Class 12 : Chemistry (English) – Chapter 8: Aldehydes, Ketones and Carboxylic Acids
EXPLANATION & SUMMARY
✨ Introduction
🔵 Aldehydes, Ketones, and Carboxylic acids form a crucial group of carbonyl compounds containing the polar carbonyl group (>C=O).
🟢 These compounds are widely found in natural products, drugs, flavours, fragrances, industrial raw materials, and biomolecules.
🟠 The high reactivity of carbonyl group arises due to polarisation of C=O bond (C δ⁺, O δ⁻).
🔴 Their chemistry includes structure, nomenclature, methods of preparation, physical and chemical properties, mechanisms of reactions, and uses.
🌿 Nomenclature
Aldehydes (-CHO):
Common names: derived from acids (e.g., formaldehyde, acetaldehyde).
IUPAC: Replace -e of alkane with -al (e.g., methanal, ethanal).
Ketones (>C=O):
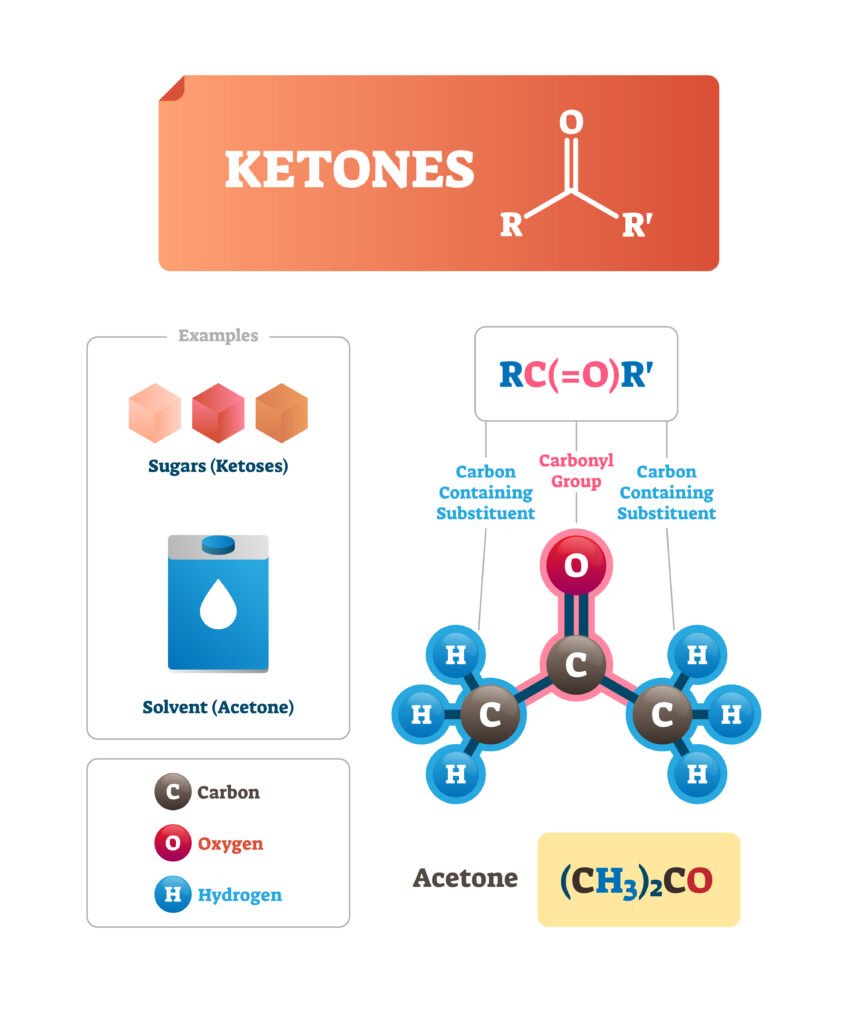
Common names: Alkyl groups + ketone (e.g., acetone, methyl ethyl ketone).
IUPAC: Replace -e of alkane with -one (e.g., propanone, butanone).
Carboxylic acids (-COOH):
Common names: Derived from Latin/Greek names of sources (formic, acetic).
IUPAC: Replace -e of alkane with -oic acid (e.g., ethanoic acid, propanoic acid).
🧪 Preparation
Aldehydes and Ketones
🟦 By oxidation of alcohols:
1° alcohols → Aldehydes
2° alcohols → Ketones
🟩 By ozonolysis of alkenes:
RCH=CHR’ + O₃ → RCHO + R’CHO / RCOCH₃
🟨 By Rosenmund reduction:
Acyl chloride + H₂ (Pd/BaSO₄) → Aldehyde
🟧 By Friedel–Crafts acylation (for aromatic ketones).
Carboxylic acids
🟦 By oxidation of primary alcohols/aldehydes with KMnO₄ or CrO₃.
🟩 From nitriles: RCN + 2H₂O + H⁺ → RCOOH + NH₄⁺.
🟨 From Grignard reagents: RMgX + CO₂ → RCOOH.
⚡ Physical Properties
🔵 Aldehydes & Ketones:
Polar, higher boiling points than hydrocarbons but lower than alcohols.
