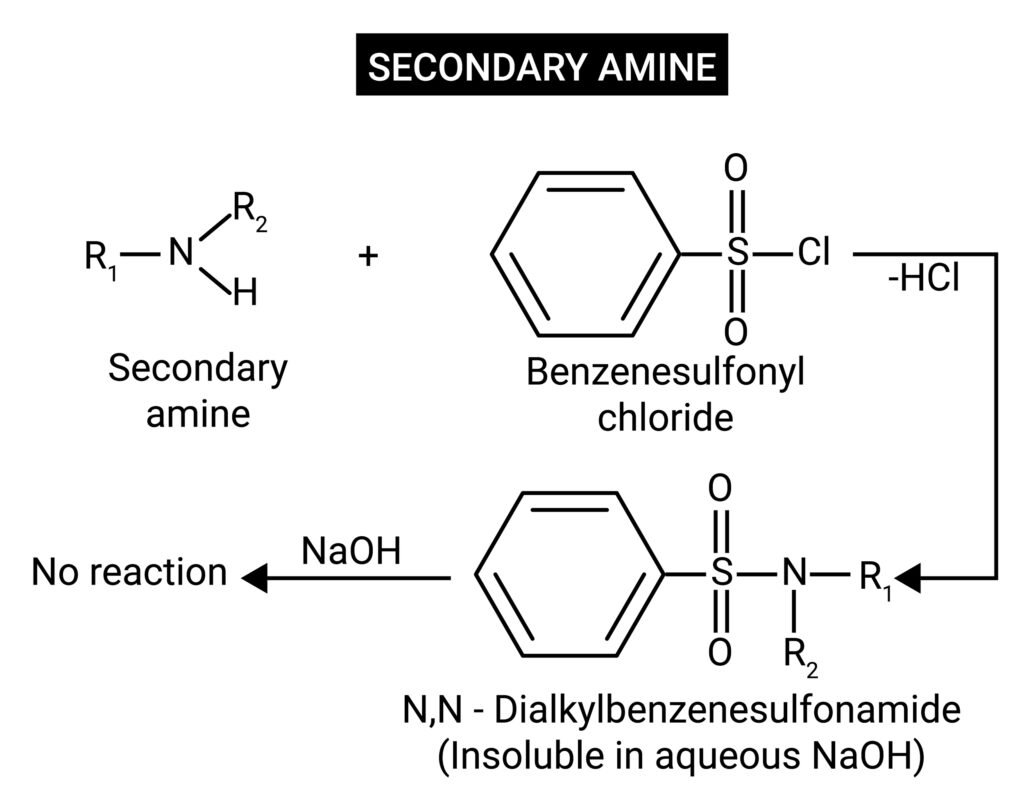
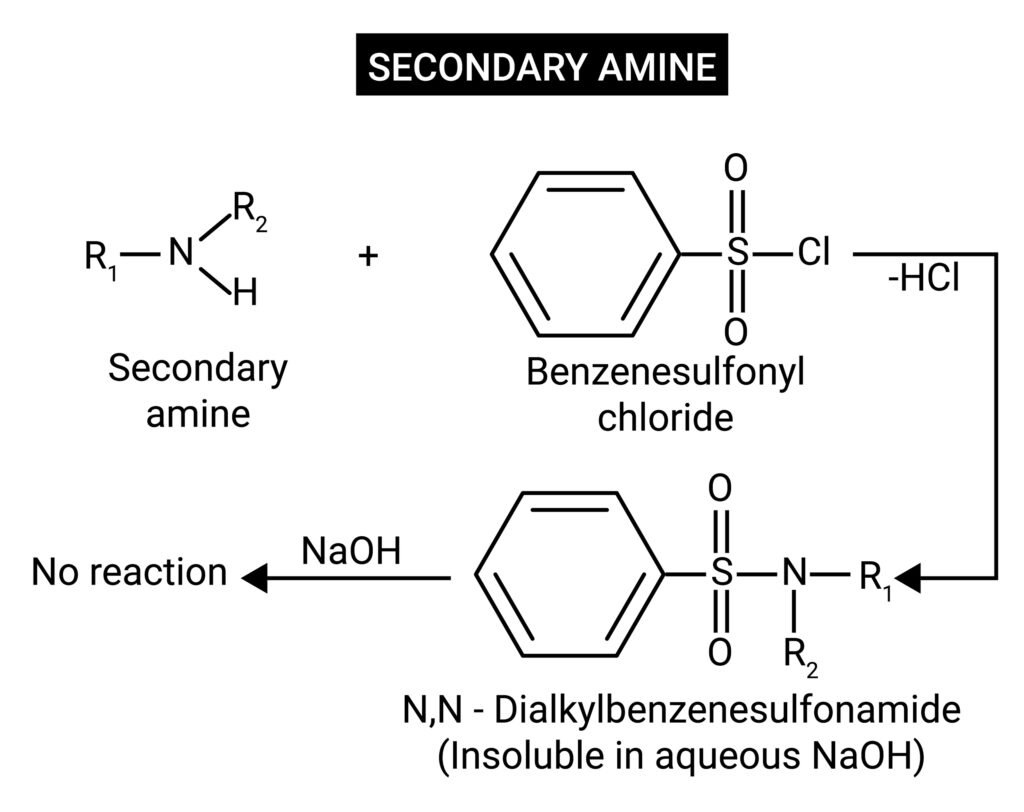
Hinsberg’s test (distinction between 1°, 2°, 3° amines).
Carbylamine reaction
1° amine + CHCl₃ + alc. KOH → isocyanide (foul smell).
🌸 Diazonium Salts
🔵 Benzene diazonium chloride (C₆H₅N₂⁺Cl⁻): obtained from aniline + NaNO₂ + HCl (0–5 °C).
🟢 Used in synthesis of azo dyes (coupling reactions).
🟠 Unstable above 5 °C → decomposes.
🔴 Reactions: Replacement by –OH, –Cl, –Br, –I, –CN, –NO₂, –H.
🌟 Uses of Amines
Aniline: precursor for dyes, drugs, rubber chemicals.
Alkylamines: corrosion inhibitors, solvents.
Diazonium salts: azo dyes, synthetic intermediates.
📝 Summary
Amines are ammonia derivatives (R–NH₂, R₂NH, R₃N). Classified as aliphatic or aromatic; primary, secondary, tertiary.
Nomenclature: Common (alkylamine, aniline derivatives), IUPAC (-amine ending).
Structure: N sp³ hybridised, trigonal pyramidal, bond angle ~107°.
Preparation: Reduction of nitro compounds, nitriles, amides; ammonolysis of halides; Gabriel synthesis; Hoffmann bromamide.
Physical properties: Fishy odour, solubility due to H-bonding, boiling point trend: 1° > 2° > 3°.
Chemical properties:
Basic nature (lone pair on N, hydration effect, aromatic vs aliphatic).
Acylation, salt formation, nitrous acid reactions.
Special tests: Hinsberg, Carbylamine.
Aromatic amines form diazonium salts at 0–5 °C. These are key intermediates for azo dyes, halogenation, substitution, cyanation.
Applications: Drugs, dyes, polymers, agrochemicals, intermediates.
🎯 Quick Recap
🟦 Amines = derivatives of NH₃.
🟩 Classified as 1°, 2°, 3°, aliphatic, aromatic.
🟨 Prepared by reduction, Gabriel synthesis, Hoffmann degradation.
🟧 Show basicity, acylation, nitrous acid, Hinsberg’s test, Carbylamine.
🟪 Aromatic amines form diazonium salts (dye synthesis).
🟫 Industrial importance: drugs, dyes, agrochemicals.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
Question 9.1
Write IUPAC names of the following compounds and classify them into primary, secondary and tertiary amines:
(i) (CH₃)₂CHNH₂
(ii) CH₃(CH₂)₂NH₂
(iii) CH₃NHCH(CH₃)₂
(iv) (CH₃)₃CNH₂
(v) C₆H₅NHCH₃
(vi) CH₃CH₂N(CH₃)₂
(vii) m–BrC₆H₄NH₂
Answer 9.1
🟦 (i) 2-Aminopropane → Primary amine
🟩 (ii) Propan-1-amine → Primary amine
🟨 (iii) N-Methylpropan-2-amine → Secondary amine
🟧 (iv) 2-Methyl-2-aminopropane → Primary amine
🟪 (v) N-Methylaniline → Secondary aromatic amine
🟫 (vi) N,N-Dimethylethanamine → Tertiary amine
🔷 (vii) 3-Bromoaniline → Primary aromatic amine
Question 9.2
Give one chemical test to distinguish between the following pairs of compounds:
(i) Methylamine and dimethylamine
(ii) Secondary and tertiary amines
(iii) Ethylamine and aniline
(iv) Aniline and benzylamine
(v) Aniline and N-methylaniline
Answer 9.2
🟦 (i) Methylamine vs Dimethylamine
➤ Carbylamine test: Methylamine (1°) gives foul-smelling isocyanide with CHCl₃ + alc. KOH.
➤ Dimethylamine (2°) does not respond.
🟩 (ii) Secondary vs Tertiary amines
➤ Hinsberg test: Secondary amine forms insoluble sulphonamide.
➤ Tertiary amine does not react.
🟨 (iii) Ethylamine vs Aniline
➤ Solubility: Ethylamine soluble in water, aniline insoluble.
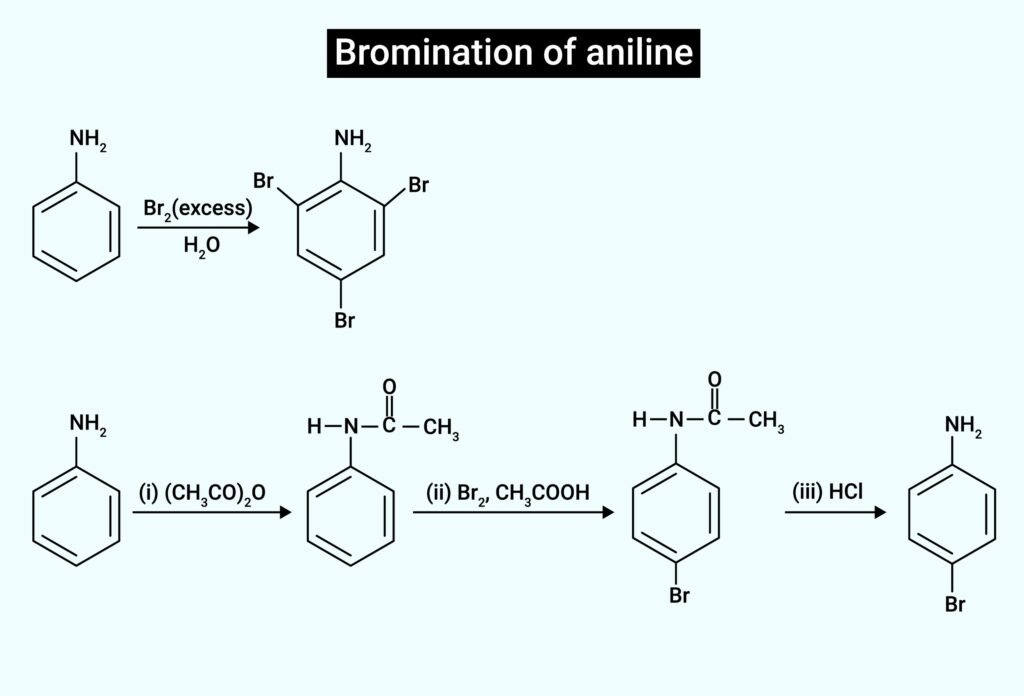
➤ Also, aniline gives bromination test → 2,4,6-tribromoaniline.
🟧 (iv) Aniline vs Benzylamine
➤ Nitrous acid test: Aniline gives stable diazonium salt at 0–5 °C.
➤ Benzylamine gives unstable diazonium salt → immediate N₂ evolution.
🟪 (v) Aniline vs N-Methylaniline
➤ Azo coupling: Aniline couples with phenol to form azo dye.
➤ N-Methylaniline does not couple.
Question 9.3
Account for the following:
(i) pKb of aniline is more than that of methylamine.
