Class 10 : Science (In English) – Lesson 1. Chemical Reactions and Equations
EXPLANATION AND SUMMARY
🌿 EXPLANATION
🔵 Introduction
Every day, we see chemical reactions taking place — burning of fuels, cooking of food, rusting of iron, and respiration in our body. A chemical reaction is a process in which one or more substances (reactants) change into new substances (products) with different properties. During these reactions, the atoms rearrange but are neither created nor destroyed, following the Law of Conservation of Mass.
🟢 1. What is a Chemical Reaction?
When substances interact chemically, new products form with properties different from the reactants.
💡 Example: When magnesium ribbon burns in air:
Mg + O₂ → 2MgO
✔️ Magnesium (reactant) combines with oxygen (reactant) to form magnesium oxide (product).
🔴 2. Characteristics of Chemical Reactions
You can identify a chemical reaction by several observable signs:
✨ Change in State: Solid, liquid, or gas form may change.
✨ Change in Colour: Fe + CuSO₄ → FeSO₄ + Cu (blue turns green)
✨ Gas Evolution: Zn + H₂SO₄ → ZnSO₄ + H₂↑
✨ Change in Temperature: CaO + H₂O → Ca(OH)₂ + heat (exothermic)
✨ Precipitate Formation: BaCl₂ + H₂SO₄ → BaSO₄↓ + 2HCl
✏️ Note: Physical changes (like melting or freezing) do not form new substances, but chemical changes always do.
🟡 3. Chemical Equations
A chemical equation is the symbolic representation of a reaction using formulas and symbols.
Word Equation: Hydrogen + Oxygen → Water
Symbolic Equation: H₂ + O₂ → H₂O
💡 Key Idea: Equations must be balanced to obey the Law of Conservation of Mass, meaning the total number of atoms on both sides must be equal.
🔵 4. Balancing Chemical Equations
To balance an equation, ensure equal numbers of atoms of each element appear on both sides.
Example: Fe + H₂O → Fe₃O₄ + H₂
Step 1: Count atoms.
Step 2: Balance iron → 3Fe + H₂O → Fe₃O₄ + H₂
Step 3: Balance oxygen and hydrogen → 3Fe + 4H₂O → Fe₃O₄ + 4H₂
✔️ Balanced Equation: 3Fe + 4H₂O → Fe₃O₄ + 4H₂
🟢 5. Types of Chemical Reactions
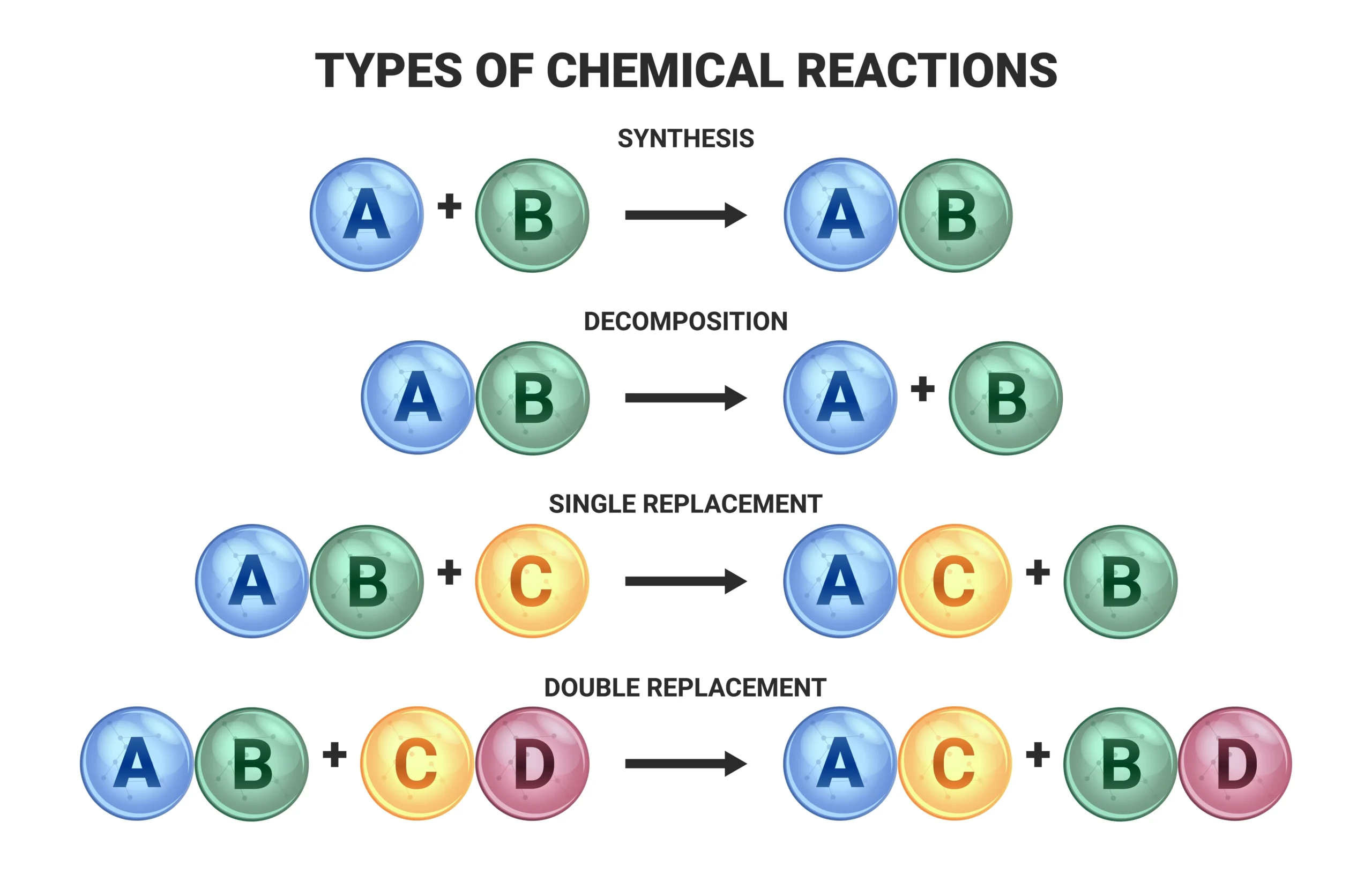
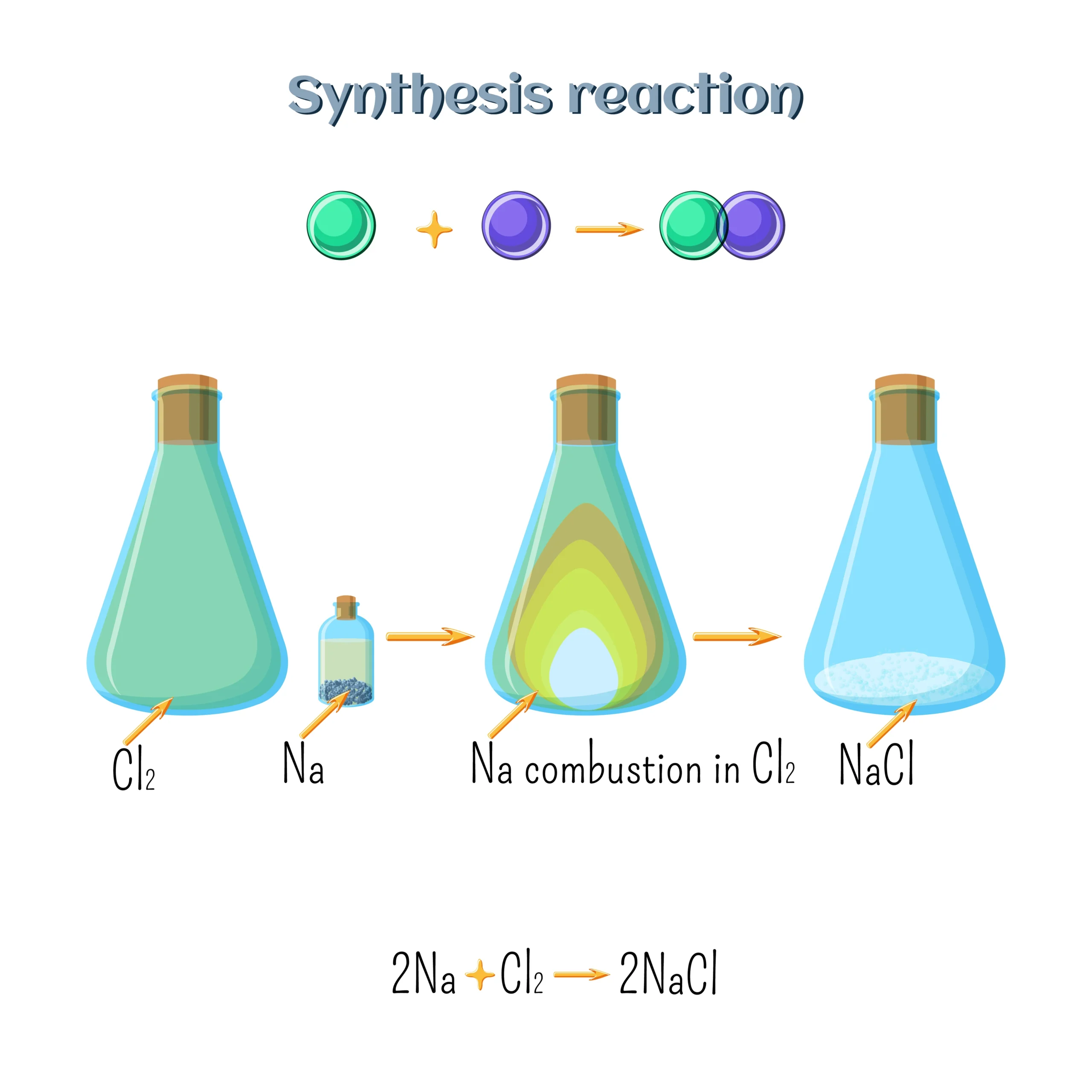
(a) Combination Reaction ➡️ Two or more substances combine to form a single product.
