Class 12 : Chemistry (English) – Chapter 10: Biomolecules
EXPLANATION & SUMMARY
✨ Introduction
🔵 Biomolecules are organic molecules that are essential for life.
🟢 They form the structural and functional basis of cells and organisms.
🟠 Classified into carbohydrates, proteins, nucleic acids, and lipids, along with vitamins and hormones.
🔴 Their study connects chemistry with biology and medicine.


🌿 Carbohydrates
Definition & Types
✔ Carbohydrates = polyhydroxy aldehydes or ketones, or compounds that yield them on hydrolysis.
Monosaccharides: Simple sugars (glucose, fructose).
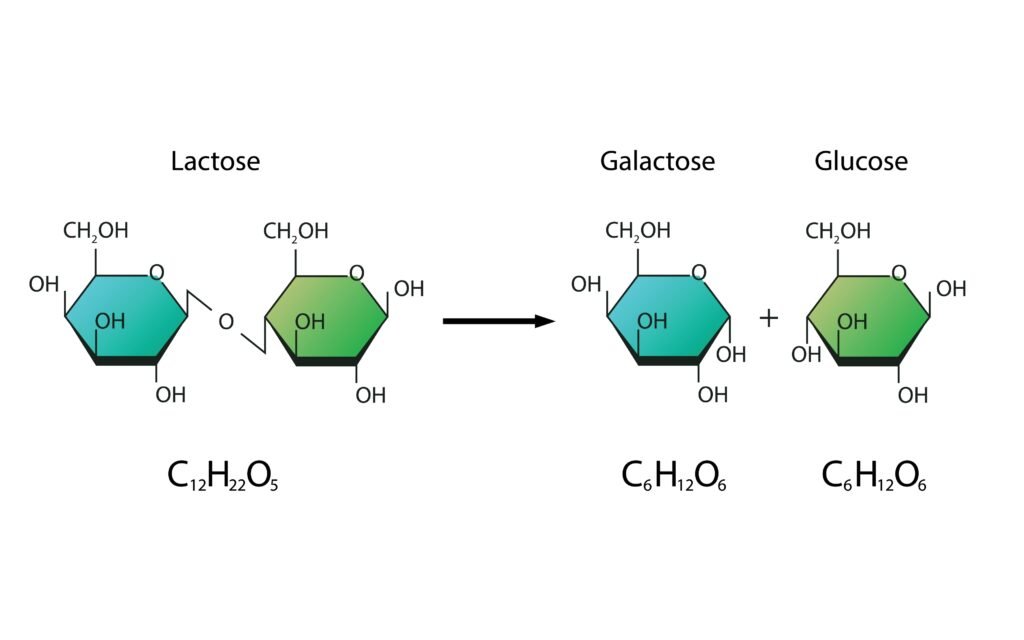
Disaccharides: 2 monosaccharide units (sucrose, maltose, lactose).
Polysaccharides: Large polymers (starch, cellulose, glycogen).
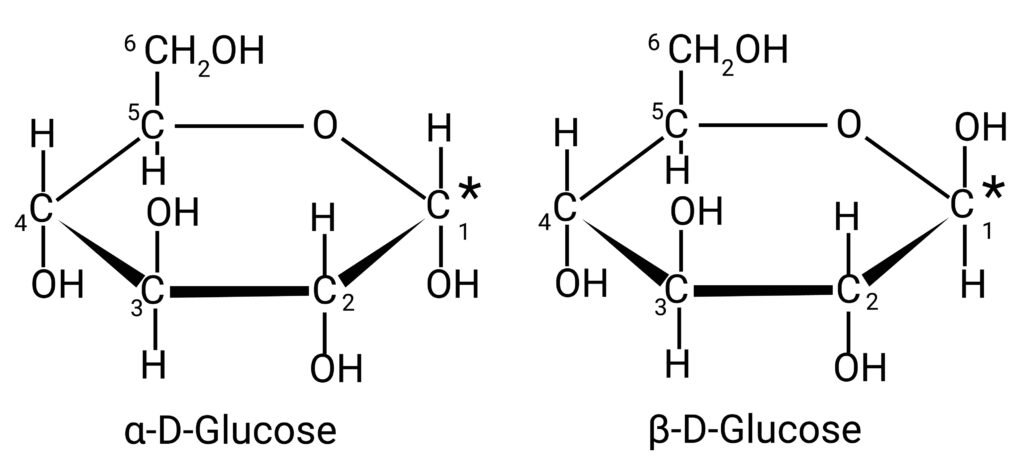
Glucose
Formula: C₆H₁₂O₆.