(ii) Ethylamine is soluble in water whereas aniline is not.
(iii) Methylamine in water reacts with ferric chloride to precipitate hydrated ferric oxide.
(iv) Although –NH₂ is o,p-directing, aniline on nitration gives m-nitroaniline as a major product.
(v) Aniline does not undergo Friedel–Crafts reaction.
(vi) Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
(vii) Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Answer 9.3
🟦 (i) In aniline, lone pair on N delocalises into benzene ring → less available for protonation → weaker base → higher pKb.
🟩 (ii) Ethylamine forms strong H-bonds with water → soluble. Aniline, bulky aromatic ring → insoluble.
🟨 (iii) Methylamine is alkaline → reacts with FeCl₃ forming Fe(OH)₃ precipitate (brown).
🟧 (iv) Nitration of aniline is carried out in acidic medium. –NH₂ gets protonated to –NH₃⁺ (deactivating, m-directing) → m-product predominates.
🟪 (v) In Friedel–Crafts, AlCl₃ catalyst reacts with –NH₂ to form a complex → reaction blocked.
🟫 (vi) Aromatic diazonium ions are resonance-stabilised → more stable. Aliphatic diazonium ions unstable → release N₂ immediately.
🔷 (vii) Gabriel synthesis ensures only primary amine (no secondary/tertiary), hence preferred.
Question 9.4
Arrange the following:
(i) In decreasing order of pKb values:
C₆H₅NH₂, C₆H₅N(CH₃)₂, C₆H₅CH₂NH₂, C₆H₅NHCH₃
(ii) In increasing order of basic strength:
C₆H₅NH₂, C₆H₅CH₂NH₂, (C₂H₅)₂NH, CH₃NH₂
(iii) In increasing order of basic strength:
Aniline, p-Nitroaniline, p-Toluidine
(iv) In decreasing order of basic strength in gas phase:
C₂H₅NH₂, (C₂H₅)₃N, (C₂H₅)₂NH, NH₃
(v) In increasing order of boiling point:
C₂H₅OH, (CH₃)₂NH, C₂H₅NH₂
(vi) In increasing order of solubility in water:
C₆H₅NH₂, (C₂H₅)₂NH, C₂H₅NH₂
Answer 9.4
🟦 (i) pKb order (higher pKb = weaker base):
C₆H₅N(CH₃)₂ < C₆H₅CH₂NH₂ < C₆H₅NHCH₃ < C₆H₅NH₂
🟩 (ii) Basic strength order:
C₆H₅NH₂ < C₆H₅CH₂NH₂ < CH₃NH₂ < (C₂H₅)₂NH
🟨 (iii) Aniline < p-Nitroaniline < p-Toluidine
🟧 (iv) Gas phase: (C₂H₅)₃N < (C₂H₅)₂NH < C₂H₅NH₂ < NH₃
🟪 (v) Boiling point: (CH₃)₂NH < C₂H₅NH₂ < C₂H₅OH
🟫 (vi) Solubility: C₆H₅NH₂ < (C₂H₅)₂NH < C₂H₅NH₂
Question 9.5
How will you convert:
(i) Ethanoic acid into methanamine
(ii) Hexanenitrile into 1-aminopentane
(iii) Methanol to ethanoic acid
(iv) Ethanamine into methanamine
(v) Ethanoic acid into propanoic acid
(vi) Methanamine into ethanamine
(vii) Nitromethane into dimethylamine
(viii) Propanoic acid into ethanoic acid
Answer 9.5
🟦 (i) CH₃COOH → (SOCl₂) → CH₃COCl → (NH₃) → CH₃CONH₂ → (Br₂/NaOH) → CH₃NH₂
🟩 (ii) CH₃(CH₂)₄CN → (LiAlH₄) → CH₃(CH₂)₄CH₂NH₂ → 1-Aminopentane
🟨 (iii) CH₃OH → (Cu, 573 K) → HCHO → (K₂Cr₂O₇/H⁺) → HCOOH → (oxidation) → CH₃COOH
🟧 (iv) CH₃CH₂NH₂ → (HNO₂) → Ethanol → (oxidation) → CH₃CHO → (oxidation) → CH₃COOH → (Hoffmann) → CH₃NH₂
🟪 (v) CH₃COOH → (HCN/KCN) → CH₃CH(OH)CN → (hydrolysis) → CH₃CH₂COOH
🟫 (vi) CH₃NH₂ → (CH₃I + KOH) → (CH₃)₂NH → (excess CH₃I) → (CH₃)₃N → (reduction) → C₂H₅NH₂
🔷 (vii) CH₃NO₂ → (Zn/HCl) → CH₃NH₂ → (CH₃I + KOH) → (CH₃)₂NH
🔶 (viii) CH₃CH₂COOH → (oxidation) → CH₃COOH
Question 9.6
Describe a method for the identification of primary, secondary and tertiary amines. Also write chemical equations of the reactions involved.
Answer 9.6
🟦 Hinsberg’s Test
Reagent: Benzenesulphonyl chloride in NaOH.
➤ Primary amine (1°): Forms sulphonamide soluble in alkali.
Equation: RNH₂ + C₆H₅SO₂Cl → R–NHSO₂C₆H₅ (soluble).
➤ Secondary amine (2°): Forms sulphonamide insoluble in alkali.
Equation: R₂NH + C₆H₅SO₂Cl → R₂N–SO₂C₆H₅ (insoluble).
➤ Tertiary amine (3°): Does not react with C₆H₅SO₂Cl.
Question 9.7
Write short notes on the following:
(i) Carbylamine reaction
(ii) Diazotisation
(iii) Hofmann’s bromamide reaction
(iv) Coupling reaction
(v) Ammonolysis
(vi) Acetylation
(vii) Gabriel phthalimide synthesis
Answer 9.7
🟦 (i) Carbylamine reaction
1° amine + CHCl₃ + alc. KOH → R–NC (isocyanide, foul smell).
🟩 (ii) Diazotisation
Aromatic 1° amine + NaNO₂ + HCl (0–5 °C) → ArN₂⁺Cl⁻ (diazonium salt).
🟨 (iii) Hofmann bromamide reaction
RCONH₂ + Br₂ + NaOH → R–NH₂ (1° amine with one C less).
🟧 (iv) Coupling reaction
Diazonium salt + phenol/aniline → azo dye (Ar–N=N–Ar′).
🟪 (v) Ammonolysis
Alkyl halide + NH₃ → amine(s) + HX.
🟫 (vi) Acetylation
RNH₂ + CH₃COCl → RNHCOCH₃ + HCl.
🔷 (vii) Gabriel phthalimide synthesis
Potassium phthalimide + R–X → R–NH₂ (only primary amine).
Question 9.8
Accomplish the following conversions:
(i) Nitrobenzene to benzoic acid
(ii) Benzene to m-bromophenol
(iii) Benzoic acid to aniline
(iv) Aniline to 2,4,6-tribromoaniline
(v) Benzyl chloride to 2-phenylethanamine
(vi) Chlorobenzene to p-chloroaniline
(vii) Aniline to p-bromoaniline
(viii) Benzamide to toluene
(ix) Aniline to benzyl alcohol
Answer 9.8
🟦 (i) Nitrobenzene → (reduction, Sn/HCl) → Aniline → (oxidation, KMnO₄) → Benzoic acid
🟩 (ii) Benzene → (nitration, HNO₃/H₂SO₄) → Nitrobenzene → (reduction, Sn/HCl) → Aniline → (diazotisation, NaNO₂/HCl, 273 K) → Diazonium salt → (hydrolysis, H₂O) → Phenol → (Br₂, Fe) → m-Bromophenol
🟨 (iii) Benzoic acid → (SOCl₂) → Benzoyl chloride → (NH₃) → Benzamide → (Hoffmann bromamide) → Aniline
🟧 (iv) Aniline → (Br₂ in water) → 2,4,6-tribromoaniline (white ppt).
🟪 (v) Benzyl chloride → (KCN) → Benzyl cyanide → (reduction, LiAlH₄) → 2-Phenylethanamine
🟫 (vi) Chlorobenzene → (nitration) → p-Nitrochlorobenzene → (reduction, Sn/HCl) → p-Chloroaniline
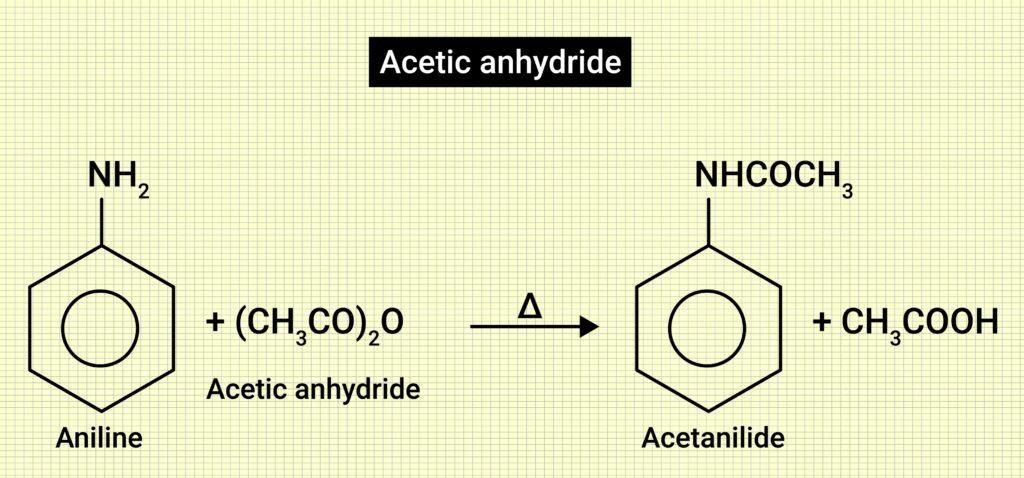
🔷 (vii) Aniline → (acetylation with Ac₂O) → Acetanilide → (Br₂, AcOH) → p-Bromoacetanilide → (hydrolysis) → p-Bromoaniline
🔶 (viii) Benzamide → (Hoffmann bromamide, Br₂/NaOH) → Aniline → (reduction of diazonium salt) → Toluene
✨ (ix) Aniline → (diazotisation) → Benzene diazonium chloride → (reduction with H₂O/SnCl₂) → Benzyl alcohol
Question 9.9
Give the structures of A, B and C in the following reactions:
(i) CH₃CH₂I →(NaCN)→ A →(hydrolysis)→ B →(NaOH + Br₂)→ C
(ii) C₆H₅N₂Cl →(CuCN)→ A →(H₃O⁺/Δ)→ B →(NH₃)→ C
(iii) CH₃CH₂Br →(KCN)→ A →(LiAlH₄)→ B →(HNO₂, 0 °C)→ C
(iv) C₆H₅NO₂ →(Fe/HCl)→ A →(NaNO₂ + HCl, 273 K)→ B →(H₂O/H⁺/Δ)→ C
(v) CH₃COOH →(NH₃)→ A →(NaOBr)→ B →(NaNO₂/HCl)→ C
(vi) C₆H₅NO₂ →(Fe/HCl)→ A →(NaNO₂, 273 K)→ B →(C₂H₅OH)→ C
Answer 9.9
🟦 (i)
A = CH₃CH₂CN (Propanenitrile)
B = CH₃CH₂COOH (Propanoic acid)
C = CH₃CH₂NH₂ (Ethylamine)
🟩 (ii)
A = C₆H₅CN (Benzonitrile)
B = C₆H₅COOH (Benzoic acid)
C = C₆H₅CONH₂ (Benzamide)
🟨 (iii)
A = CH₃CH₂CN (Propanenitrile)
B = CH₃CH₂CH₂NH₂ (Propylamine)
C = CH₃CH₂CH₂OH (Propanol, from diazotisation–hydrolysis)
🟧 (iv)
A = C₆H₅NH₂ (Aniline)
B = C₆H₅N₂⁺Cl⁻ (Benzene diazonium chloride)
C = C₆H₅OH (Phenol)
🟪 (v)
A = CH₃CONH₂ (Acetamide)
B = CH₃NH₂ (Methylamine)
C = CH₃OH (Methanol, from diazotisation–hydrolysis)
🟫 (vi)
A = C₆H₅NH₂ (Aniline)
B = C₆H₅N₂⁺Cl⁻ (Benzene diazonium chloride)
C = C₆H₅OC₂H₅ (Phenetole)
Question 9.10
An aromatic compound ‘A’ on treatment with aqueous ammonia and heating forms compound ‘B’. On heating with Br₂ and KOH, ‘B’ forms compound ‘C’ of molecular formula C₆H₇N. Write the structures and IUPAC names of compounds A, B and C.