Simple members soluble in water due to H-bonding.
Pleasant odours, used in perfumes/flavouring.
🟢 Carboxylic acids:
Very high boiling points due to strong H-bonding (dimers in liquid phase).
Soluble in water (up to C4), solubility decreases with chain length.
Have sharp, pungent odours.
🔬 Chemical Properties
Aldehydes vs Ketones
Nucleophilic addition reactions:
HCN → cyanohydrin
NaHSO₃ → bisulphite adduct
NH₂-Z (Z = alkyl/aryl) → imines, oximes, hydrazones
Reduction (NaBH₄, LiAlH₄) → alcohols
Addition of Grignard reagents → alcohols
Oxidation:
Aldehydes → acids easily (Tollen’s, Fehling’s positive).
Ketones resist oxidation but give acids with strong oxidisers.
Reduction:
Catalytic hydrogenation → alcohols.
Clemmensen, Wolff–Kishner → hydrocarbons.
Tests:
Aldehydes: Tollen’s (silver mirror), Fehling’s (red ppt Cu₂O), Schiff’s test.
Ketones: No such reactions (except methyl ketones → iodoform test).
Carboxylic Acids
Acidic nature:
Dissociate as RCOOH ⇌ RCOO⁻ + H⁺
Strength affected by electron-withdrawing/donating groups.
Reactions:
Salt formation with bases.
Esterification with alcohols (H₂SO₄ catalyst).
Amides from acid chlorides.
Reduction → primary alcohols.
Decarboxylation → hydrocarbons.
🌍 Uses
Aldehydes: Formaldehyde (plastics, disinfectant), Acetaldehyde (acetic acid manufacture).
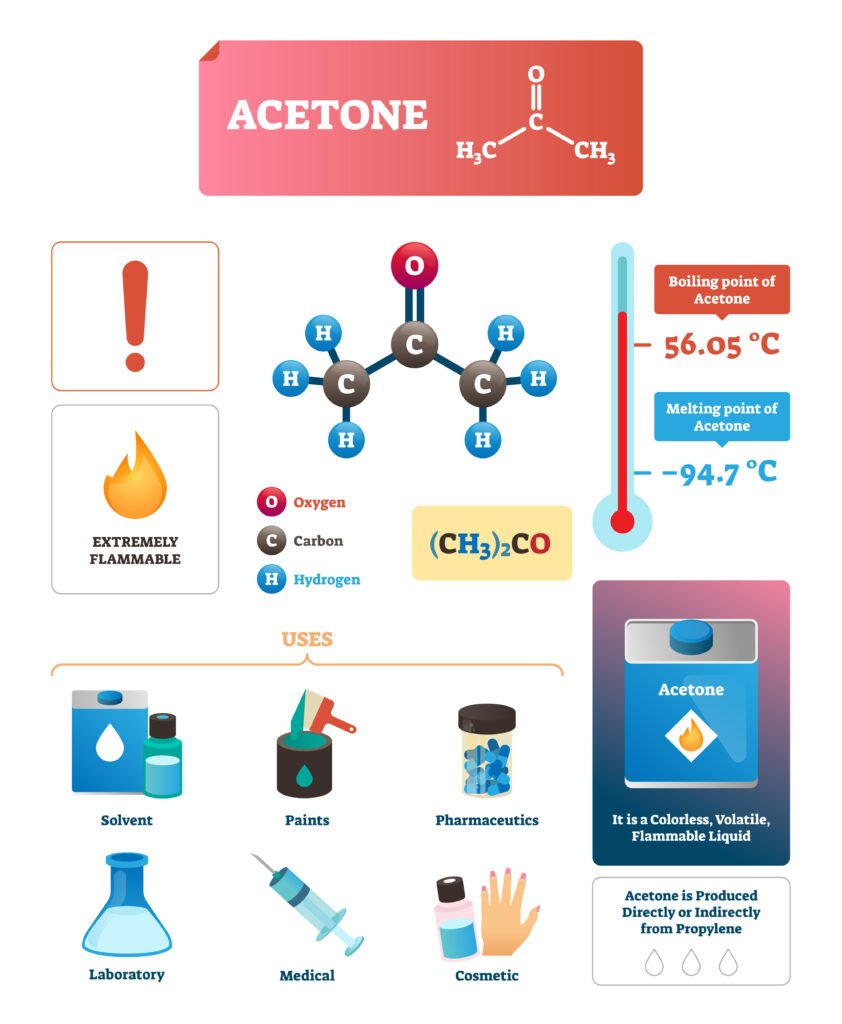
Ketones: Acetone (solvent), Acetophenone (perfume).