Open-chain and cyclic forms (pyranose/hexose).
Tests: reduction with Tollen’s and Fehling’s reagents.
Fructose
Ketohexose sugar.
Cyclic furanose form predominates.
Polysaccharides
🟩 Starch: Storage in plants, composed of amylose and amylopectin.
🟨 Cellulose: Structural polymer in plant cell walls, β-glycosidic linkages.
🟧 Glycogen: Storage in animals, similar to amylopectin but more branched.
🧬 Proteins
Amino Acids
Building blocks of proteins.
Contain amino (–NH₂) and carboxyl (–COOH) groups.
Exist as zwitterions at physiological pH.
Structure of Proteins
Primary: Linear sequence of amino acids.
Secondary: α-helix and β-pleated sheet (H-bonding).
Tertiary: 3D folding, stabilised by disulfide bonds, H-bonds, hydrophobic interactions.
Quaternary: Association of multiple polypeptide chains.
Functions
Enzymes (catalysis), hormones, structural proteins (collagen, keratin), transport (haemoglobin).
🌸 Nucleic Acids
Components
Nucleotides = Sugar + Base + Phosphate.
Bases: Purines (adenine, guanine), Pyrimidines (cytosine, thymine, uracil).
DNA
Double helix model (Watson & Crick).
Complementary base pairing (A–T, G–C).
RNA
Single-stranded.
Types: mRNA, tRNA, rRNA.
Involved in protein synthesis.
🌍 Vitamins
Organic compounds needed in small amounts.
Fat-soluble: A, D, E, K.
Water-soluble: B-complex, C.
Deficiency → diseases (scurvy, rickets, night blindness).
🧪 Hormones
Chemical messengers secreted by endocrine glands.
Types:
Peptide hormones: insulin, glucagon.
Steroid hormones: testosterone, estrogen.
Amino acid-derived: adrenaline, thyroxine.
🌟 Enzymes
Biological catalysts, mostly proteins.
Specific for substrates.
Lower activation energy.
Activity affected by pH, temperature, inhibitors.







📝 Summary
Biomolecules are molecules essential for life: carbohydrates, proteins, nucleic acids, lipids, vitamins, hormones.
Carbohydrates: Monosaccharides (glucose, fructose), disaccharides (sucrose, maltose, lactose), polysaccharides (starch, glycogen, cellulose). Provide energy and storage.
Proteins: Polymers of amino acids, levels of structure from primary to quaternary. Functions include catalysis (enzymes), structure, transport, hormones.
Nucleic acids: DNA (double helix, genetic material) and RNA (protein synthesis). Composed of nucleotides with bases (purines, pyrimidines).
Vitamins: Micronutrients required for metabolism. Fat-soluble (A, D, E, K) and water-soluble (B-complex, C). Deficiency causes disorders like rickets, scurvy.
Hormones: Regulatory biomolecules controlling growth, metabolism, and reproduction. Classified as peptides, steroids, or amine derivatives.
Enzymes: Biological catalysts, highly specific, functioning under mild conditions, essential for biochemical reactions.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
Question 10.1
What are monosaccharides?
Answer:
🔵 Monosaccharides are the simplest carbohydrates that cannot be hydrolysed further into smaller sugar units.
🟢 They are polyhydroxy aldehydes or ketones.
🟠 General formula: Cₙ(H₂O)ₙ.
🔴 Examples: Glucose, Fructose, Galactose.
Question 10.2
What are reducing sugars?
Answer:
✔ Reducing sugars are carbohydrates that can reduce Fehling’s solution or Tollen’s reagent.
💡 Reason: They contain a free aldehyde group (–CHO) or a free ketone group (–C=O) in solution.
🌿 Examples: Glucose, Fructose, Maltose, Lactose.
Question 10.3
Write two main functions of carbohydrates in plants.
Answer:
🔷 Energy storage: Starch acts as the primary storage carbohydrate.
🔶 Structural role: Cellulose forms the rigid cell wall providing mechanical strength.