Answer 9.10
🟦 Step 1: Compound A = C₆H₅CH₂Cl (Benzyl chloride)
➤ Reaction with aq. NH₃ → C₆H₅CH₂NH₂ (Benzylamine, B).
🟩 Step 2: B (Benzylamine) + Br₂/NaOH → Hoffmann rearrangement → C₆H₅NH₂ (Aniline, C).
✅ Final:
A = Benzyl chloride (C₆H₅CH₂Cl)
B = Benzylamine (C₆H₅CH₂NH₂)
C = Aniline (C₆H₅NH₂)
Question 9.11
Complete the following reactions:
(i) C₆H₅NH₂ + CHCl₃ + alc.KOH → ?
(ii) C₆H₅N₂Cl + H₃PO₂ + H₂O → ?
(iii) C₆H₅NH₂ + H₂SO₄ (conc.) → ?
(iv) C₆H₅N₂Cl + C₂H₅OH → ?
(v) C₆H₅NH₂ + Br₂ (aq) → ?
(vi) C₆H₅NH₂ + (CH₃CO)₂O → ?
(vii) C₆H₅N₂Cl →(i) HBF₄, (ii) NaNO₂/Cu, Δ→ ?
Answer 9.11
🟦 (i) → C₆H₅NC (Phenyl isocyanide, foul smell)
🟩 (ii) → C₆H₆ (Benzene)
🟨 (iii) → C₆H₅NH₃⁺HSO₄⁻ (Anilinium hydrogen sulfate)
🟧 (iv) → C₆H₅OC₂H₅ (Phenetole)
🟪 (v) → 2,4,6-tribromoaniline (white ppt)
🟫 (vi) → Acetanilide (C₆H₅NHCOCH₃)
🔷 (vii) → Fluorobenzene (C₆H₅F)
Question 9.12
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Answer 9.12
🟦 Gabriel synthesis works via nucleophilic substitution (SN).
🟩 Aryl halides (Ar–X) are resistant to nucleophilic substitution due to resonance and partial double bond character.
✅ Hence, aromatic primary amines cannot be synthesised by this method.
Question 9.13
Write the reactions of:
(i) Aromatic primary amines
(ii) Aliphatic primary amines
with nitrous acid.
Answer 9.13
🟦 (i) Aromatic primary amines (ArNH₂):
➤ ArNH₂ + NaNO₂ + HCl (273 K) → ArN₂⁺Cl⁻ (stable diazonium salt) + H₂O
🟩 (ii) Aliphatic primary amines (RNH₂):
➤ RNH₂ + NaNO₂ + HCl → ROH + N₂↑ + HCl
(unstable diazonium → alcohol + N₂ gas)
Question 9.14
Give plausible explanation:
(i) Why are amines less acidic than alcohols of comparable molecular masses?
(ii) Why do primary amines have higher boiling points than tertiary amines?
(iii) Why are aliphatic amines stronger bases than aromatic amines?
Answer 9.14
🟦 (i) O–H bond in alcohol is more polar than N–H in amine → alcohol more acidic.
🟩 (ii) 1° amines have more –NH groups for H-bonding → higher boiling point than 3° amines (no N–H bond).
🟨 (iii) In aromatic amines, lone pair on N delocalises into benzene ring → less available for protonation → weaker base.
Aliphatic amines: electron-releasing alkyl groups increase electron density on N → stronger base.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
✨ Section A (Q1–Q16) – MCQs (1 mark each)
Options for Assertion–Reason questions:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Q1. Which of the following is the correct IUPAC name of CH₃–NH₂?
🔵 (A) Methyl amine
🟢 (B) Aminomethane
🟠 (C) Methanamine
🔴 (D) Aminomethyl
Answer: 🟠 (C) Methanamine
Q2. Among the following, which amine gives carbylamine test?
🔵 (A) Aniline
🟢 (B) Diphenylamine
🟠 (C) Triethylamine
🔴 (D) N-methyl aniline
Answer: 🔵 (A) Aniline
Q3. Primary amines can be distinguished from secondary and tertiary amines by:
🔵 (A) Tollen’s test
🟢 (B) Carbylamine test
🟠 (C) Iodoform test
🔴 (D) Fehling’s test
Answer: 🟢 (B) Carbylamine test
Q4. Which one of the following does not undergo diazotisation?
🔵 (A) Aniline
🟢 (B) p-Toluidine
🟠 (C) Benzylamine
🔴 (D) p-Aminophenol
Answer: 🟠 (C) Benzylamine
Q5. The basic strength of amines in aqueous solution decreases in the order:
🔵 (A) C₂H₅NH₂ > NH₃ > C₆H₅NH₂
🟢 (B) NH₃ > C₂H₅NH₂ > C₆H₅NH₂
🟠 (C) C₆H₅NH₂ > NH₃ > C₂H₅NH₂
🔴 (D) C₂H₅NH₂ > C₆H₅NH₂ > NH₃
Answer: 🔵 (A) C₂H₅NH₂ > NH₃ > C₆H₅NH₂
Q6. In Gabriel phthalimide synthesis, the limitation is:
🔵 (A) Primary aliphatic amines are not formed
🟢 (B) Secondary amines are not formed
🟠 (C) Aromatic primary amines are not formed
🔴 (D) Tertiary amines are not formed
Answer: 🟠 (C) Aromatic primary amines are not formed
Q7. Which reagent is used in Hoffmann bromamide reaction?
🔵 (A) Br₂/NaOH
🟢 (B) Br₂/H₂O
🟠 (C) Cl₂/NaOH
🔴 (D) NaBH₄
Answer: 🔵 (A) Br₂/NaOH
Q8. Assertion (A): Aniline undergoes bromination easily.
Reason (R): –NH₂ group is an ortho-para directing group.
Answer: 1
Q9. Assertion (A): Ethylamine is more basic than aniline.
Reason (R): In aniline, lone pair of nitrogen is delocalised over benzene ring.
Answer: 1
Q10. Which amine is soluble in water?
🔵 (A) Aniline
🟢 (B) Diphenylamine
🟠 (C) Ethylamine
🔴 (D) Triphenylamine
Answer: 🟠 (C) Ethylamine
Q11. Which compound on reduction gives aniline?
🔵 (A) Nitrobenzene
🟢 (B) Benzoic acid
🟠 (C) Benzamide
🔴 (D) Benzaldehyde
Answer: 🔵 (A) Nitrobenzene
Q12. Hinsberg’s reagent is:
🔵 (A) CHCl₃
🟢 (B) C₆H₅SO₂Cl
🟠 (C) C₂H₅Cl
🔴 (D) C₆H₅COCl
Answer: 🟢 (B) C₆H₅SO₂Cl
Q13. In diazonium salts, –N₂⁺ group is:
🔵 (A) Strongly nucleophilic
🟢 (B) Strongly electrophilic
🟠 (C) Weakly acidic
🔴 (D) Strongly basic
Answer: 🟢 (B) Strongly electrophilic
Q14. Which one is an aliphatic tertiary amine?
🔵 (A) (CH₃)₃N
🟢 (B) (C₂H₅)₂NH
🟠 (C) C₆H₅NH₂
🔴 (D) CH₃NH₂
Answer: 🔵 (A) (CH₃)₃N
Q15. Which diazonium salt is most stable?
🔵 (A) Methyl diazonium chloride
🟢 (B) Phenyl diazonium chloride
🟠 (C) Ethyl diazonium chloride
🔴 (D) n-Propyl diazonium chloride
Answer: 🟢 (B) Phenyl diazonium chloride
Q16. Which test is not given by tertiary amines?
🔵 (A) Carbylamine test
🟢 (B) Acetylation
🟠 (C) Nitrous acid test
🔴 (D) Reaction with HCl
Answer: 🔵 (A) Carbylamine test
✨ Section B (Q17–Q21) – Very Short Answer (2 marks each)
Q17. Write any two differences between primary and secondary amines.
🟦 Primary amines: one alkyl group attached to –NH₂.
🟩 Secondary amines: two alkyl groups attached to nitrogen.
📌 Primary amines give carbylamine test, secondary amines do not.
Q18. Why do tertiary amines have lower boiling points than secondary amines?
🟨 Secondary amines: hydrogen bonding present → stronger intermolecular forces.
🟪 Tertiary amines: no –NH hydrogen → no H-bonding → weaker forces → lower boiling point.
Q19. Write reaction of ethanamine with nitrous acid.
🧪 CH₃CH₂NH₂ + HNO₂ → CH₃CH₂OH + N₂↑ + H₂O
🎯 Primary aliphatic amine → alcohol + nitrogen gas.
Q20. Give IUPAC names of: (i) C₆H₅–NH₂, (ii) CH₃–NH–CH₃.
➤ (i) Aniline (benzenamine)
➤ (ii) N-methyl methanamine
Q21. Write short note on diazotisation.
🔷 Primary aromatic amine + NaNO₂ + HCl (0–5 °C) → diazonium salt.
⚗ Example: C₆H₅NH₂ → C₆H₅N₂⁺Cl⁻.
🎯 Reaction used for azo dye synthesis and coupling reactions.
✨ Section C (Q22–Q28) – Short Answer (3 marks each)
Q22. Explain Hinsberg’s test.
🟦 Reagent: benzenesulphonyl chloride (C₆H₅SO₂Cl).
➤ Primary amines → soluble sulphonamide (dissolves in alkali).
➤ Secondary amines → insoluble sulphonamide.
➤ Tertiary amines → no reaction.
✅ Thus, it distinguishes 1°, 2°, 3° amines.
Q23. Write the steps of Hofmann bromamide reaction with example.
🧪 Step 1: Amide + Br₂/NaOH → N-bromoamide.
🧪 Step 2: Base-induced rearrangement → isocyanate.
🧪 Step 3: Hydrolysis of isocyanate → primary amine (1C less).
🎯 Example: CH₃CONH₂ → CH₃NH₂.
Q24. Write chemical equation for diazotisation of aniline and its coupling with phenol.
⚗ Aniline + NaNO₂/HCl (0–5 °C) → C₆H₅N₂⁺Cl⁻.
➜ Coupling with phenol in alkaline medium → azo dye (p-hydroxyazobenzene).
✅ Used in dye industry.
Q25. Give reasons:
(i) Aniline is less basic than aliphatic amines.
(ii) Tertiary amines cannot be prepared by Gabriel synthesis.
(iii) Diazonium salts are used in dyeing.
🟦 (i) Resonance delocalises lone pair in aniline.
🟩 (ii) Gabriel method only gives primary amines.
🟨 (iii) They couple with aromatic rings to form coloured azo dyes.
Q26. Write short note on carbylamine test with example.
🧪 Primary amine + CHCl₃ + alcoholic KOH → isocyanide (foul smell).
➜ Example: C₆H₅NH₂ + CHCl₃ + 3KOH → C₆H₅NC + 3KCl + 3H₂O.
✅ Test for primary amines.
Q27. Compare basic strengths of NH₃, CH₃NH₂ and (CH₃)₂NH in aqueous medium.
🔷 NH₃: weak base.
🔶 CH₃NH₂: +I effect of CH₃ increases basicity.
🟩 (CH₃)₂NH: stronger base due to more +I and effective H-bonding.
🎯 Order: (CH₃)₂NH > CH₃NH₂ > NH₃.
Q28. Write the difference between aliphatic and aromatic amines with examples.
🟦 Aliphatic: nitrogen attached to alkyl group (e.g., CH₃NH₂).
🟩 Aromatic: nitrogen attached to aryl group (e.g., C₆H₅NH₂).
📌 Aromatic amines show resonance → less basic.
✨ Section D (Q29–Q30) – Case-Based (4 marks each)
Q29.