Example: CaO + H₂O → Ca(OH)₂
✔️ Usually exothermic (releases heat).
(b) Decomposition Reaction ➡️ One compound breaks down into simpler substances.
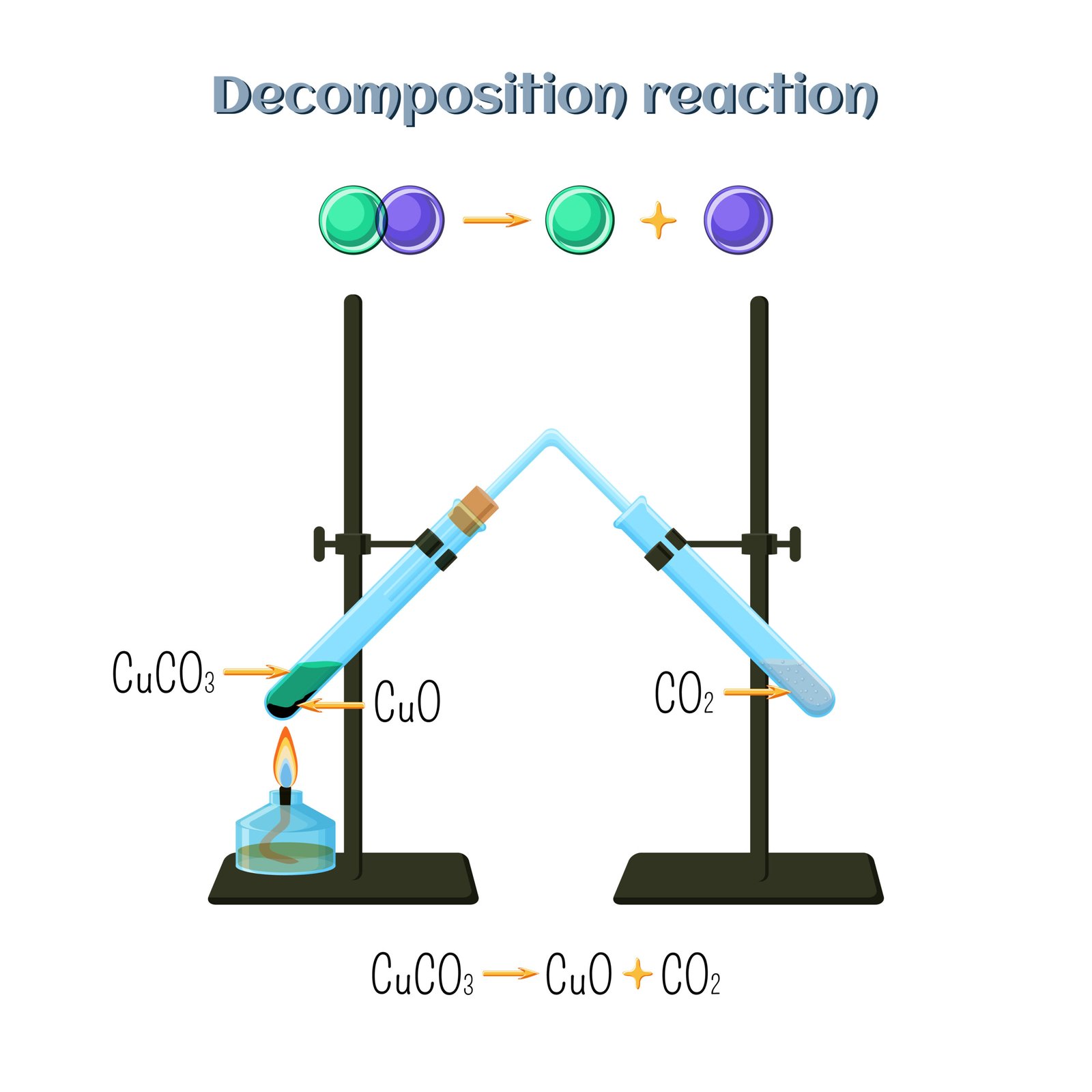
Thermal: CaCO₃ → CaO + CO₂
Electrolytic: 2H₂O → 2H₂ + O₂
Photolytic: 2AgCl → 2Ag + Cl₂ (in sunlight)
✏️ Used in photography.
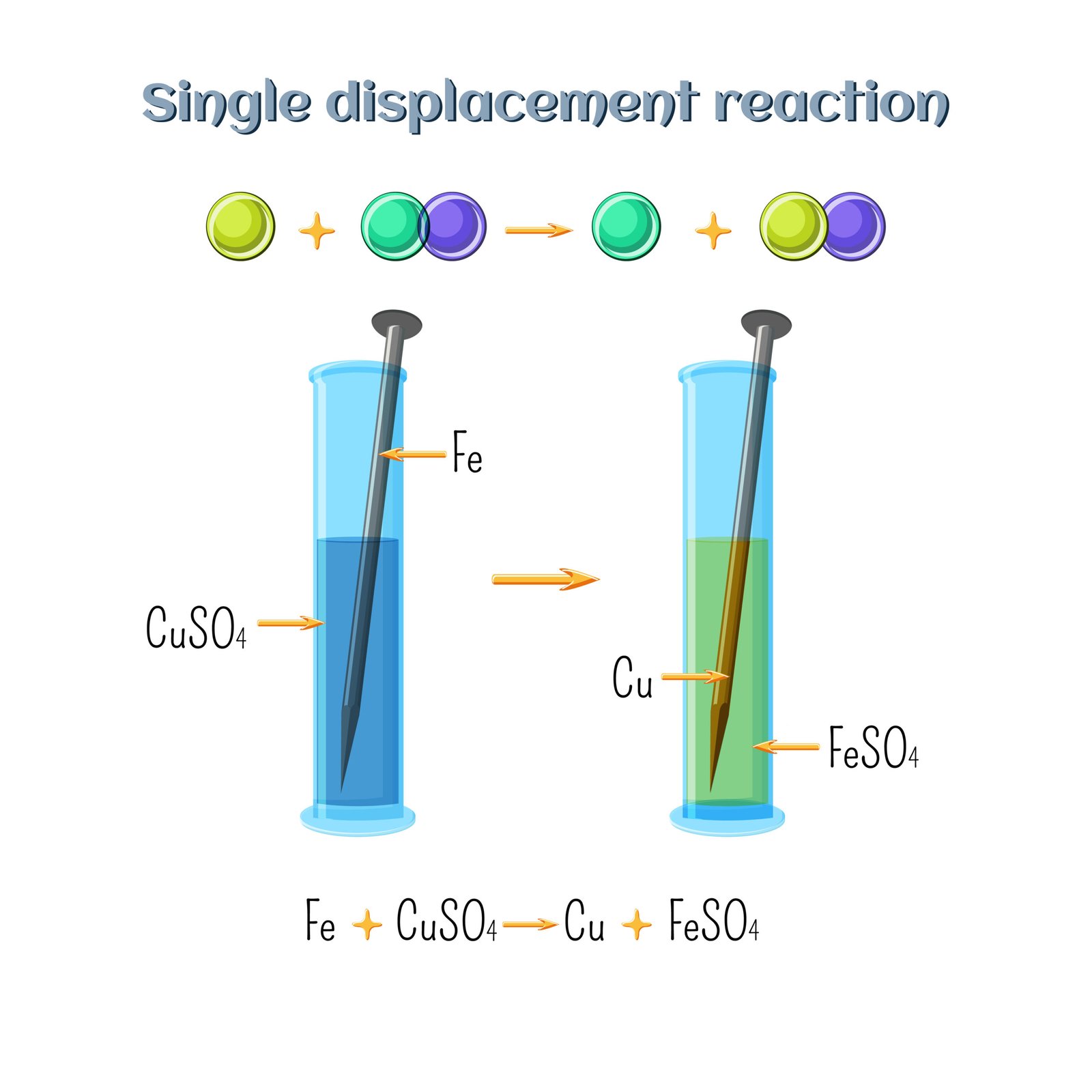
(c) Displacement Reaction ➡️ A more reactive element replaces a less reactive one.