Question 10.4
Classify the following into monosaccharides and disaccharides: Ribose, 2-deoxyribose, maltose, galactose, fructose, lactose.
Answer:
🟦 Monosaccharides: Ribose, 2-deoxyribose, Galactose, Fructose.
🟨 Disaccharides: Maltose, Lactose.
Question 10.5
What do you understand by the term glycosidic linkage?
Answer:
✔ Glycosidic linkage = covalent bond joining two monosaccharides.
🧪 It is formed when the –OH group of one sugar reacts with the anomeric carbon of another sugar with elimination of water.
🌿 Example: In maltose, glucose units are linked by α(1→4) glycosidic bond.
Question 10.6
What is glycogen? How is it different from starch?
Answer:
🟢 Glycogen: Storage polysaccharide in animals, highly branched polymer of α-D-glucose.
🟠 Difference from starch:
Starch has two components: amylose (linear) and amylopectin (branched).
Glycogen is structurally similar to amylopectin but more highly branched.
Starch = plant storage; Glycogen = animal storage.
Question 10.7
What are the hydrolysis products of (i) sucrose and (ii) lactose?
Answer:
🟦 Sucrose hydrolysis: → Glucose + Fructose.
🟩 Lactose hydrolysis: → Glucose + Galactose.
Question 10.8
What is the basic structural difference between starch and cellulose?
Answer:
🔵 Starch: Polymer of α-D-glucose units linked by α(1→4) and α(1→6) bonds.
🟠 Cellulose: Polymer of β-D-glucose units linked by β(1→4) bonds.
🌿 Due to α vs β linkages, starch is digestible by humans but cellulose is not.
Question 10.9
What happens when D-glucose is treated with the following reagents?
(i) HI
(ii) Bromine water
(iii) HNO₃
Answer:
🟦 With HI → n-hexane (reduction to straight-chain hydrocarbon).
🟨 With bromine water → Gluconic acid (oxidation of –CHO to –COOH).
🟩 With HNO₃ → Saccharic acid (oxidation of both –CHO and terminal –CH₂OH).
Question 10.10
Enumerate the reactions of D-glucose which cannot be explained by its open chain structure.
Answer:
✔ Observed reactions:
Glucose forms pentaacetate with acetic anhydride (5 –OH groups).
Glucose does not give Schiff’s test for free aldehyde.
Glucose does not form hydrogen sulphite addition product.
Exists in α- and β- forms (anomers).
🟢 These facts prove glucose has a cyclic hemiacetal structure not explained by simple open chain.
Question 10.11
What are essential and non-essential amino acids? Give two examples of each type.
Answer:
🟦 Essential amino acids: Cannot be synthesised by the body, must be obtained from diet.
Examples: Valine, Leucine, Lysine, Phenylalanine.
🟩 Non-essential amino acids: Can be synthesised in the body.
Examples: Glycine, Alanine, Glutamic acid, Aspartic acid.
Question 10.12
Define the following as related to proteins:
(i) Peptide linkage
(ii) Primary structure
(iii) Denaturation
Answer:
🟦 Peptide linkage: –CO–NH– bond formed between –COOH of one amino acid and –NH₂ of another.
🟨 Primary structure: Linear sequence of amino acids in polypeptide chain.
🟩 Denaturation: Loss of biological activity of protein due to disruption of secondary/tertiary structure by heat, pH, etc.
Question 10.13
What are the common types of secondary structure of proteins?
Answer:
✔ Secondary structure refers to regular folding due to hydrogen bonding.
🟦 Two common types:
α-helix (spiral, right-handed).
β-pleated sheet (zigzag, sheet-like).
Question 10.14
What type of bonding helps in stabilising the α-helix structure of proteins?
Answer:
🟦 α-helix is stabilised by intra-chain hydrogen bonds.
🟩 These bonds form between –C=O of one amino acid and –NH of another located four residues ahead in the chain.
✔ This coiling gives a right-handed helix.
Question 10.15
Differentiate between globular and fibrous proteins.
Answer:
🔷 Globular proteins
Spherical, compact.
Soluble in water.
Biological roles: enzymes, hormones, transport proteins (e.g., insulin, hemoglobin).
🔶 Fibrous proteins
Long, thread-like.
Insoluble in water.