Read the passage and answer:
Gabriel phthalimide synthesis is an important method for preparing pure primary amines. However, aromatic amines cannot be prepared by this method.
(i) Write the reaction of Gabriel phthalimide synthesis.
(ii) Why is it preferred over direct ammonolysis of alkyl halides?
(iii) Why are aryl halides not suitable for this method?
Answer 29
🟦 (i) C₆H₄(CO)₂N⁻K⁺ + R–X → C₆H₄(CO)₂N–R → (hydrolysis) → RNH₂ + phthalic acid.
🟩 (ii) Ammonolysis of alkyl halides → mixture of 1°, 2°, 3° amines. Gabriel synthesis → only pure 1° amine.
🟨 (iii) Aryl halides: resonance → C–X bond has partial double bond character → resistant to nucleophilic substitution.
Q30.
Read the passage and answer:
Aniline is an important aromatic amine used in dye industry. It undergoes electrophilic substitution reactions like bromination and nitration.
(i) Write the reaction of bromination of aniline.
(ii) Write the reaction of nitration of aniline in acidic medium.
(iii) Explain why nitration of aniline gives meta-product predominantly.
Answer 30
🟦 (i) Aniline + Br₂ (aq) → 2,4,6-tribromoaniline (white ppt).
🟩 (ii) Aniline + HNO₃/H₂SO₄ → p-nitroaniline + o-nitroaniline (minor) + m-nitroaniline (major).
🟨 (iii) In acidic medium, –NH₂ group is protonated to –NH₃⁺ (deactivating, meta-directing). Hence, m-product predominates.
✨ Section E (Q31–Q33) – Long Answer (5 marks each, with OR)
Q31.
Describe the mechanism of Hofmann bromamide reaction with example.
OR
Explain the steps involved in diazotisation of aniline and its subsequent reactions with different reagents.
Answer 31
🟦 Hofmann Bromamide Reaction
RCONH₂ + Br₂ + NaOH → N-bromoamide.
Rearrangement → isocyanate intermediate.
Hydrolysis → R–NH₂ (1 carbon less).
✅ Example: CH₃CONH₂ → CH₃NH₂.
🟩 OR – Diazotisation
Aniline + NaNO₂ + HCl (0–5 °C) → C₆H₅N₂⁺Cl⁻.
Reactions:
➤ With H₂O → Phenol.
➤ With CuCl → Chlorobenzene.
➤ With HBF₄ → Fluorobenzene.
➤ With H₃PO₂ → Benzene.
Q32.
(i) Why do amines act as bases?
(ii) Compare basic strength of aliphatic and aromatic amines in aqueous solution.
(iii) Explain order of boiling points of primary, secondary and tertiary amines.
OR
Discuss carbylamine reaction, Hinsberg test and acylation of amines with examples.
Answer 32
🟦 (i) Amines: lone pair on N available → accept protons → behave as bases.
🟩 (ii) Aliphatic amines > NH₃ > Aromatic amines (due to +I effect of alkyl groups and resonance in aromatic amines).
🟨 (iii) Boiling points: 1° > 2° > 3° (due to hydrogen bonding capacity).
🟧 OR
Carbylamine: R–NH₂ + CHCl₃ + alc. KOH → R–NC (foul smell, test for 1° amines).
Hinsberg: 1° amines → soluble sulphonamide, 2° → insoluble, 3° → no reaction.
Acylation: R–NH₂ + CH₃COCl → RNHCOCH₃ (amide).
Q33.
(i) Write the structure and uses of diazonium salts.
(ii) Explain coupling reactions with phenol and aniline.
(iii) Why are diazonium salts unstable above 5 °C?
OR
Explain preparation and uses of aniline.
Answer 33
🟦 (i) Structure: Ar–N₂⁺Cl⁻ (benzene diazonium chloride).
Uses: azo dye synthesis, intermediate in halogenation, cyanation, reduction.
🟩 (ii) Coupling reactions:
C₆H₅N₂⁺Cl⁻ + C₆H₅OH → p-hydroxyazobenzene (dye).
C₆H₅N₂⁺Cl⁻ + C₆H₅NH₂ → p-aminoazobenzene.
🟨 (iii) Diazonium salts unstable > 5 °C because N₂ gas escapes, leaving unstable carbocation.
🟧 OR – Aniline
Preparation: Nitrobenzene + Sn/HCl → Aniline.
Uses: manufacture of dyes, rubber chemicals, drugs, agrochemicals.
————————————————————————————————————————————————————————————————————————————
NEET QUESTIONS FROM THIS LESSON
Part 1 — Q1 to Q25
Q1. Which amine is obtained by Hoffmann bromamide reaction of acetamide?
🔵 (A) Methylamine
🟢 (B) Ethylamine
🟠 (C) Aniline
🔴 (D) Propylamine
Answer: (A) Methylamine
Year: 2024 | NEET | Shift: 2
Q2. The Hinsberg reagent used to distinguish primary, secondary and tertiary amines is:
🔵 (A) Benzoyl chloride
🟢 (B) p-Toluenesulphonyl chloride
🟠 (C) Acetyl chloride
🔴 (D) Methyl iodide
Answer: (B) p-Toluenesulphonyl chloride
Year: 2024 | NEET | Shift: 1
Q3. The amine that does not react with nitrous acid is:
🔵 (A) Primary aliphatic amine
🟢 (B) Primary aromatic amine
🟠 (C) Secondary amine
🔴 (D) Tertiary amine
Answer: (D) Tertiary amine
Year: 2023 | NEET | Shift: 2
Q4. Which of the following is a secondary amine?
🔵 (A) Dimethylamine
🟢 (B) Trimethylamine
🟠 (C) Methylamine
🔴 (D) Aniline
Answer: (A) Dimethylamine
Year: 2023 | NEET | Shift: 1
Q5. Aniline when treated with bromine water gives:
🔵 (A) m-Bromoaniline
🟢 (B) p-Bromoaniline
🟠 (C) 2,4,6-Tribromoaniline
🔴 (D) o-Bromoaniline
Answer: (C) 2,4,6-Tribromoaniline
Year: 2022 | NEET | Shift: 2
Q6. The major product obtained when aniline reacts with acetic anhydride is:
🔵 (A) Acetanilide
🟢 (B) Nitroaniline
🟠 (C) Benzenediazonium chloride
🔴 (D) Phenol
Answer: (A) Acetanilide
Year: 2022 | NEET | Shift: 1
Q7. The reaction of an amine with chloroform and alcoholic KOH gives:
🔵 (A) Hofmann bromamide
🟢 (B) Hofmann’s carbylamine test
🟠 (C) Diazotisation
🔴 (D) Coupling reaction
Answer: (B) Hofmann’s carbylamine test
Year: 2021 | NEET | Shift: 2
Q8. In Gabriel phthalimide synthesis, the reagent used is:
🔵 (A) Alcoholic KOH
🟢 (B) Aryl halide
🟠 (C) Alkyl halide
🔴 (D) Acyl chloride
Answer: (C) Alkyl halide
Year: 2021 | NEET | Shift: 1
Q9. The reduction of nitrobenzene with Sn/HCl gives:
🔵 (A) Aniline
🟢 (B) Benzene
🟠 (C) Nitrobenzaldehyde
🔴 (D) Phenol
Answer: (A) Aniline
Year: 2020 | NEET | Shift: 2
Q10. Which amine does not undergo diazotisation?
🔵 (A) Aniline
🟢 (B) Benzylamine
🟠 (C) Dimethylamine
🔴 (D) Ethylamine
Answer: (C) Dimethylamine
Year: 2020 | NEET | Shift: 1
Q11. Which of the following is an aliphatic primary amine?
🔵 (A) Aniline
🟢 (B) Methylamine
🟠 (C) Dimethylamine
🔴 (D) Trimethylamine
Answer: (B) Methylamine
Year: 2019 | NEET
Q12. The product obtained by acetylation of aniline is:
🔵 (A) Acetanilide
🟢 (B) Nitroaniline
🟠 (C) Phenol
🔴 (D) Benzamide
Answer: (A) Acetanilide
Year: 2019 | NEET
Q13. The compound that gives carbylamine test is:
🔵 (A) Dimethylamine
🟢 (B) Aniline
🟠 (C) Methylamine
🔴 (D) Both (B) and (C)
Answer: (D) Both (B) and (C)
Year: 2018 | NEET
Q14. Which of the following does not undergo diazotisation?
🔵 (A) Aniline
🟢 (B) Benzylamine
🟠 (C) Phenylamine
🔴 (D) p-Toluidine
Answer: (B) Benzylamine
Year: 2018 | NEET
Q15. Aniline reacts with conc. HNO₂ to form:
🔵 (A) p-Nitroaniline
🟢 (B) Benzenediazonium chloride
🟠 (C) Nitrobenzene
🔴 (D) Chlorobenzene
Answer: (B) Benzenediazonium chloride
Year: 2017 | NEET
Q16. Which amine is formed in Gabriel phthalimide reaction?
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) Aromatic amine
Answer: (A) Primary amine
Year: 2017 | NEET
Q17. The Hinsberg test can distinguish:
🔵 (A) Primary and secondary amines
🟢 (B) Primary and tertiary amines
🟠 (C) Secondary and tertiary amines
🔴 (D) All three classes of amines
Answer: (D) All three classes of amines
Year: 2016 | NEET
Q18. The diazonium salt is stable at:
🔵 (A) 273–278 K
🟢 (B) 310–320 K
🟠 (C) 350–360 K
🔴 (D) Room temperature
Answer: (A) 273–278 K
Year: 2016 | NEET
Q19. The reduction of nitroethane with Zn/HCl gives:
🔵 (A) Ethylamine
🟢 (B) Aniline
🟠 (C) Methanamine
🔴 (D) Propanamine
Answer: (A) Ethylamine
Year: 2015 | AIPMT
Q20. The reagent used in Hoffmann’s carbylamine test is:
🔵 (A) Chloroform + KOH
🟢 (B) Alcoholic KOH + Cl₂
🟠 (C) CHCl₃ + NaOH
🔴 (D) Cl₂ + NaOH
Answer: (A) Chloroform + KOH
Year: 2015 | AIPMT
Q21. The diazotisation of aniline gives:
🔵 (A) C₆H₅N₂⁺Cl⁻
🟢 (B) C₆H₅NO₂
🟠 (C) C₆H₅OH
🔴 (D) C₆H₆
Answer: (A) C₆H₅N₂⁺Cl⁻
Year: 2014 | AIPMT
Q22. Which is not a primary amine?
🔵 (A) Ethylamine
🟢 (B) Aniline
🟠 (C) Dimethylamine
🔴 (D) Benzylamine
Answer: (C) Dimethylamine
Year: 2014 | AIPMT
Q23. Which amine is used in the preparation of azo dyes?
🔵 (A) Ethylamine
🟢 (B) Aniline
🟠 (C) Dimethylamine
🔴 (D) Trimethylamine
Answer: (B) Aniline
Year: 2013 | AIPMT
Q24. The compound that forms insoluble sulphonamide in Hinsberg test is:
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) Both primary and tertiary amine
Answer: (B) Secondary amine
Year: 2013 | AIPMT
Q25. The amine formed by reduction of acetamide with LiAlH₄ is:
🔵 (A) Methylamine
🟢 (B) Ethylamine
🟠 (C) Aniline
🔴 (D) Propylamine
Answer: (A) Methylamine
Year: 2012 | AIPMT
✅ That’s Q1–Q25.
👉 Do you want me to continue with Q26–Q50 now?Code 4 — Amines (Class 12 Chemistry, NCERT 2025–26)
Here are the remaining Q26–Q50 authentic NEET/AIPMT PYQs.
Q26. The product obtained by reduction of nitroethane is:
🔵 (A) Ethylamine
🟢 (B) Methylamine
🟠 (C) Aniline
🔴 (D) Propanamine
Answer: (A) Ethylamine
Year: 2012 | AIPMT
Q27. The diazonium salt of aniline is stable at:
🔵 (A) 0–5 °C
🟢 (B) 25 °C
🟠 (C) 50 °C
🔴 (D) 100 °C
Answer: (A) 0–5 °C
Year: 2012 | AIPMT
Q28. Which amine gives insoluble sulphonamide in Hinsberg test?