Example: Zn + CuSO₄ → ZnSO₄ + Cu
(d) Double Displacement Reaction ➡️ Exchange of ions between two compounds.
Example: BaCl₂ + Na₂SO₄ → 2NaCl + BaSO₄↓
(e) Redox Reaction ➡️ Both oxidation and reduction occur simultaneously.
Example: ZnO + C → Zn + CO
🔴 6. Oxidation and Reduction
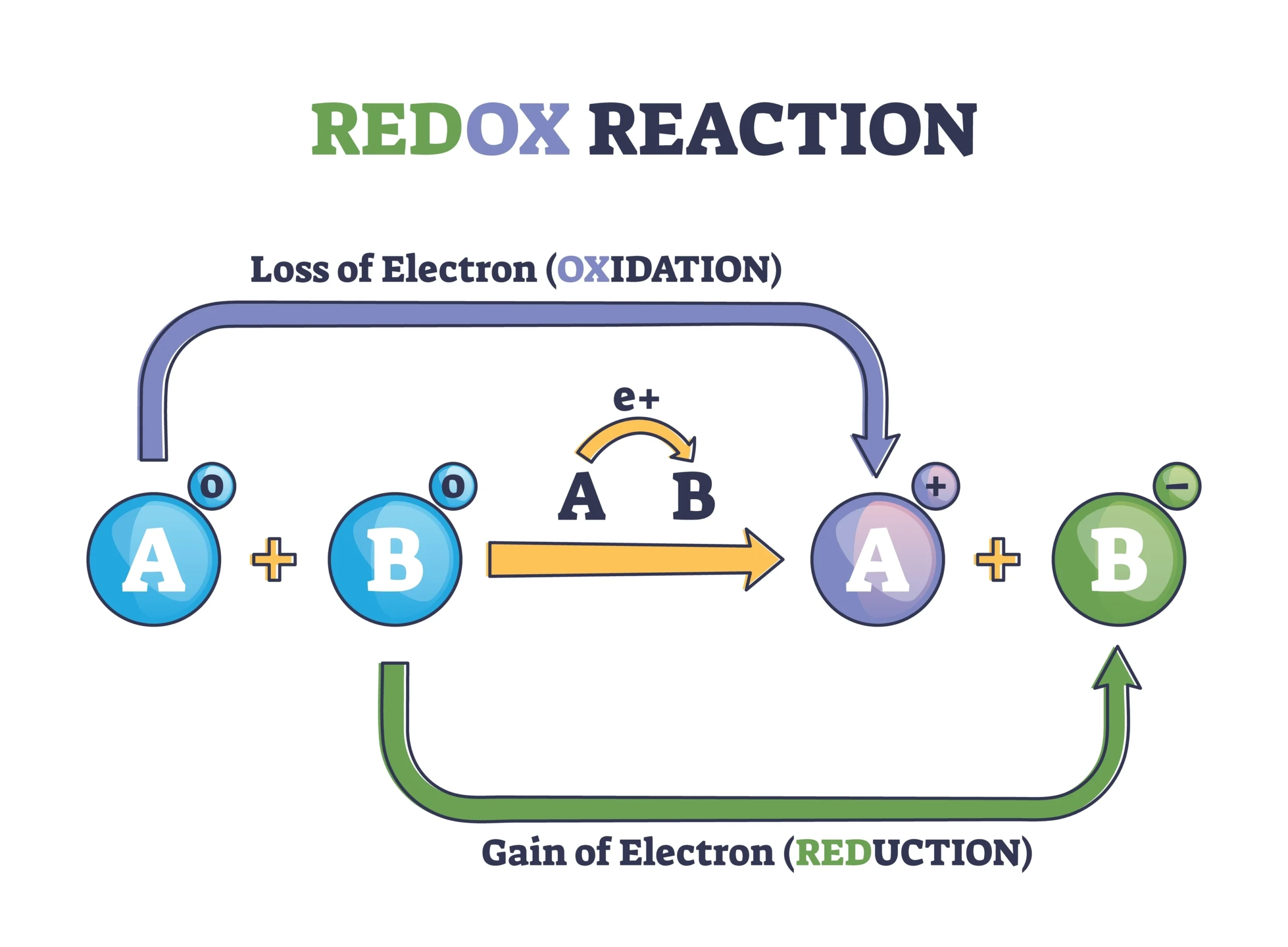
Oxidation: Addition of oxygen or removal of hydrogen.
Reduction: Addition of hydrogen or removal of oxygen.
💡 Example: CuO + H₂ → Cu + H₂O
✔️ CuO gets reduced, H₂ gets oxidized.
Oxidizing agent → causes oxidation.
Reducing agent → causes reduction.
🟡 7. Effects of Oxidation in Daily Life
(a) Corrosion: Metals react with air and moisture, forming oxides that damage the surface.
Example: 4Fe + 3O₂ + 6H₂O → 4Fe(OH)₃ → Fe₂O₃·xH₂O (rust)
✔️ Prevention: painting, oiling, galvanizing, electroplating, alloying.
(b) Rancidity: Oils and fats oxidize when exposed to air, causing bad odor and taste.
✔️ Prevention: airtight storage, antioxidants, refrigeration, nitrogen flushing.
🔵 8. Exothermic and Endothermic Reactions
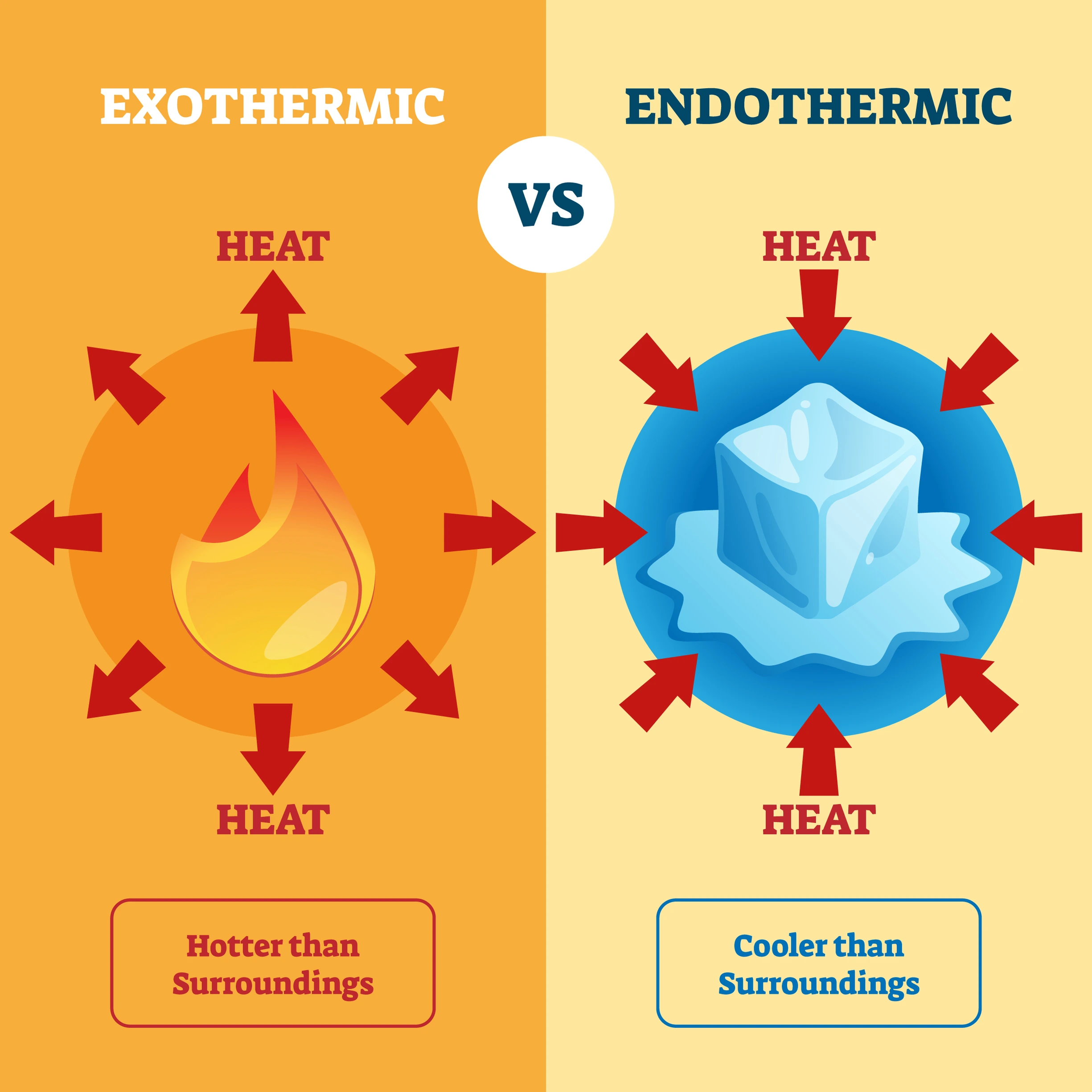
Exothermic: Heat is released. Example: CaO + H₂O → Ca(OH)₂ + heat
Endothermic: Heat is absorbed. Example: N₂ + O₂ → 2NO
✔️ Exothermic reactions increase temperature; endothermic reactions lower it.