Structural roles (e.g., keratin, collagen).
Question 10.16
How do you explain the amphoteric behaviour of amino acids?
Answer:
🟦 Amino acids contain both acidic (–COOH) and basic (–NH₂) groups.
🟨 They exist as zwitterions (–NH₃⁺ and –COO⁻ simultaneously).
✔ Thus, they react with:
Acids → act as base.
Bases → act as acid.
Question 10.17
What are enzymes?
Answer:
🟢 Enzymes are biological catalysts, mostly proteins.
🟦 They speed up biochemical reactions without being consumed.
🌿 Example: Amylase (starch → maltose), Pepsin (protein digestion).
Question 10.18
What is the effect of denaturation on the structure of proteins?
Answer:
✔ Denaturation = disruption of secondary and tertiary structures.
🟦 Primary structure remains intact.
🟨 Loss of biological activity occurs (e.g., coagulation of egg white on boiling).
Question 10.19
How are vitamins classified? Name the vitamin responsible for the coagulation of blood.
Answer:
🟩 Classification:
Fat-soluble vitamins: A, D, E, K.
Water-soluble vitamins: B-complex, C.
🟦 Vitamin responsible for blood coagulation → Vitamin K.
Question 10.20
Why are vitamin A and vitamin C essential to us? Give their important sources.
Answer:
🟢 Vitamin A
Functions: vision, healthy skin, growth.
Sources: carrots, butter, fish liver oil.
🟠 Vitamin C
Functions: healthy gums, wound healing, prevents scurvy.
Sources: citrus fruits, amla, tomatoes.
Question 10.21
What are nucleic acids? Mention their two important functions.
Answer:
🟦 Nucleic acids = polymers of nucleotides (DNA, RNA).
🟨 Functions:
Carry genetic information from one generation to next.
Direct protein synthesis in cells.
Question 10.22
What is the difference between a nucleoside and a nucleotide?
Answer:
✔ Nucleoside = Base + Sugar (no phosphate).
✔ Nucleotide = Base + Sugar + Phosphate group.
Question 10.23
The two strands in DNA are not identical but are complementary. Explain.
Answer:
🟩 Bases pair by hydrogen bonding:
Adenine (A) with Thymine (T).
Guanine (G) with Cytosine (C).
🟦 Hence, the sequence on one strand dictates the complementary sequence on the other.
Question 10.24
Write the important structural and functional differences between DNA and RNA.
Answer:
🔷 DNA
Sugar: deoxyribose.
Bases: A, G, C, T.
Structure: double helix.
Function: genetic material in most organisms.
🔶 RNA
Sugar: ribose.
Bases: A, G, C, U.
Structure: single-stranded.
Function: involved in protein synthesis (mRNA, tRNA, rRNA).
Question 10.25
What are the different types of RNA found in the cell?
Answer:
🟦 Three types:
mRNA (messenger RNA): carries genetic code from DNA.
tRNA (transfer RNA): brings amino acids during protein synthesis.
rRNA (ribosomal RNA): structural and catalytic role in ribosomes.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
Section A (Q1–Q16, MCQs)
(16 × 1 = 16 Marks)
Options for Assertion–Reason Questions:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Q1. Which of the following is a disaccharide?
🔵 (A) Glucose
🟢 (B) Galactose
🟠 (C) Maltose
🔴 (D) Fructose
Answer: 🟠 (C) Maltose
Q2. The bond linking monosaccharide units in polysaccharides is:
🔵 (A) Glycosidic bond
🟢 (B) Peptide bond
🟠 (C) Hydrogen bond
🔴 (D) Ester bond
Answer: 🔵 (A) Glycosidic bond
Q3. Which one is a reducing sugar?
🔵 (A) Sucrose
🟢 (B) Lactose
🟠 (C) Maltose
🔴 (D) Both (B) and (C)
Answer: 🔴 (D) Both (B) and (C)
Q4. Which vitamin prevents scurvy?
🔵 (A) Vitamin A
🟢 (B) Vitamin B₁
🟠 (C) Vitamin C
🔴 (D) Vitamin D
Answer: 🟠 (C) Vitamin C
Q5. Which form of glucose is found in polysaccharides like starch?