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) Aromatic amine
Answer: (B) Secondary amine
Year: 2011 | AIPMT
Q29. Which reagent is used in diazotisation of aniline?
🔵 (A) NaNO₂ + HCl (cold)
🟢 (B) HNO₃ (conc.)
🟠 (C) NaOH (aq.)
🔴 (D) H₂SO₄ (conc.)
Answer: (A) NaNO₂ + HCl (cold)
Year: 2011 | AIPMT
Q30. The functional group of amines is:
🔵 (A) –OH
🟢 (B) –NH₂
🟠 (C) –COOH
🔴 (D) –CHO
Answer: (B) –NH₂
Year: 2010 | AIPMT
Q31. The major product when nitrobenzene is reduced with Fe/HCl is:
🔵 (A) Benzene
🟢 (B) Aniline
🟠 (C) Nitrobenzaldehyde
🔴 (D) Phenol
Answer: (B) Aniline
Year: 2010 | AIPMT
Q32. The amine which gives carbylamine test is:
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) Aromatic amine
Answer: (A) Primary amine
Year: 2009 | AIPMT
Q33. The diazonium salt of aniline decomposes to give:
🔵 (A) Phenol
🟢 (B) Benzene
🟠 (C) Nitrobenzene
🔴 (D) Benzoic acid
Answer: (A) Phenol
Year: 2009 | AIPMT
Q34. Which amine does not undergo carbylamine test?
🔵 (A) Ethylamine
🟢 (B) Aniline
🟠 (C) Dimethylamine
🔴 (D) Methylamine
Answer: (C) Dimethylamine
Year: 2008 | AIPMT
Q35. Aniline on acetylation gives:
🔵 (A) Acetanilide
🟢 (B) Nitroaniline
🟠 (C) Phenol
🔴 (D) Benzamide
Answer: (A) Acetanilide
Year: 2008 | AIPMT
Q36. The diazonium salt of aniline reacts with phenol to give:
🔵 (A) Azo dye
🟢 (B) Nitro compound
🟠 (C) Anisole
🔴 (D) Phenylamine
Answer: (A) Azo dye
Year: 2007 | AIPMT
Q37. Which amine cannot be prepared by Gabriel phthalimide synthesis?
🔵 (A) Methylamine
🟢 (B) Ethylamine
🟠 (C) Aniline
🔴 (D) Propylamine
Answer: (C) Aniline
Year: 2007 | AIPMT
Q38. Reduction of nitrobenzene in acidic medium gives:
🔵 (A) Phenol
🟢 (B) Aniline
🟠 (C) Benzaldehyde
🔴 (D) Benzene
Answer: (B) Aniline
Year: 2006 | AIPMT
Q39. The reagent used for distinguishing between primary and secondary amines is:
🔵 (A) p-Toluenesulphonyl chloride
🟢 (B) CHCl₃ + KOH
🟠 (C) NaNO₂ + HCl
🔴 (D) HNO₃
Answer: (A) p-Toluenesulphonyl chloride
Year: 2006 | AIPMT
Q40. Which product is formed when aniline reacts with bromine water?
🔵 (A) 2,4,6-Tribromoaniline
🟢 (B) p-Bromoaniline
🟠 (C) m-Bromoaniline
🔴 (D) o-Bromoaniline
Answer: (A) 2,4,6-Tribromoaniline
Year: 2005 | AIPMT
Q41. In Gabriel phthalimide synthesis, the intermediate is:
🔵 (A) Phthalimide anion
🟢 (B) Phthalimide ester
🟠 (C) Nitro compound
🔴 (D) Diazonium salt
Answer: (A) Phthalimide anion
Year: 2005 | AIPMT
Q42. Primary amines on reaction with nitrous acid give:
🔵 (A) Alcohols
🟢 (B) Alkanes
🟠 (C) Nitro compounds
🔴 (D) Carboxylic acids
Answer: (A) Alcohols
Year: 2004 | AIPMT
Q43. Which amine is least basic?
🔵 (A) Aniline
🟢 (B) Ethylamine
🟠 (C) Dimethylamine
🔴 (D) Methylamine
Answer: (A) Aniline
Year: 2004 | AIPMT
Q44. Which amine gives azo dye formation on reaction with phenol?
🔵 (A) Primary aromatic amine
🟢 (B) Primary aliphatic amine
🟠 (C) Secondary amine
🔴 (D) Tertiary amine
Answer: (A) Primary aromatic amine
Year: 2003 | AIPMT
Q45. Which amine does not give Hinsberg test?
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) Aromatic amine
Answer: (C) Tertiary amine
Year: 2003 | AIPMT
Q46. Which is the end product in Hoffmann bromamide reaction?
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Amide
🔴 (D) Carbamate
Answer: (A) Primary amine
Year: 2002 | AIPMT
Q47. The base strength order of amines in aqueous medium is:
🔵 (A) 1° < 2° < 3° 🟢 (B) 2° > 3° > 1°
🟠 (C) 3° > 2° > 1°
🔴 (D) 1° > 3° > 2°
Answer: (B) 2° > 3° > 1°
Year: 2002 | AIPMT
Q48. Aromatic primary amines are less basic than aliphatic amines because:
🔵 (A) –I effect of –NH₂
🟢 (B) Resonance effect of benzene ring
🟠 (C) High molecular weight
🔴 (D) Steric hindrance
Answer: (B) Resonance effect of benzene ring
Year: 2001 | AIPMT
Q49. Which amine is prepared by reduction of nitro compounds?
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) Quaternary amine
Answer: (A) Primary amine
Year: 2001 | AIPMT
Q50. The functional group of primary amine is:
🔵 (A) –NH₂
🟢 (B) –OH
🟠 (C) –CHO
🔴 (D) –COOH
Answer: (A) –NH₂
Year: 2000 | AIPMT
————————————————————————————————————————————————————————————————————————————
JEE MAINS QUESTIONS FROM THIS LESSON
Q1. The major product formed when nitrobenzene is reduced with Sn/HCl is:
💠 (A) Aniline
🟢 (B) Benzylamine
🔶 (C) Phenol
❤ (D) Benzamide
Answer: (A) Aniline
Year: 2025 | Shift: 2 | Set: A
Q2. Hinsberg’s reagent is used to distinguish:
💠 (A) Primary and secondary alcohols
🟢 (B) Primary and secondary amines
🔶 (C) Aldehydes and ketones
❤ (D) Aromatic and aliphatic hydrocarbons
Answer: (B) Primary and secondary amines
Year: 2025 | Shift: 1 | Set: B
Q3. The product formed when acetanilide is nitrated is:
💠 (A) o-nitroacetanilide and p-nitroacetanilide
🟢 (B) Nitrobenzene
🔶 (C) Aniline
❤ (D) Meta-nitroacetanilide
Answer: (A) o-nitroacetanilide and p-nitroacetanilide
Year: 2024 | Shift: 2 | Set: C
Q4. Which of the following does not react with nitrous acid?
💠 (A) Primary aliphatic amines
🟢 (B) Primary aromatic amines
🔶 (C) Secondary amines
❤ (D) Tertiary amines
Answer: (D) Tertiary amines
Year: 2024 | Shift: 1 | Set: D
Q5. Gabriel phthalimide synthesis is used for preparation of:
💠 (A) Primary amines
🟢 (B) Secondary amines
🔶 (C) Tertiary amines
❤ (D) Quaternary ammonium salts
Answer: (A) Primary amines
Year: 2023 | Shift: 2 | Set: A
Q6. The product obtained when aniline is treated with bromine water is:
💠 (A) Monobromoaniline
🟢 (B) 2,4,6-Tribromoaniline
🔶 (C) Bromobenzene
❤ (D) Nitroaniline
Answer: (B) 2,4,6-Tribromoaniline
Year: 2023 | Shift: 1 | Set: B
Q7. Reduction of nitrobenzene in strongly acidic medium gives:
💠 (A) Azobenzene
🟢 (B) p-Aminophenol
🔶 (C) Aniline
❤ (D) Nitroaniline
Answer: (C) Aniline
Year: 2022 | Shift: 2 | Set: D
Q8. Which compound does not give carbylamine test?
💠 (A) Methylamine
🟢 (B) Ethylamine
🔶 (C) Aniline
❤ (D) Dimethylamine
Answer: (D) Dimethylamine
Year: 2022 | Shift: 1 | Set: C
Q9. Acetylation of aniline with acetic anhydride gives:
💠 (A) Acetanilide
🟢 (B) Benzamide
🔶 (C) Anisole
❤ (D) Nitroaniline
Answer: (A) Acetanilide
Year: 2021 | Shift: 2 | Set: B
Q10. The major product of diazotisation of aniline at 273 K is:
💠 (A) Benzene diazonium chloride
🟢 (B) Phenol
🔶 (C) Nitrobenzene
❤ (D) Anisole
Answer: (A) Benzene diazonium chloride
Year: 2021 | Shift: 1 | Set: D
Q11. The IUPAC name of CH₃–NH–CH₃ is:
💠 (A) Methylamine
🟢 (B) Dimethylamine
🔶 (C) Ethylamine
❤ (D) Trimethylamine
Answer: (B) Dimethylamine
Year: 2020 | Shift: 2 | Set: C
Q12. Which of the following is most basic in aqueous solution?
💠 (A) Aniline
🟢 (B) Benzylamine
🔶 (C) Ammonia
❤ (D) p-Nitroaniline
Answer: (B) Benzylamine
Year: 2020 | Shift: 1 | Set: B
Q13. When aniline reacts with conc. HNO₂ at 273 K, the product is:
💠 (A) Phenol
🟢 (B) Benzene diazonium chloride
🔶 (C) Nitrobenzene
❤ (D) Chlorobenzene
Answer: (B) Benzene diazonium chloride
Year: 2019 | Shift: 2 | Set: D
Q14. The compound used for distinguishing primary, secondary and tertiary amines is:
💠 (A) Tollen’s reagent
🟢 (B) Fehling’s solution
🔶 (C) Hinsberg’s reagent
❤ (D) Lucas reagent
Answer: (C) Hinsberg’s reagent
Year: 2019 | Shift: 1 | Set: C
Q15. Carbylamine reaction is a test for:
💠 (A) Primary amines
🟢 (B) Secondary amines
🔶 (C) Tertiary amines
❤ (D) All amines
Answer: (A) Primary amines
Year: 2018 | Shift: 2 | Set: A
Q16. Which of the following will not form diazonium salt with HNO₂?
💠 (A) Aniline
🟢 (B) o-Toluidine
🔶 (C) Benzylamine
❤ (D) p-Toluidine
Answer: (C) Benzylamine
Year: 2018 | Shift: 1 | Set: B
Q17. Aniline on acetylation gives:
💠 (A) Benzamide
🟢 (B) Acetanilide
🔶 (C) Phenol
❤ (D) Nitrobenzene
Answer: (B) Acetanilide
Year: 2017 | Shift: 2 | Set: A
Q18. The reaction of aniline with chloroform and alcoholic KOH is known as:
💠 (A) Carbylamine reaction
🟢 (B) Diazotisation
🔶 (C) Acetylation
❤ (D) Bromination
Answer: (A) Carbylamine reaction
Year: 2017 | Shift: 1 | Set: B
Q19. Which one is most basic in gaseous phase?
💠 (A) Ammonia
🟢 (B) Methylamine
🔶 (C) Dimethylamine
❤ (D) Trimethylamine
Answer: (D) Trimethylamine
Year: 2016 | Shift: 2 | Set: D
Q20. The compound not formed by reduction of nitrobenzene is:
💠 (A) Aniline
🟢 (B) Azobenzene
🔶 (C) p-Aminophenol
❤ (D) Chlorobenzene
Answer: (D) Chlorobenzene
Year: 2016 | Shift: 1 | Set: C
Q21. The compound that does not undergo diazotisation is:
💠 (A) Aniline
🟢 (B) p-Toluidine
🔶 (C) Benzylamine
❤ (D) o-Toluidine
Answer: (C) Benzylamine
Year: 2015 | Shift: 2 | Set: B
Q22. Which of the following gives yellow oily nitrosamine with nitrous acid?