🟢 9. Importance of Balancing
✔️ Ensures the Law of Conservation of Mass is followed.
✔️ Allows correct calculation of reactant and product quantities.
✔️ Essential for industrial chemical safety and efficiency.
🔴 10. Real-Life Importance of Chemical Reactions
✔️ Respiration → oxidation of glucose for energy.
✔️ Photosynthesis → plants produce food using CO₂ and sunlight.
✔️ Combustion → fuels release energy.
✔️ Digestion → breakdown of food into simpler molecules.
✔️ Industry → used in making fertilizers, soaps, medicines, and plastics.
🟡 11. Practice Example (Balancing)
Hydrogen sulphide burns in air to form sulphur dioxide and water.
Unbalanced: H₂S + O₂ → SO₂ + H₂O
Balanced: 2H₂S + 3O₂ → 2SO₂ + 2H₂O
✔️ Balanced on both sides (H, S, O all equal).
🔵 12. Practice Example (Reaction Types)
2KClO₃ → 2KCl + 3O₂ → Decomposition (thermal)
Zn + CuSO₄ → ZnSO₄ + Cu → Displacement
H₂ + Cl₂ → 2HCl → Combination
AgNO₃ + NaCl → AgCl + NaNO₃ → Double Displacement
🟢 13. Everyday Applications
🌿 Rusting → Oxidation of iron in moisture.
🌿 Blackening of silver ornaments → Formation of silver sulphide.
🌿 Burning of LPG → Exothermic oxidation.
🌿 Photosynthesis → Reduction of CO₂ to glucose.
🌿 Respiration → Oxidation of glucose releasing energy.
🔴 14. Key Concepts
✔️ Chemical equations follow the Law of Conservation of Mass.
✔️ Every oxidation is accompanied by a reduction.
✔️ Balancing ensures accurate chemical calculations.
✔️ Corrosion and rancidity are real-life oxidation effects.
✔️ Reactions power life processes and industries.
✳️ SUMMARY
Chemical reactions transform reactants into new products with unique properties. Observable signs include change in color, gas evolution, temperature variation, and precipitate formation. Equations are balanced to conserve mass. The main types are Combination, Decomposition, Displacement, Double Displacement, and Redox. Oxidation means gain of oxygen or loss of hydrogen; reduction means gain of hydrogen or loss of oxygen. Examples like corrosion and rancidity show oxidation in daily life. Balancing ensures safety, correctness, and predictability in all chemical processes.
📝 QUICK RECAP
1️⃣ Chemical reaction = reactants → products (new properties)
2️⃣ Indicators → color change, gas release, heat, precipitate, state change
3️⃣ Balancing → ensures conservation of mass
4️⃣ Reaction types → Combination, Decomposition, Displacement, Double Displacement, Redox
5️⃣ Oxidation = +O₂ or −H₂; Reduction = +H₂ or −O₂
6️⃣ Real-life examples → rusting, rancidity, respiration, photosynthesis
————————————————————————————————————————————————————————————————————————–
QUESTIONS FROM TEXTBOOK:-
🔵 Question
- Which of the statements about the reaction below are incorrect?
2PbO(s) + C(s) → 2Pb(s) + CO₂(g)
(a) Lead is getting reduced.
(b) Carbon dioxide is getting oxidised.
(c) Carbon is getting oxidised.
(d) Lead oxide is getting reduced.
(i) (a) and (b)
(ii) (a) and (c)
(iii) (a), (b) and (c)
(iv) all
🟢 Answer
💡 First, understand reduction / oxidation in this reaction:
➡️ 2PbO(s) + C(s) → 2Pb(s) + CO₂(g)
🔵 Step 1: Observe PbO → Pb
🟢 Lead oxide (PbO) loses oxygen to form lead (Pb).
🟢 Losing oxygen = reduction.
✔️ So PbO is reduced to Pb.
🔵 Step 2: Observe C → CO₂
🟡 Carbon combines with oxygen to form CO₂.
🟡 Gaining oxygen = oxidation.
✔️ So carbon is oxidised to CO₂.
🔵 Step 3: Is CO₂ getting oxidised?
🔴 No. CO₂ is already the oxidised product. It is not getting further oxidised here.
Now check each statement:
(a) “Lead is getting reduced.”
✏️ Here, lead (Pb) is produced from PbO. Pb itself is not reduced — PbO is reduced.
🔴 So statement (a) is incorrect.
(b) “Carbon dioxide is getting oxidised.”
🔴 CO₂ is formed; it is not being oxidised further.
🔴 So statement (b) is incorrect.
(c) “Carbon is getting oxidised.”
✔️ This is TRUE (carbon → CO₂ by gain of oxygen).
(d) “Lead oxide is getting reduced.”
✔️ This is TRUE (PbO loses oxygen and becomes Pb).
🧠 So the INCORRECT statements are (a) and (b).
✔️ Final: The correct choice is
(i) (a) and (b).
✔️ Final Answer: (i)
🔵 Question
2. Fe₂O₃ + 2Al → Al₂O₃ + 2Fe
The above reaction is an example of a
(a) combination reaction.
(b) double displacement reaction.
(c) decomposition reaction.
(d) displacement reaction.
🟢 Answer
💡 Concept: In a displacement reaction, a more reactive element displaces a less reactive element from its compound.
🔵 Step 1: Compare reactivity.
🟢 Aluminium (Al) is more reactive than iron (Fe).
🔵 Step 2: Look at the reaction.
➡️ Fe₂O₃ + 2Al → Al₂O₃ + 2Fe
🟡 Aluminium removes (displaces) iron from iron(III) oxide.
🔵 Step 3: Identify the reaction type.
🧠 Iron (Fe) is set free.
🧠 Aluminium takes oxygen and forms aluminium oxide (Al₂O₃).
➡️ This is a displacement / redox process known as thermite reaction.
✔️ Final Answer: (d) displacement reaction.
✏️ Note: It is also known as a highly exothermic reaction used in welding, but in terms of NCERT classification, it is displacement.
🔵 Question
3. What happens when dilute hydrochloric acid is added to iron fillings? Tick the correct answer.
(a) Hydrogen gas and iron chloride are produced.
(b) Chlorine gas and iron hydroxide are produced.
(c) No reaction takes place.
(d) Iron salt and water are produced.
🟢 Answer
💡 Reaction between metal (iron) and dilute acid (HCl):
➡️ Metal + dilute acid → Salt + Hydrogen gas
🔵 Step 1: Write the reaction.
➡️ Fe + 2HCl → FeCl₂ + H₂↑
🔵 Step 2: Identify products.
🟢 FeCl₂ = iron(II) chloride (an iron salt).
🟢 H₂ = hydrogen gas.
🔵 Step 3: Match with options.
(a) says “Hydrogen gas and iron chloride are produced.”
✔️ This matches FeCl₂ (iron chloride) + H₂.
(d) says “Iron salt and water are produced.”
🔴 Water is not produced here.
(b) is wrong: no chlorine gas is produced.
(c) is wrong: reaction clearly happens.
✔️ Final Answer: (a) Hydrogen gas and iron chloride are produced.
✔️ Also write the balanced equation: Fe + 2HCl → FeCl₂ + H₂
🔵 Question
4. What is a balanced chemical equation? Why should chemical equations be balanced?
🟢 Answer
💡 Concept:
🔵 A balanced chemical equation is a chemical equation in which the number of atoms of each element is the same on both sides of the equation (reactant side = product side).