🔵 (A) D(+) Glucose
🟢 (B) L(−) Glucose
🟠 (C) Both D and L
🔴 (D) Neither D nor L
Answer: 🔵 (A) D(+) Glucose
Q6. The difference between cellulose and starch is due to:
🔵 (A) Type of monosaccharide
🟢 (B) Type of glycosidic linkage
🟠 (C) Molecular formula
🔴 (D) Degree of polymerisation
Answer: 🟢 (B) Type of glycosidic linkage
Q7. Which nucleic acid has thymine as a base?
🔵 (A) DNA
🟢 (B) RNA
🟠 (C) Both
🔴 (D) None
Answer: 🔵 (A) DNA
Q8. Which one is a fibrous protein?
🔵 (A) Haemoglobin
🟢 (B) Keratin
🟠 (C) Myoglobin
🔴 (D) Enzyme
Answer: 🟢 (B) Keratin
Q9. The hydrolysis of sucrose gives:
🔵 (A) Glucose + Glucose
🟢 (B) Glucose + Galactose
🟠 (C) Glucose + Fructose
🔴 (D) Fructose + Galactose
Answer: 🟠 (C) Glucose + Fructose
Q10. Which vitamin is fat soluble?
🔵 (A) Vitamin B
🟢 (B) Vitamin C
🟠 (C) Vitamin D
🔴 (D) None
Answer: 🟠 (C) Vitamin D
Q11. Assertion (A): Glycogen is a storage carbohydrate in animals.
Reason (R): Glycogen has a highly branched structure, making it suitable for quick energy release.
Answer: 1 (Both A and R true, R explains A)
Q12. Assertion (A): Cellulose cannot be digested by humans.
Reason (R): Humans lack the enzyme cellulase.
Answer: 1 (Both A and R true, R explains A)
Q13. Which of the following is NOT an amino acid?
🔵 (A) Glycine
🟢 (B) Alanine
🟠 (C) Cytosine
🔴 (D) Serine
Answer: 🟠 (C) Cytosine
Q14. Which type of bond stabilises the α-helix structure of proteins?
🔵 (A) Covalent bond
🟢 (B) Ionic bond
🟠 (C) Hydrogen bond
🔴 (D) Peptide bond
Answer: 🟠 (C) Hydrogen bond
Q15. Which RNA carries amino acids to ribosomes?
🔵 (A) mRNA
🟢 (B) tRNA
🟠 (C) rRNA
🔴 (D) All of these
Answer: 🟢 (B) tRNA
Q16. Which of the following is an essential amino acid?
🔵 (A) Glycine
🟢 (B) Alanine
🟠 (C) Valine
🔴 (D) Serine
Answer: 🟠 (C) Valine
Section B (Q17–Q21, Very Short Answer, 2 marks each)
(5 × 2 = 10 Marks, ~30 words each)
Q17. Define monosaccharides with examples.
🟦 Monosaccharides are the simplest carbohydrates which cannot be hydrolysed further.
🟢 Examples — Glucose, Fructose.
Q18. State two main functions of carbohydrates in plants.
🌿 Provide energy through respiration.
🌿 Store energy in the form of starch.
Q19. Name two essential amino acids.
✔ Valine
✔ Leucine
Q20. What are nucleosides?
💡 Nucleosides = Nitrogen base + Sugar (ribose/deoxyribose), without phosphate group.
Q21. Name two water-soluble vitamins and their deficiency diseases.
🟢 Vitamin B₁ — Beriberi
🟠 Vitamin C — Scurvy
Section C (Q22–Q28, Short Answer, 3 marks each)
(7 × 3 = 21 Marks, ~50 words each)
Q22. Differentiate between reducing and non-reducing sugars with examples.
🔷 Reducing sugars: Have free aldehyde/ketone group → reduce Tollen’s or Fehling’s reagent. (Examples: Glucose, Maltose, Lactose).
🔶 Non-reducing sugars: No free group, do not reduce reagents. (Example: Sucrose).
Q23. Write two differences between globular and fibrous proteins.
🟩 Globular proteins: Spherical, soluble, functional (e.g., enzymes, haemoglobin).
🟨 Fibrous proteins: Long, insoluble, structural (e.g., keratin, collagen).
Q24. Explain peptide linkage.
🧪 A peptide linkage is formed between –COOH group of one amino acid and –NH₂ group of another with removal of H₂O.