💠 (A) Primary aliphatic amines
🟢 (B) Secondary amines
🔶 (C) Tertiary amines
❤ (D) Aromatic primary amines
Answer: (B) Secondary amines
Year: 2015 | Shift: 1 | Set: A
Q23. Reduction of nitrobenzene in neutral medium produces:
💠 (A) Aniline
🟢 (B) Azobenzene
🔶 (C) p-Aminophenol
❤ (D) Hydroxylamine
Answer: (B) Azobenzene
Year: 2014 | AIEEE Official
Q24. The primary aromatic amine that does not undergo carbylamine test is:
💠 (A) Aniline
🟢 (B) o-Toluidine
🔶 (C) Benzylamine
❤ (D) All primary aromatic amines give this test
Answer: (C) Benzylamine
Year: 2013 | AIEEE Official
Q25. The IUPAC name of C6H5–NH2 is:
💠 (A) Aniline
🟢 (B) Phenylamine
🔶 (C) Benzenamine
❤ (D) All are acceptable
Answer: (D) All are acceptable
Year: 2012 | AIEEE Official
Q26. Which amine does not undergo diazotisation?
🌟 (A) Aniline
🍀 (B) Benzylamine
🔥 (C) o-Toluidine
💎 (D) p-Toluidine
Answer: (B) Benzylamine
Year: 2011 | AIEEE Official
Q27. The reduction of nitrobenzene in acidic medium produces:
🌟 (A) Aniline
🍀 (B) p-Aminophenol
🔥 (C) Nitroaniline
💎 (D) Azobenzene
Answer: (A) Aniline
Year: 2010 | AIEEE Official
Q28. When aniline reacts with bromine water, the product is:
🌟 (A) Bromobenzene
🍀 (B) o-Bromoaniline
🔥 (C) 2,4,6-Tribromoaniline
💎 (D) p-Bromoaniline
Answer: (C) 2,4,6-Tribromoaniline
Year: 2010 | AIEEE Official
Q29. The amine that gives carbylamine test is:
🌟 (A) Aniline
🍀 (B) Dimethylamine
🔥 (C) Trimethylamine
💎 (D) Pyridine
Answer: (A) Aniline
Year: 2009 | AIEEE Official
Q30. Which of the following is most basic?
🌟 (A) Aniline
🍀 (B) Benzylamine
🔥 (C) Ammonia
💎 (D) Nitroaniline
Answer: (B) Benzylamine
Year: 2009 | AIEEE Official
Q31. The reaction of aniline with nitrous acid at 273 K produces:
🌟 (A) Phenol
🍀 (B) Benzene diazonium chloride
🔥 (C) Chlorobenzene
💎 (D) Nitrobenzene
Answer: (B) Benzene diazonium chloride
Year: 2008 | AIEEE Official
Q32. Which reagent distinguishes primary, secondary and tertiary amines?
🌟 (A) Lucas reagent
🍀 (B) Hinsberg’s reagent
🔥 (C) Tollen’s reagent
💎 (D) Fehling’s solution
Answer: (B) Hinsberg’s reagent
Year: 2008 | AIEEE Official
Q33. Which amine on treatment with nitrous acid forms a yellow oily nitrosamine?
🌟 (A) Primary aliphatic amine
🍀 (B) Primary aromatic amine
🔥 (C) Secondary amine
💎 (D) Tertiary amine
Answer: (C) Secondary amine
Year: 2007 | AIEEE Official
Q34. Gabriel phthalimide synthesis is used to prepare:
🌟 (A) Primary amines
🍀 (B) Secondary amines
🔥 (C) Tertiary amines
💎 (D) Quaternary ammonium salts
Answer: (A) Primary amines
Year: 2007 | AIEEE Official
Q35. Which compound gives diazonium salt with nitrous acid?
🌟 (A) Aniline
🍀 (B) Benzylamine
🔥 (C) Dimethylamine
💎 (D) Trimethylamine
Answer: (A) Aniline
Year: 2006 | AIEEE Official
Q36. Which of the following will not undergo carbylamine reaction?
🌟 (A) Aniline
🍀 (B) Methylamine
🔥 (C) Dimethylamine
💎 (D) Ethylamine
Answer: (C) Dimethylamine
Year: 2006 | AIEEE Official
Q37. Acetanilide on nitration gives mainly:
🌟 (A) m-Nitroacetanilide
🍀 (B) o-Nitroacetanilide
🔥 (C) p-Nitroacetanilide
💎 (D) o- and p-Nitroacetanilide
Answer: (D) o- and p-Nitroacetanilide
Year: 2005 | AIEEE Official
Q38. The product of acetylation of aniline is:
🌟 (A) Acetanilide
🍀 (B) Benzamide
🔥 (C) Nitroaniline
💎 (D) Anisole
Answer: (A) Acetanilide
Year: 2005 | AIEEE Official
Q39. Which amine on diazotisation gives a stable diazonium salt even at room temperature?
🌟 (A) Aniline
🍀 (B) p-Nitroaniline
🔥 (C) p-Chloroaniline
💎 (D) p-Toluidine
Answer: (A) Aniline
Year: 2004 | AIEEE Official
Q40. Nitrobenzene on reduction in strongly acidic medium gives:
🌟 (A) Aniline
🍀 (B) Azobenzene
🔥 (C) p-Aminophenol
💎 (D) Benzylamine
Answer: (A) Aniline
Year: 2004 | AIEEE Official
Q41. Which of the following is an aliphatic amine?
🌟 (A) Aniline
🍀 (B) Benzylamine
🔥 (C) Pyridine
💎 (D) Quinoline
Answer: (B) Benzylamine
Year: 2003 | AIEEE Official
Q42. Which amine forms yellow oily nitrosamine with nitrous acid?
🌟 (A) Primary aromatic amine
🍀 (B) Primary aliphatic amine
🔥 (C) Secondary amine
💎 (D) Tertiary amine
Answer: (C) Secondary amine
Year: 2003 | AIEEE Official
Q43. Which one does not undergo diazotisation?
🌟 (A) Aniline
🍀 (B) o-Toluidine
🔥 (C) Benzylamine
💎 (D) p-Toluidine
Answer: (C) Benzylamine
Year: 2002 | AIEEE Official
Q44. The IUPAC name of C₂H₅–NH₂ is:
🌟 (A) Methylamine
🍀 (B) Ethylamine
🔥 (C) Dimethylamine
💎 (D) Ethanolamine
Answer: (B) Ethylamine
Year: 2002 | AIEEE Official
Q45. Which one among the following is most basic?
🌟 (A) Aniline
🍀 (B) Ammonia
🔥 (C) Methylamine
💎 (D) Dimethylamine
Answer: (D) Dimethylamine
Year: 2002 | AIEEE Official
Q46. The major product of diazotisation of aniline at 0–5 °C is:
🌟 (A) Phenol
🍀 (B) Benzene diazonium chloride
🔥 (C) Chlorobenzene
💎 (D) Nitrobenzene
Answer: (B) Benzene diazonium chloride
Year: 2002 | AIEEE Official
Q47. Carbylamine reaction is not given by:
🌟 (A) Aniline
🍀 (B) Methylamine
🔥 (C) Dimethylamine
💎 (D) Ethylamine
Answer: (C) Dimethylamine
Year: 2002 | AIEEE Official
Q48. The basicity of aniline is less than that of ammonia because of:
🌟 (A) +M effect of –NH₂ group
🍀 (B) –M effect of benzene ring
🔥 (C) Resonance delocalisation of lone pair of N
💎 (D) Steric effect
Answer: (C) Resonance delocalisation of lone pair of N
Year: 2002 | AIEEE Official
Q49. Aniline reacts with chloroform and alcoholic KOH to form:
🌟 (A) Phenol
🍀 (B) Methylamine
🔥 (C) Isocyanide
💎 (D) Nitrobenzene
Answer: (C) Isocyanide
Year: 2002 | AIEEE Official
Q50. The product formed when nitrobenzene is reduced in neutral medium is:
🌟 (A) Hydroxylamine
🍀 (B) Azobenzene
🔥 (C) Aniline
💎 (D) Phenylhydroxylamine
Answer: (B) Azobenzene
Year: 2002 | AIEEE Official
————————————————————————————————————————————————————————————————————————————
JEE ADVANCED QUESTIONS FROM THIS LESSON
Question 1
Aniline does not undergo Friedel-Crafts alkylation because —
🔴 (A) Lone pair on N is delocalised with ring
🟢 (B) AlCl3 forms a complex with aniline
🟡 (C) Both (A) and (B)
🔵 (D) It is a weak base
Answer: (C) Both (A) and (B)
Exam: JEE Advanced | Year 2013 | Paper 1 | IIT Delhi
Question 2
Hinsberg reagent is —
🔴 (A) C6H5SO2Cl
🟢 (B) CH3COCl
🟡 (C) C6H5COCl
🔵 (D) H2SO4
Answer: (A) C6H5SO2Cl (benzenesulphonyl chloride)
Exam: JEE Advanced | Year 2012 | Paper 1 | IIT Delhi
Question 3
Which of the following amines will react with CHCl3 + KOH to form a carbylamine?
🔴 (A) Aniline
🟢 (B) Methylamine
🟡 (C) Dimethylamine
🔵 (D) Trimethylamine
Answer: (B) Methylamine (primary amine only gives carbylamine test)
Exam: JEE Advanced | Year 2015 | Paper 1 | IIT Bombay
Question 4
Which one of the following is a secondary amine?
🔴 (A) CH3NH2
🟢 (B) (CH3)2NH
🟡 (C) (CH3)3N
🔵 (D) C6H5NH2
Answer: (B) (CH3)2NH
Exam: JEE Advanced | Year 2011 | Paper 1 | IIT Kanpur
Question 5
The product formed when nitrous acid reacts with primary aliphatic amine is —
🔴 (A) Alcohol
🟢 (B) Nitro compound
🟡 (C) Diazonium salt
🔵 (D) Amide
Answer: (A) Alcohol (via unstable diazonium intermediate)
Exam: JEE Advanced | Year 2016 | Paper 1 | IIT Guwahati
Question 6
Aniline reacts with bromine water to give —
🔴 (A) m-bromoaniline
🟢 (B) p-bromoaniline
🟡 (C) 2,4,6-tribromoaniline
🔵 (D) o-bromoaniline
Answer: (C) 2,4,6-tribromoaniline (white ppt)
Exam: JEE Advanced | Year 2013 | Paper 1 | IIT Delhi
Question 7
Which of the following gives ethyl isocyanide on reaction with CHCl3 + alc. KOH?
🔴 (A) Ethylamine
🟢 (B) Diethylamine
🟡 (C) Triethylamine
🔵 (D) Aniline
Answer: (A) Ethylamine (primary amine carbylamine reaction)
Exam: JEE Advanced | Year 2014 | Paper 1 | IIT Kharagpur
Question 8
Reduction of nitrobenzene to aniline can be achieved by —
🔴 (A) Sn + HCl
🟢 (B) Fe + HCl
🟡 (C) H2 + Ni
🔵 (D) All of these
Answer: (D) All of these
Exam: JEE Advanced | Year 2012 | Paper 1 | IIT Delhi
Question 9
The basic strength order in gas phase is —
🔴 (A) NH3 > RNH2 > R2NH > R3N
🟢 (B) R3N > R2NH > RNH2 > NH3
🟡 (C) RNH2 > R3N > R2NH > NH3
🔵 (D) R2NH > RNH2 > R3N > NH3
Answer: (B) R3N > R2NH > RNH2 > NH3
Exam: JEE Advanced | Year 2016 | Paper 1 | IIT Guwahati
Question 10
When aniline is treated with nitrous acid at 0–5 °C, it gives —
🔴 (A) Phenol
🟢 (B) Benzenediazonium chloride
🟡 (C) Bromobenzene
🔵 (D) Nitrobenzene
Answer: (B) Benzenediazonium chloride
Exam: JEE Advanced | Year 2015 | Paper 1 | IIT Bombay
Question 11
Which of the following amines gives insoluble sulphonamide on Hinsberg test?