🟢 Reason it must be balanced:
🔵 Step 1: According to the law of conservation of mass,
➡️ mass can neither be created nor destroyed in a chemical reaction.
🔵 Step 2: If atoms on the left and right are not equal, mass would not “match”.
🔵 Step 3: Therefore we adjust the coefficients to make both sides equal.
✔️ Final: We balance equations so that they obey the law of conservation of mass.
✏️ Note: Changing only the numbers in front of formulas (coefficients) is allowed. Changing the actual formula is NOT allowed.
🔵 Question
5. Translate the following statements into chemical equations and then balance them.
(a) Hydrogen gas combines with nitrogen to form ammonia.
(b) Hydrogen sulphide gas burns in air to give water and sulphur dioxide.
(c) Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate.
(d) Potassium metal reacts with water to give potassium hydroxide and hydrogen gas.
🟢 Answer
💡 Concept: We will first write the word equation as a skeletal equation, then balance it.
(a) Hydrogen gas combines with nitrogen to form ammonia.
🔵 Step 1: Skeletal equation → H₂ + N₂ → NH₃
🔵 Step 2: Balance nitrogen and hydrogen.
➡️ N₂ + 3H₂ → 2NH₃
✔️ Final balanced: N₂ + 3H₂ → 2NH₃
(b) Hydrogen sulphide gas burns in air to give water and sulphur dioxide.
🔵 Step 1: Skeletal equation → H₂S + O₂ → H₂O + SO₂
🔵 Step 2: Balance.
➡️ 2H₂S + 3O₂ → 2H₂O + 2SO₂
✔️ Final balanced: 2H₂S + 3O₂ → 2H₂O + 2SO₂
(c) Barium chloride reacts with aluminium sulphate to give aluminium chloride and a precipitate of barium sulphate.
🔵 Step 1: Skeletal equation → BaCl₂ + Al₂(SO₄)₃ → AlCl₃ + BaSO₄
🔵 Step 2: Balance carefully.
➡️ 3BaCl₂ + Al₂(SO₄)₃ → 2AlCl₃ + 3BaSO₄
✔️ Final balanced: 3BaCl₂ + Al₂(SO₄)₃ → 2AlCl₃ + 3BaSO₄
✏️ Note: BaSO₄ is an insoluble white precipitate. This is double displacement + precipitation.
(d) Potassium metal reacts with water to give potassium hydroxide and hydrogen gas.
🔵 Step 1: Skeletal equation → K + H₂O → KOH + H₂
🔵 Step 2: Balance.
➡️ 2K + 2H₂O → 2KOH + H₂
✔️ Final balanced: 2K + 2H₂O → 2KOH + H₂
✔️ Final summary for Q5:
🟢 (a) N₂ + 3H₂ → 2NH₃
🟢 (b) 2H₂S + 3O₂ → 2H₂O + 2SO₂
🟢 (c) 3BaCl₂ + Al₂(SO₄)₃ → 2AlCl₃ + 3BaSO₄
🟢 (d) 2K + 2H₂O → 2KOH + H₂
🔵 Question
6. Balance the following chemical equations.
(a) HNO₃ + Ca(OH)₂ → Ca(NO₃)₂ + H₂O
(b) NaOH + H₂SO₄ → Na₂SO₄ + H₂O
(c) NaCl + AgNO₃ → AgCl + NaNO₃
(d) BaCl₂ + H₂SO₄ → BaSO₄ + HCl
🟢 Answer
(a) HNO₃ + Ca(OH)₂ → Ca(NO₃)₂ + H₂O
🔵 Step 1: Check atoms.
➡️ Ca:1 both sides
➡️ N: left 1, right 2
➡️ O,H need balancing
🔵 Step 2: Put 2 in front of HNO₃ and 2 in front of H₂O.
➡️ 2HNO₃ + Ca(OH)₂ → Ca(NO₃)₂ + 2H₂O
✔️ Final balanced: 2HNO₃ + Ca(OH)₂ → Ca(NO₃)₂ + 2H₂O
(b) NaOH + H₂SO₄ → Na₂SO₄ + H₂O
🔵 Step 1: Sodium atoms. Need 2 Na on left.
➡️ 2NaOH + H₂SO₄ → Na₂SO₄ + H₂O
🔵 Step 2: Now check H and O.
Left H: (2 from 2NaOH) + 2 from H₂SO₄ = 4 total
Right H in H₂O is 2, so multiply H₂O by 2.
➡️ 2NaOH + H₂SO₄ → Na₂SO₄ + 2H₂O
✔️ Final balanced: 2NaOH + H₂SO₄ → Na₂SO₄ + 2H₂O
(c) NaCl + AgNO₃ → AgCl + NaNO₃
💡 This is already balanced.
➡️ 1 Na, 1 Cl, 1 Ag, 1 NO₃ on both sides.
✔️ Final balanced: NaCl + AgNO₃ → AgCl + NaNO₃
(d) BaCl₂ + H₂SO₄ → BaSO₄ + HCl
🔵 Step 1: Check atoms.
Left: Ba=1, Cl=2, H=2, S=1, O=4
Right: Ba=1, SO₄=1, HCl has 1 H and 1 Cl
🔵 Step 2: Need 2 HCl on the right.
➡️ BaCl₂ + H₂SO₄ → BaSO₄ + 2HCl
✔️ Final balanced: BaCl₂ + H₂SO₄ → BaSO₄ + 2HCl
🔵 Question
7. Write the balanced chemical equations for the following reactions.
(a) Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water
(b) Zinc + Silver nitrate → Zinc nitrate + Silver
(c) Aluminium + Copper chloride → Aluminium chloride + Copper
(d) Barium chloride + Potassium sulphate → Barium sulphate + Potassium chloride
🟢 Answer
💡 We will first write the skeletal equations, then balance each.
(a) Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water
🔵 Step 1: Skeletal equation
➡️ Ca(OH)₂ + CO₂ → CaCO₃ + H₂O
🔵 Step 2: Check atoms: Ca = 1, C = 1, O and H already balanced.
✔️ Final balanced equation: Ca(OH)₂ + CO₂ → CaCO₃ + H₂O
(b) Zinc + Silver nitrate → Zinc nitrate + Silver
🔵 Step 1: Skeletal equation
➡️ Zn + AgNO₃ → Zn(NO₃)₂ + Ag
🔵 Step 2: Nitrate group NO₃ appears twice on the right.
To balance, we need 2 AgNO₃ on the left and 2 Ag on the right.
➡️ Zn + 2AgNO₃ → Zn(NO₃)₂ + 2Ag
✔️ Final balanced equation: Zn + 2AgNO₃ → Zn(NO₃)₂ + 2Ag
✏️ Note: This is a displacement reaction. Zinc displaces silver.
(c) Aluminium + Copper chloride → Aluminium chloride + Copper
🔵 Step 1: Skeletal equation
➡️ Al + CuCl₂ → AlCl₃ + Cu
🔵 Step 2: Balance Cl and Al.
We need 2 Al atoms and 3 CuCl₂ to supply 6 Cl atoms.
Then we get 2AlCl₃ and 3Cu.
➡️ 2Al + 3CuCl₂ → 2AlCl₃ + 3Cu
✔️ Final balanced equation: 2Al + 3CuCl₂ → 2AlCl₃ + 3Cu
(d) Barium chloride + Potassium sulphate → Barium sulphate + Potassium chloride
🔵 Step 1: Skeletal equation
➡️ BaCl₂ + K₂SO₄ → BaSO₄ + KCl
🔵 Step 2: Balance K and Cl by making 2KCl.
➡️ BaCl₂ + K₂SO₄ → BaSO₄ + 2KCl
✔️ Final balanced equation: BaCl₂ + K₂SO₄ → BaSO₄ + 2KCl
✔️ Final summary for Q7:
🟢 (a) Ca(OH)₂ + CO₂ → CaCO₃ + H₂O
🟢 (b) Zn + 2AgNO₃ → Zn(NO₃)₂ + 2Ag
🟢 (c) 2Al + 3CuCl₂ → 2AlCl₃ + 3Cu
🟢 (d) BaCl₂ + K₂SO₄ → BaSO₄ + 2KCl
🔵 Question
8. Write the balanced chemical equation for the following and identify the type of reaction in each case.
(a) Potassium bromide(aq) + Barium iodide(aq) → Potassium iodide(aq) + Barium bromide(s)
(b) Zinc carbonate(s) → Zinc oxide(s) + Carbon dioxide(g)
(c) Hydrogen(g) + Chlorine(g) → Hydrogen chloride(g)
(d) Magnesium(s) + Hydrochloric acid(aq) → Magnesium chloride(aq) + Hydrogen(g)
🟢 Answer
(a) Potassium bromide(aq) + Barium iodide(aq) → Potassium iodide(aq) + Barium bromide(s)
🔵 Step 1: Skeletal equation
➡️ KBr(aq) + BaI₂(aq) → KI(aq) + BaBr₂(s)
🔵 Step 2: Balance K and I.