⚗ Example: –CO–NH–
Q25. Differentiate between DNA and RNA.
🔷 DNA: Double stranded, contains deoxyribose, bases = A, G, C, T.
🔶 RNA: Single stranded, contains ribose, bases = A, G, C, U.
Q26. What are enzymes? Write two characteristics.
💡 Enzymes = Biological catalysts, mostly proteins.
✅ Highly specific in action.
✅ Work under mild conditions (pH, temperature).
Q27. What is glycogen? How is it different from starch?
🟦 Glycogen = Storage polysaccharide in animals.
🟩 Highly branched structure, stored in liver/muscles.
🟨 Starch = Plant storage polysaccharide with amylose + amylopectin.
Q28. Write the hydrolysis products of sucrose and lactose.
✔ Sucrose → Glucose + Fructose
✔ Lactose → Glucose + Galactose
Section D (Q29–Q30, Case-Based, 4 marks each)
Q29. Read the passage and answer:
Glucose is an important monosaccharide found in fruits and honey. It can exist in open-chain as well as cyclic forms. In aqueous solution, it shows mutarotation due to interconversion of its α- and β- forms.
(i) What is the molecular formula of glucose?
(ii) What is mutarotation?
(iii) Name the cyclic structures of α- and β-glucose.
Answer 29
🟦 (i) Molecular formula = C₆H₁₂O₆.
🟩 (ii) Mutarotation = Change in optical rotation due to interconversion between α- and β- anomers via open-chain form.
🟨 (iii) α-D-glucopyranose and β-D-glucopyranose.
Q30. Read the passage and answer:
Proteins are polymers of amino acids and are essential biomolecules. They exhibit different levels of structural organisation, from primary to quaternary. Their biological activity is sensitive to changes in conditions.
(i) What is the primary structure of protein?
(ii) Which bond is responsible for secondary structure?
(iii) What happens when a protein is denatured?
Answer 30
🟦 (i) Primary structure = Linear sequence of amino acids in a polypeptide chain.
🟩 (ii) Secondary structure = Stabilised by hydrogen bonds (α-helix, β-sheet).
🟨 (iii) Denaturation → Loss of secondary and tertiary structure, loss of biological activity, primary structure remains intact.
Section E (Q31–Q33, Long Answer, 5 marks each, with OR choice)
Q31.
Explain the structural difference between starch and cellulose.
OR
Explain the difference between DNA and RNA.
Answer 31
🟦 Starch vs Cellulose
Both are polysaccharides of glucose.
Starch: α-D-glucose units linked by α(1→4) and α(1→6) linkages (amylose + amylopectin). Digestible by humans.
Cellulose: β-D-glucose units linked by β(1→4) bonds, forms long straight chains with H-bonding. Indigestible to humans.
🟩 OR – DNA vs RNA
DNA: Double stranded, sugar = deoxyribose, bases = A, G, C, T. Genetic material.
RNA: Single stranded, sugar = ribose, bases = A, G, C, U. Role in protein synthesis.
Q32.
Discuss the classification of vitamins with examples.
OR
Explain the functions and deficiency diseases of Vitamin A, D, E, K.
Answer 32
🟦 Classification of Vitamins
Fat soluble: A, D, E, K (stored in liver, can be toxic in excess).
Water soluble: B-complex, C (excess excreted in urine).
🟩 OR – Functions & Deficiencies
Vitamin A: Vision, skin health; deficiency → night blindness.
Vitamin D: Calcium absorption; deficiency → rickets.
Vitamin E: Antioxidant; deficiency → reproductive failure.
Vitamin K: Blood clotting; deficiency → delayed clotting.
Q33.
Describe the role of nucleic acids in transmission of heredity.
OR
Explain the functions of different types of RNA.
Answer 33
🟦 Nucleic Acids & Heredity
DNA carries genetic information in sequence of bases.
Replication ensures transfer to daughter cells.
Base pairing ensures accuracy (A–T, G–C).
Genes on DNA code for proteins which determine traits.
🟩 OR – Types of RNA
mRNA: Carries genetic code from DNA to ribosome.
tRNA: Brings amino acids during translation.
rRNA: Structural and catalytic role in ribosomes.
✔ Together, RNA types ensure accurate protein synthesis.
————————————————————————————————————————————————————————————————————————————