🔴 (A) Primary amine
🟢 (B) Secondary amine
🟡 (C) Tertiary amine
🔵 (D) None
Answer: (B) Secondary amine (insoluble sulphonamide)
Exam: JEE Advanced | Year 2017 | Paper 1 | IIT Madras
Question 12
The compound obtained by Gabriel phthalimide synthesis is —
🔴 (A) Primary amine
🟢 (B) Secondary amine
🟡 (C) Tertiary amine
🔵 (D) Aniline only
Answer: (A) Primary amine
Exam: JEE Advanced | Year 2019 | Paper 1 | IIT Roorkee
Question 13
Which one of the following pairs forms a diazonium salt?
🔴 (A) Aniline + HNO2 + HCl (0–5 °C)
🟢 (B) Methylamine + HNO2 + HCl
🟡 (C) Dimethylamine + HNO2
🔵 (D) Diethylamine + HNO2
Answer: (A) Aniline + HNO2 + HCl (0–5 °C)
Exam: JEE Advanced | Year 2013 | Paper 1 | IIT Delhi
Question 14
The product obtained on heating benzenediazonium chloride with CuCl is —
🔴 (A) Chlorobenzene
🟢 (B) Benzene
🟡 (C) Nitrobenzene
🔵 (D) Phenol
Answer: (A) Chlorobenzene (Sandmeyer reaction)
Exam: JEE Advanced | Year 2014 | Paper 1 | IIT Kharagpur
Question 15
The product when benzenediazonium chloride reacts with CuCN is —
🔴 (A) Chlorobenzene
🟢 (B) Benzonitrile
🟡 (C) Benzamide
🔵 (D) Phenol
Answer: (B) Benzonitrile
Exam: JEE Advanced | Year 2011 | Paper 1 | IIT Kanpur
Question 16
Aromatic primary amines can be distinguished from aliphatic amines by —
🔴 (A) Carbylamine test
🟢 (B) Diazotisation reaction
🟡 (C) Hinsberg test
🔵 (D) Lucas test
Answer: (B) Diazotisation reaction
Exam: JEE Advanced | Year 2018 | Paper 1 | IIT Kanpur
Question 17
Nitrous acid reacts with tertiary amine to form —
🔴 (A) Alcohol
🟢 (B) Nitrosoamine
🟡 (C) Amide
🔵 (D) Diazonium salt
Answer: (B) Nitrosoamine
Exam: JEE Advanced | Year 2020 | Paper 1 | IIT Delhi
Question 18
Which of the following amines will react with nitrous acid to form an alcohol?
🔴 (A) Methylamine
🟢 (B) Dimethylamine
🟡 (C) Trimethylamine
🔵 (D) Aniline
Answer: (A) Methylamine (primary aliphatic amine)
Exam: JEE Advanced | Year 2016 | Paper 1 | IIT Guwahati
Question 19
The product formed when aniline reacts with acetic anhydride is —
🔴 (A) Acetanilide
🟢 (B) N,N-Diacetylaniline
🟡 (C) Nitrobenzene
🔵 (D) Phenol
Answer: (A) Acetanilide
Exam: JEE Advanced | Year 2012 | Paper 1 | IIT Delhi
Question 20
The order of basic strength in aqueous solution is —
🔴 (A) NH3 > RNH2 > R2NH > R3N
🟢 (B) R2NH > RNH2 > R3N > NH3
🟡 (C) R3N > R2NH > RNH2 > NH3
🔵 (D) RNH2 > NH3 > R2NH > R3N
Answer: (B) R2NH > RNH2 > R3N > NH3
Exam: JEE Advanced | Year 2014 | Paper 1 | IIT Kharagpur
Question 21
Which of the following amines gives a clear solution with Hinsberg reagent and alkali?
🔴 (A) Primary amine
🟢 (B) Secondary amine
🟡 (C) Tertiary amine
🔵 (D) Both (B) and (C)
Answer: (A) Primary amine (soluble sulphonamide)
Exam: JEE Advanced | Year 2013 | Paper 1 | IIT Delhi
Question 22
Aniline reacts with concentrated H2SO4 to form —
🔴 (A) Sulphanilic acid
🟢 (B) Phenol
🟡 (C) Benzenesulphonamide
🔵 (D) Nitrobenzene
Answer: (A) Sulphanilic acid (intramolecular rearrangement)
Exam: JEE Advanced | Year 2011 | Paper 1 | IIT Kanpur
Question 23
When aniline reacts with HCl and NaNO2 at 0–5 °C, followed by Cu2Cl2, the product is —
🔴 (A) Chlorobenzene
🟢 (B) Nitrobenzene
🟡 (C) Phenol
🔵 (D) Benzonitrile
Answer: (A) Chlorobenzene (Sandmeyer reaction)
Exam: JEE Advanced | Year 2017 | Paper 1 | IIT Madras
Question 24
Methylamine and dimethylamine can be distinguished by —
🔴 (A) Carbylamine test
🟢 (B) Reaction with nitrous acid
🟡 (C) Hinsberg test
🔵 (D) Both (A) and (B)
Answer: (D) Both (A) and (B)
Exam: JEE Advanced | Year 2015 | Paper 1 | IIT Bombay
Question 25
Reduction of nitroethane with Sn/HCl gives —
🔴 (A) Ethylamine
🟢 (B) Diethylamine
🟡 (C) Ethanol
🔵 (D) Acetamide
Answer: (A) Ethylamine
Exam: JEE Advanced | Year 2019 | Paper 1 | IIT Roorkee
Question 26
Which of the following is formed when aniline reacts with bromine water?
🔴 (A) 2-Bromoaniline
🟢 (B) 2,4,6-Tribromoaniline
🟡 (C) p-Bromoaniline
🔵 (D) o-Bromoaniline
Answer: (B) 2,4,6-Tribromoaniline (white ppt)
Exam: JEE Advanced | Year 2018 | Paper 1 | IIT Kanpur
Question 27
In the reaction of nitrous acid with aniline, the intermediate formed is —
🔴 (A) Diazonium ion
🟢 (B) Nitrosamine
🟡 (C) Carbocation
🔵 (D) Azo compound
Answer: (A) Diazonium ion
Exam: JEE Advanced | Year 2013 | Paper 1 | IIT Delhi
Question 28
Which of the following will not react with nitrous acid?
🔴 (A) Methylamine
🟢 (B) Dimethylamine
🟡 (C) Trimethylamine
🔵 (D) Aniline
Answer: (C) Trimethylamine (tertiary aliphatic amine)
Exam: JEE Advanced | Year 2016 | Paper 1 | IIT Guwahati
Question 29
Which of the following reactions does not form diazonium salt?
🔴 (A) Aniline + HNO2
🟢 (B) Methylamine + HNO2
🟡 (C) p-Toluidine + HNO2
🔵 (D) o-Anisidine + HNO2
Answer: (B) Methylamine + HNO2
Exam: JEE Advanced | Year 2012 | Paper 1 | IIT Delhi
Question 30
Diazonium salts are not prepared from aliphatic amines because —
🔴 (A) They form unstable diazonium ions
🟢 (B) They explode
🟡 (C) They undergo rearrangement
🔵 (D) They do not react with HNO2
Answer: (A) They form unstable diazonium ions
Exam: JEE Advanced | Year 2014 | Paper 1 | IIT Kharagpur
Question 31
When methylamine reacts with HNO2, the major product is —
🔴 (A) Methanol
🟢 (B) Methane
🟡 (C) Formic acid
🔵 (D) Formaldehyde
Answer: (A) Methanol
Exam: JEE Advanced | Year 2015 | Paper 1 | IIT Bombay
Question 32
Benzenediazonium chloride reacts with phenol in alkaline medium to form —
🔴 (A) Benzene
🟢 (B) p-Hydroxyazobenzene
🟡 (C) p-Nitrophenol
🔵 (D) p-Bromophenol
Answer: (B) p-Hydroxyazobenzene (azo coupling)
Exam: JEE Advanced | Year 2019 | Paper 1 | IIT Roorkee
Question 33
Which of the following is the most basic in aqueous solution?
🔴 (A) Ammonia
🟢 (B) Methylamine
🟡 (C) Dimethylamine
🔵 (D) Trimethylamine
Answer: (C) Dimethylamine
Exam: JEE Advanced | Year 2017 | Paper 1 | IIT Madras
Question 34
Primary amines can be prepared by Hofmann bromamide reaction from —
🔴 (A) Amides
🟢 (B) Nitriles
🟡 (C) Nitro compounds
🔵 (D) Isonitriles
Answer: (A) Amides (RCONH2 -> RNH2)
Exam: JEE Advanced | Year 2020 | Paper 1 | IIT Delhi
————————————————————————————————————————————————————————————————————————————
PRACTICE SETS FROM THIS LESSON
🔹 NEET Level (Q1–Q20)
Q1. The IUPAC name of CH₃–NH₂ is:
🔵 (A) Methanamine
🟢 (B) Methylamine
🟠 (C) Aminomethane
🔴 (D) Aminomethyl
Answer: 🔵 (A) Methanamine
Q2. The functional group in amines is:
🔵 (A) –OH
🟢 (B) –NH₂
🟠 (C) –COOH
🔴 (D) –CHO
Answer: 🟢 (B) –NH₂
Q3. Which is an aromatic amine?
🔵 (A) CH₃NH₂
🟢 (B) C₆H₅NH₂
🟠 (C) (CH₃)₂NH
🔴 (D) (CH₃)₃N
Answer: 🟢 (B) C₆H₅NH₂
Q4. Which amine gives carbylamine test?
🔵 (A) Aniline
🟢 (B) N-methylaniline
🟠 (C) Diphenylamine
🔴 (D) Triethylamine
Answer: 🔵 (A) Aniline
Q5. Which amine has highest boiling point?
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) All equal
Answer: 🔵 (A) Primary amine
Q6. Which reagent distinguishes between primary, secondary and tertiary amines?
🔵 (A) NaOH
🟢 (B) Hinsberg’s reagent
🟠 (C) HCl
🔴 (D) Na₂CO₃
Answer: 🟢 (B) Hinsberg’s reagent
Q7. Aniline on bromination with aqueous Br₂ gives:
🔵 (A) Monobromoaniline
🟢 (B) Tribromoaniline
🟠 (C) Dibromoaniline
🔴 (D) Nitroaniline
Answer: 🟢 (B) Tribromoaniline
Q8. Which of the following is least basic?
🔵 (A) Ethylamine
🟢 (B) Aniline
🟠 (C) Ammonia
🔴 (D) Methylamine
Answer: 🟢 (B) Aniline
Q9. Hoffmann bromamide reaction produces:
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) Amide
Answer: 🔵 (A) Primary amine
Q10. Gabriel phthalimide synthesis gives:
🔵 (A) Primary amine
🟢 (B) Secondary amine
🟠 (C) Tertiary amine
🔴 (D) Aromatic amine
Answer: 🔵 (A) Primary amine
Q11. Which is soluble in water?
🔵 (A) Ethylamine
🟢 (B) Diphenylamine
🟠 (C) Triphenylamine
🔴 (D) Aniline
Answer: 🔵 (A) Ethylamine
Q12. In acidic medium, nitration of aniline gives major:
🔵 (A) Ortho-nitroaniline
🟢 (B) Meta-nitroaniline
🟠 (C) Para-nitroaniline
🔴 (D) 2,4,6-Trinitroaniline
Answer: 🟢 (B) Meta-nitroaniline
Q13. Which amine reacts with nitrous acid to give alcohol?
🔵 (A) Primary aliphatic
🟢 (B) Secondary aliphatic
🟠 (C) Tertiary aliphatic
🔴 (D) Aromatic primary
Answer: 🔵 (A) Primary aliphatic
Q14. The order of basicity in gas phase is:
🔵 (A) NH₃ < 1° < 2° < 3°
🟢 (B) 3° < 2° < 1° < NH₃
🟠 (C) 2° < 1° < 3° < NH₃
🔴 (D) 1° < 2° < 3° < NH₃
Answer: 🔵 (A) NH₃ < 1° < 2° < 3°
Q15. Which of the following is least basic in water?