We need 2KI on the right and 2KBr on the left.
➡️ 2KBr(aq) + BaI₂(aq) → 2KI(aq) + BaBr₂(s)
🟡 Type of reaction: Double displacement + precipitation (BaBr₂ solid forms).
✔️ Final: 2KBr(aq) + BaI₂(aq) → 2KI(aq) + BaBr₂(s)
✔️ Reaction type: Double displacement / precipitation reaction
(b) Zinc carbonate(s) → Zinc oxide(s) + Carbon dioxide(g)
🔵 Step 1: Skeletal equation
➡️ ZnCO₃(s) → ZnO(s) + CO₂(g)
🔵 Step 2: Atoms are already balanced.
✔️ Final: ZnCO₃(s) → ZnO(s) + CO₂(g)
🟡 Reaction type: Thermal decomposition (heated zinc carbonate breaks down).
(c) Hydrogen(g) + Chlorine(g) → Hydrogen chloride(g)
🔵 Step 1: Skeletal equation
➡️ H₂(g) + Cl₂(g) → HCl(g)
🔵 Step 2: Balance by making 2HCl.
➡️ H₂(g) + Cl₂(g) → 2HCl(g)
🟡 Reaction type: Combination reaction (two elements combine to form a single product).
✔️ Final: H₂ + Cl₂ → 2HCl
✔️ Type: Combination
(d) Magnesium(s) + Hydrochloric acid(aq) → Magnesium chloride(aq) + Hydrogen(g)
🔵 Step 1: Skeletal equation
➡️ Mg(s) + HCl(aq) → MgCl₂(aq) + H₂(g)
🔵 Step 2: MgCl₂ needs 2Cl, so put 2HCl.
➡️ Mg + 2HCl → MgCl₂ + H₂
🟡 Reaction type: Displacement reaction (Mg displaces hydrogen).
✔️ Final: Mg + 2HCl → MgCl₂ + H₂
✔️ Type: Displacement reaction
🔵 Question
9. What does one mean by exothermic and endothermic reactions? Give examples.
🟢 Answer
💡 Concept: Energy (heat) change is an important sign of chemical change.
🔵 Exothermic reaction:
➡️ A reaction which releases heat energy to the surroundings.
✔️ Example: C + O₂ → CO₂ + heat
✔️ Example (in daily life): Burning of fuel like methane.
🟢 Endothermic reaction:
➡️ A reaction which absorbs heat energy from the surroundings.
✔️ Example: N₂ + O₂ → 2NO (at very high temperature)
✔️ Example: Photosynthesis (overall process absorbs energy from sunlight).
🟡 Summary:
🔵 Exothermic = heat out (hot).
🟢 Endothermic = heat in (cool).
🔵 Question
10. Why is respiration considered an exothermic reaction? Explain.
🟢 Answer
💡 Concept: Respiration is a chemical reaction inside cells.
🔵 Step 1: In respiration, glucose is oxidised.
➡️ Glucose + O₂ → CO₂ + H₂O + energy
🔵 Step 2: The energy released is in the form of heat.
🟢 This heat energy keeps our body warm and supports life processes.
✔️ Final: Respiration is an exothermic reaction because energy (heat) is released when food (glucose) is broken down during oxidation.
🔵 Question
11. Why are decomposition reactions called the opposite of combination reactions? Write equations for these reactions.
🟢 Answer
💡 Combination reaction:
🔵 Two or more substances combine to form a single product.
➡️ Example: CaO + H₂O → Ca(OH)₂
💡 Decomposition reaction:
🔵 A single compound breaks down into two or more simpler substances.
➡️ Example: 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂ (on heating)
🟡 Reasoning:
🔵 In combination, simpler substances join to form something more complex.
🟢 In decomposition, a complex substance splits into simpler substances.
✔️ Final: They are opposite processes.
🔵 Question
12. Write one equation each for decomposition reactions where energy is supplied in the form of heat, light or electricity.
🟢 Answer
💡 We will give 3 different kinds of decomposition reactions:
🔵 (i) Decomposition by heat (thermal decomposition):
➡️ CaCO₃ → CaO + CO₂
✔️ Heat causes calcium carbonate to form calcium oxide and carbon dioxide.
🔵 (ii) Decomposition by light (photochemical decomposition):
➡️ 2AgCl → 2Ag + Cl₂
✔️ Silver chloride breaks down in presence of sunlight.
🔵 (iii) Decomposition by electricity (electrolytic decomposition):
➡️ 2H₂O → 2H₂ + O₂
✔️ Passing electric current through acidified water splits it into hydrogen and oxygen.
✏️ Note: All three are decomposition reactions, but the energy source is different.
🔵 Question
13. What is the difference between displacement and double displacement reactions? Write equations for these reactions.
🟢 Answer
💡 Definitions first:
🔵 Displacement reaction:
➡️ A more reactive element displaces a less reactive element from its compound.
✔️ Example: Zn + CuSO₄ → ZnSO₄ + Cu
🟡 Here, Zn displaces Cu.
🔵 Double displacement reaction:
➡️ Two compounds exchange ions to form new compounds.
➡️ Often a precipitate is formed.
✔️ Example: BaCl₂ + Na₂SO₄ → BaSO₄ + 2NaCl
🟡 BaSO₄ is an insoluble precipitate.
✔️ Summary:
🔵 Displacement = one element replaces another.
🟢 Double displacement = ions are exchanged between two compounds.
🔵 Question
14. In the refining of silver, the recovery of silver from silver nitrate solution involved displacement by copper metal. Write down the reaction involved.
🟢 Answer
💡 Copper can displace silver because copper is more reactive than silver.
🔵 Step 1: Silver nitrate reacts with copper metal.
➡️ AgNO₃ + Cu → Cu(NO₃)₂ + Ag
🔵 Step 2: Balance the equation.
We need 2AgNO₃ to match Ag atoms.
➡️ 2AgNO₃ + Cu → Cu(NO₃)₂ + 2Ag
✔️ Final balanced equation:
✔️ 2AgNO₃ + Cu → Cu(NO₃)₂ + 2Ag
🟡 Reaction type: Displacement reaction (Cu displaces Ag).
🔵 Question
15. What do you mean by a precipitation reaction? Explain by giving examples.
🟢 Answer
💡 Concept:
🔵 A precipitation reaction is a reaction in which two aqueous solutions react to form an insoluble solid (called a precipitate).
🔵 Step 1: Example from salts:
➡️ BaCl₂(aq) + Na₂SO₄(aq) → BaSO₄(s) + 2NaCl(aq)
🟡 Here, BaSO₄(s) is an insoluble white precipitate.
🔵 Step 2: Another example:
➡️ AgNO₃(aq) + NaCl(aq) → AgCl(s) + NaNO₃(aq)
🟡 AgCl(s) is a white precipitate.
✔️ Final: Formation of an insoluble solid confirms that a precipitation reaction has occurred.
🔵 Question
16. Explain the following in terms of gain or loss of oxygen with two examples each.
(a) Oxidation
(b) Reduction
🟢 Answer
💡 Oxidation and reduction can be described using oxygen.
(a) Oxidation
🔵 Definition:
➡️ Oxidation is a process in which a substance gains oxygen OR loses hydrogen.
🔵 Examples:
➡️ 2Mg + O₂ → 2MgO
🟡 Magnesium gains oxygen, so magnesium is oxidised.
➡️ H₂S + O₂ → SO₂ + H₂O
🟡 Hydrogen sulphide gains oxygen to form sulphur dioxide.
(b) Reduction
🔵 Definition:
➡️ Reduction is a process in which a substance loses oxygen OR gains hydrogen.
🔵 Examples:
➡️ CuO + H₂ → Cu + H₂O
🟡 CuO loses oxygen to form Cu, so CuO is reduced.
➡️ Fe₂O₃ + 2Al → Al₂O₃ + 2Fe
🟡 Fe₂O₃ loses oxygen to form Fe; iron(III) oxide is reduced.
✔️ Final:
🔵 Oxidation = gain of oxygen.
🟢 Reduction = loss of oxygen.
⚡ Both happen together in redox reactions.
🔵 Question
17. A shiny brown coloured element ‘X’ on heating in air becomes black in colour. Name the element ‘X’ and the black coloured compound formed.
🟢 Answer
💡 This is a classic surface oxidation change.
🔵 Step 1: Shiny brown metal → This describes copper (Cu).
🔵 Step 2: On heating in air, copper forms a black coating of copper(II) oxide.
➡️ 2Cu + O₂ → 2CuO
🟡 CuO is black.
✔️ Final:
🔵 Element ‘X’ = Copper
🔵 Black compound formed = Copper(II) oxide (CuO)
🔵 Question
18. Why do we apply paint on iron articles?
🟢 Answer
💡 Idea: Iron rusts when it reacts with air and moisture.
🔵 Step 1: Iron reacts with oxygen and water to form rust (hydrated iron(III) oxide).
➡️ Fe + O₂ + H₂O → Fe₂O₃·xH₂O (rust)
🔵 Step 2: Paint forms a protective layer.
🟡 This layer prevents direct contact of the iron surface with air and water.
✔️ Final:
We apply paint to iron articles to prevent rusting (corrosion) by cutting off air and moisture.
🔵 Question
19. Oil and fat containing food items are flushed with nitrogen. Why?
🟢 Answer
💡 Concept: Rancidity.
🔵 Step 1: Fats and oils react with oxygen in air.
🟡 This slow oxidation produces unpleasant smell and taste.
🔵 Step 2: Replacing air with nitrogen gas removes oxygen.
🟢 Less oxygen = slower oxidation.
✔️ Final:
Food packets are flushed with nitrogen to slow down oxidation of fats and thus prevent rancidity (spoiling).
🔵 Question
20. Explain the following terms with one example each.
(a) Corrosion
(b) Rancidity
🟢 Answer
(a) Corrosion
🔵 Definition:
➡️ Corrosion is the slow process in which metals are eaten up by their reaction with air, water, or chemicals on their surface.
🔵 Classic example:
➡️ Iron + Oxygen + Water → Rust (Fe₂O₃·xH₂O)
🟡 The reddish-brown rust layer is corrosion of iron.
(b) Rancidity
🔵 Definition:
➡️ Rancidity is the process in which oils or fats in food get oxidised and develop an unpleasant smell and taste.
🔵 Everyday example:
➡️ Chips packets are flushed with nitrogen to slow rancidity.
✔️ Final: Rancidity makes oily food taste and smell bad because of oxidation of fats.
————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS FOR EXAMS
[CBSE MODEL QUESTION PAPER]
ESPECIALLY MADE FROM THIS LESSON ONLY
🔵 Section A – Very Short Answer (1 mark each)
Question 1
Write the chemical equation for the reaction between magnesium and oxygen.
Answer
🔵 Step 1: Magnesium burns in oxygen to form magnesium oxide.
➡️ Reaction: 2Mg + O₂ → 2MgO
✔️ Final: 2Mg + O₂ → 2MgO
Question 2
What type of reaction is represented by the equation:
Zn + H₂SO₄ → ZnSO₄ + H₂ ?
Answer
💡 Concept: When one element replaces another in a compound, it is a displacement reaction.
✔️ Final: Displacement Reaction
Question 3
Define a chemical equation.
Answer
🔵 A chemical equation is a symbolic representation of a chemical reaction using the chemical formulae of reactants and products with their physical states.
Question 4
What does the symbol (aq) indicate in a chemical equation?
Answer
🔵 (aq) stands for aqueous, meaning the substance is dissolved in water.
✔️ Example: NaCl(aq) → sodium chloride solution.
Question 5
Identify the reactants and products in the reaction:
Fe + S → FeS
Answer
🟢 Reactants: Fe (iron) and S (sulphur)
🟢 Product: FeS (iron sulphide)
Question 6
Balance the following chemical equation:
H₂ + Cl₂ → HCl
Answer
🔵 Step 1: Count atoms on both sides.
Left: H=2, Cl=2; Right: H=1, Cl=1
🔵 Step 2: Multiply HCl by 2 → H₂ + Cl₂ → 2HCl
✔️ Final balanced equation: H₂ + Cl₂ → 2HCl
🔵 Section B – Short Answer-I (2 marks each)
Question 7
State the law of conservation of mass and show its application with an example.
Answer
💡 Concept: Mass is neither created nor destroyed in a chemical reaction.
🔵 Example:
CaCO₃ → CaO + CO₂
➡️ Total mass of reactants = Total mass of products
✔️ Therefore, the law of conservation of mass holds true.
Question 8
Write the balanced chemical equation for the reaction between aluminium and hydrochloric acid.
Answer
🔵 Step 1: Write the unbalanced equation.
Al + HCl → AlCl₃ + H₂
🔵 Step 2: Balance it.
2Al + 6HCl → 2AlCl₃ + 3H₂
✔️ Final: 2Al + 6HCl → 2AlCl₃ + 3H₂
Question 9
What happens when a magnesium ribbon is burnt in air? Write the reaction.
Answer
🔵 Step 1: Magnesium burns with a dazzling white flame in air forming magnesium oxide.
➡️ 2Mg + O₂ → 2MgO
✔️ Final: Magnesium oxide (MgO) is formed.
Question 10
Why is it necessary to balance a chemical equation?
Answer
💡 Concept: A balanced chemical equation obeys the law of conservation of mass.
✔️ It ensures equal number of atoms of each element on both sides of the equation.
Question 11
What type of reaction occurs when an acid reacts with a base? Give one example.
Answer
💡 Concept: Acid + Base → Salt + Water (Neutralization)
➡️ Example: NaOH + HCl → NaCl + H₂O
✔️ Final: Neutralization Reaction
Question 12
Give an example of a combination reaction other than magnesium oxide formation.
Answer
🟢 Example: Calcium oxide reacts with water to form calcium hydroxide.
➡️ CaO + H₂O → Ca(OH)₂
✔️ Final: CaO + H₂O → Ca(OH)₂
🔵 Section C – Short Answer II (3 Marks Each)
Question 13
Differentiate between exothermic and endothermic reactions with examples.
Answer
💡 Concept:
- Exothermic reaction → heat is released.
- Endothermic reaction → heat is absorbed.
🔵 Exothermic Example:
➡️ C + O₂ → CO₂ + Heat
🔵 Endothermic Example:
➡️ N₂ + O₂ → 2NO (requires heat)
✔️ Summary: Exothermic = energy given out 🌡️, Endothermic = energy taken in ❄️.
Question 14
What is a precipitation reaction? Explain with one example.
Answer
💡 Concept: When two aqueous solutions react to form an insoluble solid (precipitate), it is a precipitation reaction.
🔵 Example:
➡️ AgNO₃(aq) + NaCl(aq) → AgCl(s) + NaNO₃(aq)
🟢 AgCl is a white precipitate.
✔️ Final: Formation of insoluble AgCl solid is precipitation.
Question 15
Explain the difference between oxidation and reduction with one example.
Answer
💡 Concept:
- Oxidation = gain of oxygen or loss of hydrogen.
- Reduction = loss of oxygen or gain of hydrogen.
🔵 Example:
➡️ CuO + H₂ → Cu + H₂O
🟢 CuO is reduced to Cu (loss of oxygen).
🟢 H₂ is oxidised to H₂O (gain of oxygen).
✔️ Final: Both oxidation and reduction occur simultaneously.
Question 16
What is a displacement reaction? Give one example of a metal displacing another.
Answer
💡 Concept: A more reactive element displaces a less reactive element from its compound.
🔵 Example:
➡️ Zn + CuSO₄ → ZnSO₄ + Cu
✔️ Zinc displaces copper from copper sulphate — displacement reaction.
Question 17
What is corrosion? Mention one method to prevent it.
Answer
💡 Concept: Corrosion is the gradual destruction of metals due to reaction with moisture, air, or chemicals.
🔵 Example: Rusting of iron – 4Fe + 3O₂ + 6H₂O → 4Fe(OH)₃
🟢 Prevention:
➡️ By galvanization (coating iron with zinc).
✔️ Final: Corrosion can be prevented by coating or painting the surface.
Question 18
Define rancidity. How can it be prevented?
Answer
💡 Concept: Rancidity is oxidation of oils/fats when exposed to air, producing foul smell or taste.
🟢 Prevention:
🔵 Step 1: Store food in airtight containers.
🔵 Step 2: Keep away from light and heat.
🔵 Step 3: Add antioxidants like BHA or BHT.
✔️ Final: Prevent rancidity by avoiding oxidation of fats.
Question 19
Explain the process of rusting of iron. What conditions are necessary for rusting to occur?
Answer
💡 Concept: Rusting is oxidation of iron in the presence of air (oxygen) and moisture (water).