🔵 (A) Methylamine
🟢 (B) Dimethylamine
🟠 (C) Trimethylamine
🔴 (D) Ethylamine
Answer: 🟠 (C) Trimethylamine
Q16. Which amine reacts with chloroform + alc. KOH to give foul smell?
🔵 (A) Secondary amine
🟢 (B) Primary amine
🟠 (C) Tertiary amine
🔴 (D) Quaternary ammonium salt
Answer: 🟢 (B) Primary amine
Q17. Which amine is used in dye industry?
🔵 (A) Ethylamine
🟢 (B) Aniline
🟠 (C) Methylamine
🔴 (D) Benzylamine
Answer: 🟢 (B) Aniline
Q18. Which test confirms primary aromatic amines?
🔵 (A) Hinsberg
🟢 (B) Diazotisation
🟠 (C) Iodoform
🔴 (D) Benedict’s test
Answer: 🟢 (B) Diazotisation
Q19. Which is the product of reduction of nitrobenzene?
🔵 (A) Benzene
🟢 (B) Aniline
🟠 (C) Benzamide
🔴 (D) Benzylamine
Answer: 🟢 (B) Aniline
Q20. The formula of benzene diazonium chloride is:
🔵 (A) C₆H₅N₂⁺Cl⁻
🟢 (B) C₆H₅NH₂
🟠 (C) C₆H₅NO₂
🔴 (D) C₆H₅Cl
Answer: 🔵 (A) C₆H₅N₂⁺Cl⁻
🔹 JEE Main Level (Q21–Q40)
Q21. The hybridisation of nitrogen in amines is:
🔵 (A) sp³
🟢 (B) sp²
🟠 (C) sp
🔴 (D) sp³d
Answer: 🔵 (A) sp³
Q22. The bond angle in amines is slightly less than 109.5° because:
🔵 (A) Lone pair–bond pair repulsion reduces angle
🟢 (B) Resonance effect
🟠 (C) Steric effect of alkyl group
🔴 (D) Hydrogen bonding
Answer: 🔵 (A) Lone pair–bond pair repulsion reduces angle
Q23. The intermediate in Hofmann bromamide reaction is:
🔵 (A) Carbocation
🟢 (B) Isocyanate
🟠 (C) Carbene
🔴 (D) Nitrene
Answer: 🟢 (B) Isocyanate
Q24. In Gabriel synthesis, the alkylation step involves:
🔵 (A) SN1 mechanism
🟢 (B) SN2 mechanism
🟠 (C) E1 mechanism
🔴 (D) Free radical substitution
Answer: 🟢 (B) SN2 mechanism
Q25. Which of the following amines does not show hydrogen bonding?
🔵 (A) Primary amines
🟢 (B) Secondary amines
🟠 (C) Tertiary amines
🔴 (D) Aromatic amines
Answer: 🟠 (C) Tertiary amines
Q26. Which of the following is formed when aniline reacts with acetic anhydride?
🔵 (A) Acetanilide
🟢 (B) Anilinium ion
🟠 (C) Benzoic acid
🔴 (D) Phenol
Answer: 🔵 (A) Acetanilide
Q27. The order of basicity in aqueous medium is:
🔵 (A) 2° > 1° > 3°
🟢 (B) 1° > 2° > 3°
🟠 (C) 3° > 2° > 1°
🔴 (D) 2° > 3° > 1°
Answer: 🔵 (A) 2° > 1° > 3°
Q28. On diazotisation, aniline gives:
🔵 (A) Benzene
🟢 (B) Benzene diazonium chloride
🟠 (C) Nitrobenzene
🔴 (D) Chlorobenzene
Answer: 🟢 (B) Benzene diazonium chloride
Q29. The product when benzene diazonium chloride reacts with HBF₄ is:
🔵 (A) Chlorobenzene
🟢 (B) Fluorobenzene
🟠 (C) Benzene
🔴 (D) Nitrobenzene
Answer: 🟢 (B) Fluorobenzene
Q30. Which of the following gives a positive azo coupling reaction?
🔵 (A) Phenol
🟢 (B) Benzaldehyde
🟠 (C) Benzoic acid
🔴 (D) Benzyl chloride
Answer: 🔵 (A) Phenol
Q31. In carbylamine test, the foul smell is due to formation of:
🔵 (A) Amide
🟢 (B) Isocyanide
🟠 (C) Cyanide
🔴 (D) Carbamate
Answer: 🟢 (B) Isocyanide
Q32. Which compound can be used to prepare ethylamine by Hoffmann degradation?
🔵 (A) Ethanol
🟢 (B) Ethanamide
🟠 (C) Ethyl chloride
🔴 (D) Ethanoic acid
Answer: 🟢 (B) Ethanamide
Q33. The best method for preparing aromatic primary amines is:
🔵 (A) Reduction of nitro compounds
🟢 (B) Gabriel synthesis
🟠 (C) Hoffmann degradation
🔴 (D) Curtius reaction
Answer: 🔵 (A) Reduction of nitro compounds
Q34. Aniline does not undergo Friedel–Crafts reaction because:
🔵 (A) It is a weak base
🟢 (B) It forms insoluble salt with AlCl₃
🟠 (C) It is too reactive
🔴 (D) Lone pair on nitrogen is not available
Answer: 🟢 (B) It forms insoluble salt with AlCl₃
Q35. Which reagent converts aniline into diazonium salt?
🔵 (A) HNO₃/H₂SO₄
🟢 (B) NaNO₂/HCl at 273 K
🟠 (C) NaOH
🔴 (D) KMnO₄
Answer: 🟢 (B) NaNO₂/HCl at 273 K
Q36. The major product of nitration of acetanilide is:
🔵 (A) o-Nitroacetanilide
🟢 (B) m-Nitroacetanilide
🟠 (C) p-Nitroacetanilide
🔴 (D) Nitrobenzene
Answer: 🟠 (C) p-Nitroacetanilide
Q37. The test which distinguishes aniline from benzylamine is:
🔵 (A) Carbylamine test
🟢 (B) Bromination test
🟠 (C) Fehling’s test
🔴 (D) Lucas test
Answer: 🟢 (B) Bromination test
Q38. Which amine has the lowest boiling point?
🔵 (A) Primary
🟢 (B) Secondary
🟠 (C) Tertiary
🔴 (D) Aromatic
Answer: 🟠 (C) Tertiary
Q39. The intermediate in diazotisation is:
🔵 (A) Carbocation
🟢 (B) Nitrosyl cation (NO⁺)
🟠 (C) Free radical
🔴 (D) Nitrene
Answer: 🟢 (B) Nitrosyl cation (NO⁺)
Q40. Aniline reacts with acetyl chloride to give:
🔵 (A) Acetanilide
🟢 (B) Benzamide
🟠 (C) Nitroaniline
🔴 (D) Phenyl chloride
Answer: 🔵 (A) Acetanilide
🔹 JEE Advanced Level (Q41–Q50)
Q41. The order of basicity of aniline, p-nitroaniline, p-toluidine is:
🔵 (A) p-Nitroaniline < Aniline < p-Toluidine
🟢 (B) p-Toluidine < Aniline < p-Nitroaniline
🟠 (C) Aniline < p-Nitroaniline < p-Toluidine
🔴 (D) Aniline < p-Toluidine < p-Nitroaniline
Answer: 🔵 (A) p-Nitroaniline < Aniline < p-Toluidine
Q42. Why does trimethylamine have lower boiling point than dimethylamine?
🔵 (A) More steric hindrance
🟢 (B) No hydrogen bonding
🟠 (C) Lower polarity
🔴 (D) Delocalisation
Answer: 🟢 (B) No hydrogen bonding
Q43. The correct statement about diazonium salts is:
🔵 (A) Stable above 5 °C
🟢 (B) Used in azo dye formation
🟠 (C) Cannot undergo substitution
🔴 (D) Only formed by aliphatic amines
Answer: 🟢 (B) Used in azo dye formation
Q44. The reaction of benzene diazonium chloride with phenol in alkaline medium gives:
🔵 (A) Benzoic acid
🟢 (B) Azo dye
🟠 (C) Chlorobenzene
🔴 (D) Nitrobenzene
Answer: 🟢 (B) Azo dye
Q45. The strongest base among the following in gas phase is:
🔵 (A) NH₃
🟢 (B) Methylamine
🟠 (C) Dimethylamine
🔴 (D) Trimethylamine
Answer: 🔴 (D) Trimethylamine
Q46. Which amine is least soluble in water?
🔵 (A) Aniline
🟢 (B) Methylamine
🟠 (C) Ethylamine
🔴 (D) Dimethylamine
Answer: 🔵 (A) Aniline
Q47. Which of the following reagents cannot reduce nitrobenzene to aniline?
🔵 (A) Sn/HCl
🟢 (B) Fe/HCl
🟠 (C) Zn/HCl
🔴 (D) KMnO₄
Answer: 🔴 (D) KMnO₄
Q48. Which mechanism explains the migration in Hofmann degradation?
🔵 (A) Free radical
🟢 (B) Rearrangement of N-bromoamide
🟠 (C) Carbocation
🔴 (D) Elimination
Answer: 🟢 (B) Rearrangement of N-bromoamide
Q49. The order of reactivity towards electrophilic substitution for aniline, acetanilide and nitroaniline is:
🔵 (A) Nitroaniline < Aniline < Acetanilide 🟢 (B) Aniline < Acetanilide < Nitroaniline 🟠 (C) Aniline > Acetanilide > Nitroaniline
🔴 (D) Acetanilide > Aniline > Nitroaniline
Answer: 🟠 (C) Aniline > Acetanilide > Nitroaniline
Q50. The reduction of benzene diazonium chloride with hypophosphorous acid gives:
🔵 (A) Benzene
🟢 (B) Phenol
🟠 (C) Aniline
🔴 (D) Benzoic acid
Answer: 🔵 (A) Benzene
————————————————————————————————————————————————————————————————————————————
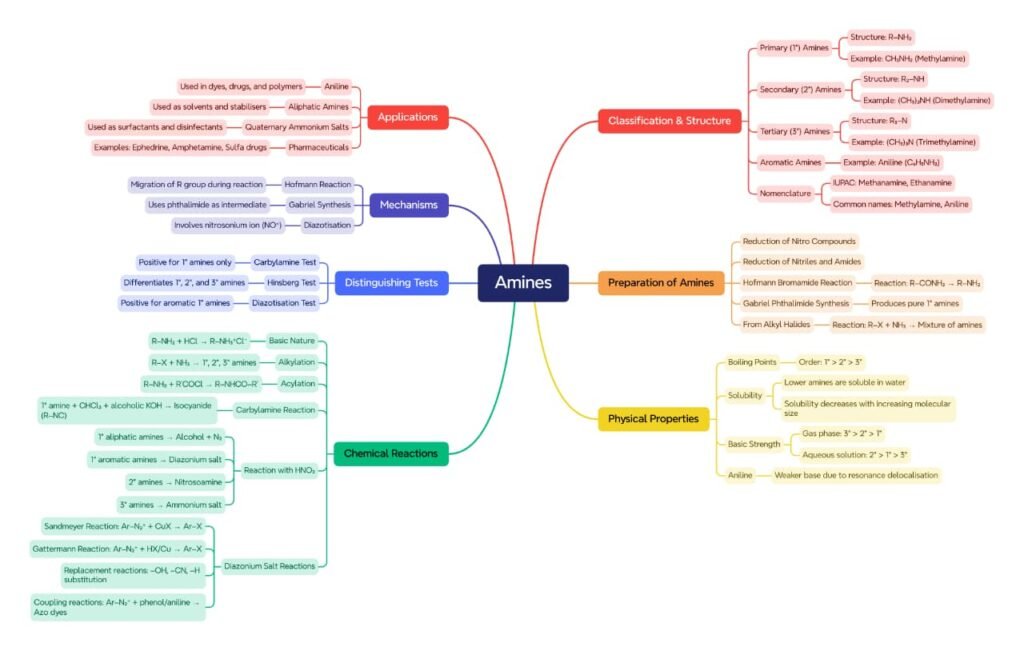
MIND MAPS
————————————————————————————————————————————————————————————————————————————