🔵 Reaction: Fe + O₂ + H₂O → Fe₂O₃·xH₂O (hydrated ferric oxide)
🟢 Conditions:
1️⃣ Presence of water
2️⃣ Presence of oxygen
✔️ Final: Rust (Fe₂O₃·xH₂O) forms only when both air and moisture are present.
Question 20
Differentiate between combination and decomposition reactions with examples.
Answer
🔵 Combination Reaction: Two or more substances combine to form a single product.
➡️ Example: 2H₂ + O₂ → 2H₂O
🔵 Decomposition Reaction: One compound breaks down into two or more simpler substances.
➡️ Example: 2H₂O → 2H₂ + O₂ (by electrolysis)
✔️ Final: Combination = joining; Decomposition = breaking down.
Question 21 (With Internal Choice)
(A) What happens when ferrous sulphate crystals are heated? Write the chemical reaction.
OR
(B) Write the reaction when lead nitrate is heated and state the type of reaction.
Answer
🟢 (A) On heating, ferrous sulphate decomposes into ferric oxide, sulphur dioxide, and sulphur trioxide.
➡️ 2FeSO₄ → Fe₂O₃ + SO₂ + SO₃
✔️ Decomposition Reaction
OR
🟢 (B) Lead nitrate decomposes to lead oxide, nitrogen dioxide, and oxygen.
➡️ 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂
✔️ Thermal Decomposition Reaction
Question 22 (With Internal Choice)
(A) Explain the role of oxidation and reduction in a redox reaction with an example.
OR
(B) What are double displacement reactions? Illustrate with an example.
Answer
🟢 (A) In redox reactions, oxidation and reduction occur together.
➡️ Example: Zn + CuSO₄ → ZnSO₄ + Cu
🔵 Zinc → oxidized (loss of electrons)
🔵 Copper → reduced (gain of electrons)
✔️ Final: Both oxidation and reduction occur simultaneously — redox reaction.
OR
🟢 (B) Double displacement reaction involves exchange of ions between reactants.
➡️ Example: Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl
✔️ Formation of BaSO₄ (white precipitate) indicates a double displacement reaction.
🔴 Section D – Long Answer (4 Marks Each)
Question 23
Explain the different types of chemical reactions with one example of each.
Answer
💡 Concept: There are several basic types of chemical reactions as per NCERT.
🔵 1. Combination Reaction:
➡️ Two or more substances combine to form a single compound.
Example: CaO + H₂O → Ca(OH)₂
🔵 2. Decomposition Reaction:
➡️ One compound breaks down into two or more simpler substances.
Example: 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂
🔵 3. Displacement Reaction:
➡️ A more reactive element displaces a less reactive one.
Example: Zn + CuSO₄ → ZnSO₄ + Cu
🔵 4. Double Displacement Reaction:
➡️ Exchange of ions between two compounds.
Example: Na₂SO₄ + BaCl₂ → BaSO₄ + 2NaCl
✔️ Final: Each reaction shows a different pattern of chemical change based on combination, breakdown, or exchange of elements.
Question 24
What is a redox reaction? Explain with a suitable example and identify oxidising and reducing agents.
Answer
💡 Concept:
A redox reaction involves both oxidation and reduction occurring simultaneously.
🔵 Example: Zn + H₂SO₄ → ZnSO₄ + H₂
🟢 Zinc → oxidised (loss of electrons)
🟢 Hydrogen ions → reduced (gain of electrons)
🔵 Oxidising Agent: H₂SO₄ (provides H⁺ ions, causes oxidation of Zn)
🔵 Reducing Agent: Zn (donates electrons, causes reduction of H⁺)
✔️ Final: Oxidation and reduction always occur together; one substance loses electrons while another gains them.
Question 25
Describe any three methods used for preventing corrosion.
Answer
💡 Concept: Corrosion weakens metals, hence prevention is vital.
🔵 1️⃣ Painting or Oiling:
➡️ Coats metal surface to stop contact with air and moisture.
🔵 2️⃣ Galvanisation:
➡️ Coating iron with a thin layer of zinc to prevent rusting.
🔵 3️⃣ Alloying:
➡️ Mixing a metal with another element to improve resistance.
Example: Stainless steel (Iron + Nickel + Chromium) resists rust.
✔️ Final: Corrosion can be minimised by creating a barrier between metal and environmental agents.
Question 26 (With Internal Choice)
(A) Write the balanced chemical equations for the following and identify the type of reaction in each case:
(i) Calcium hydroxide reacts with carbon dioxide.
(ii) Heating of lead nitrate.
OR
(B) Explain thermal decomposition and photochemical decomposition with one example each.
Answer
🟢 (A) (i) Ca(OH)₂ + CO₂ → CaCO₃ + H₂O
➡️ Type: Combination Reaction
🟢 (A) (ii) 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂
➡️ Type: Decomposition Reaction
✔️ OR
🟢 (B)
🔵 Thermal Decomposition: Breaking of a compound by heat.
➡️ 2HgO → 2Hg + O₂
🔵 Photochemical Decomposition: Breaking of compound by light energy.
➡️ 2AgCl → 2Ag + Cl₂ (in sunlight)
✔️ Both are types of decomposition reactions.
Question 27
Explain the process of balancing a chemical equation with an example.
Answer
💡 Concept: Balancing ensures the law of conservation of mass is obeyed—same number of atoms on both sides.
🔵 Example: Fe + H₂O → Fe₃O₄ + H₂
➡️ Step 1: Count atoms on both sides.
LHS: Fe = 1, H = 2, O = 1
➡️ Step 2: Adjust coefficients → 3Fe + 4H₂O → Fe₃O₄ + 4H₂
✔️ Final balanced equation: 3Fe + 4H₂O → Fe₃O₄ + 4H₂
✔️ Law of mass conservation satisfied.
Question 28 (With Internal Choice)
(A) What are decomposition reactions? Classify them and give one example of each type.
OR
(B) What happens when water is added to quicklime? Write the equation and mention the type of reaction.
Answer
🟢 (A)
💡 Decomposition Reaction: A single compound breaks into simpler substances.
🔵 (i) Thermal Decomposition:
➡️ CaCO₃ → CaO + CO₂
🔵 (ii) Electrolytic Decomposition:
➡️ 2H₂O → 2H₂ + O₂
🔵 (iii) Photochemical Decomposition:
➡️ 2AgBr → 2Ag + Br₂
✔️ Each uses a different source of energy (heat, electricity, or light).
OR
🟢 (B)
CaO + H₂O → Ca(OH)₂ + Heat
➡️ Type: Exothermic Combination Reaction
✔️ Quicklime reacts vigorously with water releasing heat.
Question 29
Explain any four characteristics of a chemical reaction.
Answer
💡 Concept: Observable features show a chemical change has occurred.
🔵 1️⃣ Change in Colour:
➡️ Example: Fe + CuSO₄ → FeSO₄ + Cu (blue to green).
🔵 2️⃣ Evolution of Gas:
➡️ Example: Zn + H₂SO₄ → ZnSO₄ + H₂↑
🔵 3️⃣ Change in Temperature (Heat):
➡️ Example: C + O₂ → CO₂ + Heat (exothermic).
🔵 4️⃣ Formation of Precipitate:
➡️ Example: BaCl₂ + Na₂SO₄ → BaSO₄ + 2NaCl (white precipitate).
✔️ Final: These characteristics confirm that a new substance has formed.
Question 30 (With Internal Choice)
(A) Write observations for the following reactions and name the type of reaction:
(i) Iron nails dipped in copper sulphate solution.
(ii) Heating ferrous sulphate crystals.
OR
(B) Explain the effects of oxidation in our daily life with examples.
Answer
🟢 (A) (i) Iron nails turn reddish-brown, and blue colour of solution fades.
➡️ Reaction: Fe + CuSO₄ → FeSO₄ + Cu
✔️ Type: Displacement Reaction
🟢 (A) (ii) Green ferrous sulphate crystals turn brown; gases SO₂ and SO₃ are released.
➡️ Reaction: 2FeSO₄ → Fe₂O₃ + SO₂ + SO₃
✔️ Type: Decomposition Reaction
OR
🟢 (B)
💡 Effects of Oxidation in Daily Life:
🔵 (1) Rancidity: Fats and oils in food oxidize → bad smell.
🔵 (2) Corrosion: Metals like iron rust due to oxidation → Fe₂O₃·xH₂O.
🔵 (3) Respiration: Biological oxidation of glucose → CO₂ + H₂O + Energy.
✔️ Final: Oxidation has both harmful (rusting, rancidity) and useful (respiration) effects.
——————————————————————————————————————————————————————————————————————————–
MIND MAP
————————————————————————————————————————————————————————————————————————–