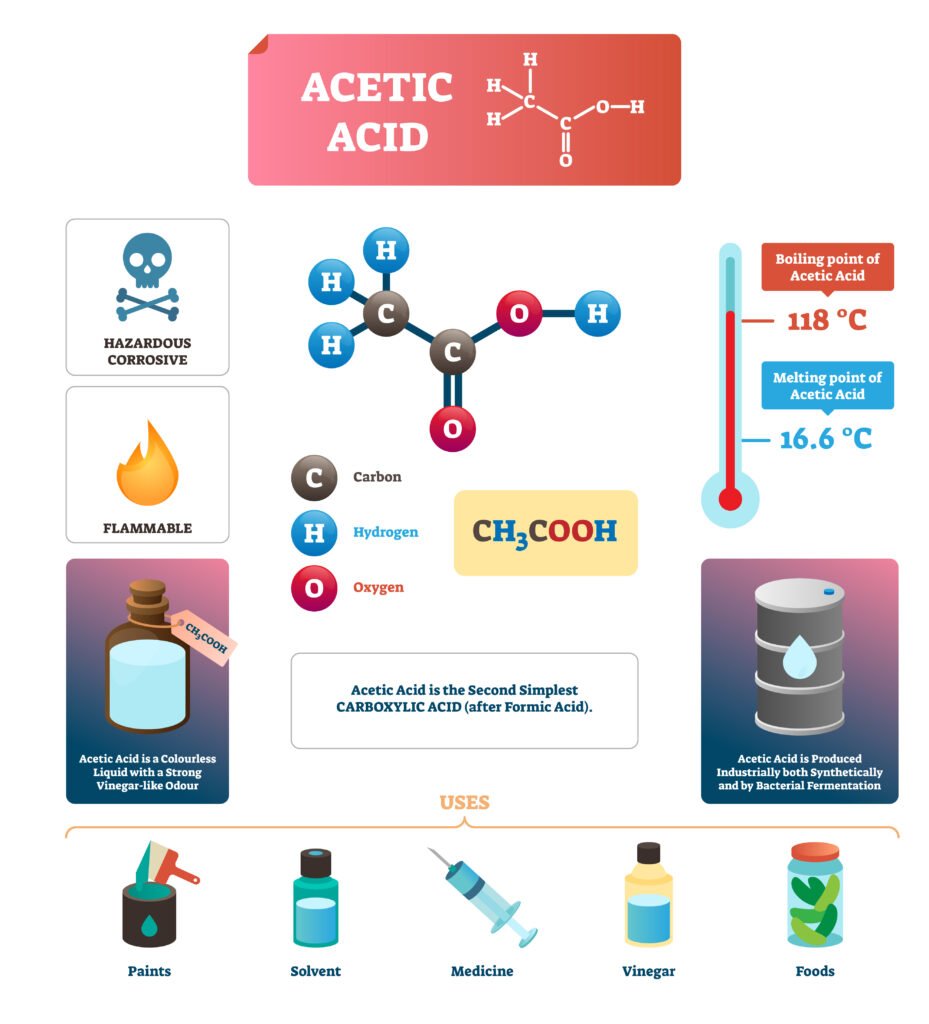
Carboxylic acids: Acetic acid (vinegar, esters, solvents), Benzoic acid (food preservative), Fatty acids (lipids).
📝 Summary
Aldehydes (-CHO), Ketones (>C=O), and Carboxylic acids (-COOH) are vital carbonyl compounds.
Nomenclature follows IUPAC rules: -al, -one, -oic acid endings.
Prepared by oxidation of alcohols, ozonolysis, Rosenmund reduction, Friedel–Crafts acylation (for aldehydes/ketones), and oxidation of primary alcohols, nitriles, Grignard reagents (for acids).
Physical properties depend on C=O polarity and hydrogen bonding. Acids have highest boiling points due to dimerisation.
Aldehydes/ketones undergo nucleophilic addition (HCN, NH₂-Z, Grignard reagents), oxidation (Tollen’s, Fehling’s for aldehydes), reduction (alcohols/hydrocarbons), and characteristic tests.
Carboxylic acids show acidic behaviour, esterification, salt formation, reduction, and decarboxylation.
Used extensively in industry (solvents, polymers, preservatives) and biology (lipids, metabolites).
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
Question 8.1
What is meant by the following terms? Give an example of the reaction in each case
Answer
🔵 (i) Cyanohydrin
Compound containing both –OH and –CN groups on the same carbon.
➡ Example: CH₃CHO + HCN → CH₃–CH(OH)–CN
🟢 (ii) Acetal
Product formed when aldehyde reacts with two equivalents of alcohol in presence of dry HCl.
➡ Example: CH₃CHO + 2C₂H₅OH → CH₃–CH(OC₂H₅)₂
🟠 (iii) Semicarbazone
Formed by reaction of aldehyde/ketone with semicarbazide.
➡ Example: CH₃COCH₃ + H₂N–NH–CONH₂ → CH₃C=NNHCONH₂
🔴 (iv) Aldol
β-Hydroxy aldehyde or ketone formed by aldol condensation.
➡ Example: 2CH₃CHO → CH₃–CH(OH)–CH₂–CHO
🟡 (v) Hemiacetal
Formed when one molecule of alcohol adds to an aldehyde.
➡ Example: CH₃CHO + C₂H₅OH → CH₃–CH(OH)(OC₂H₅)
🔵 (vi) Oxime
Compound formed by reaction of aldehyde/ketone with hydroxylamine.
➡ Example: CH₃CHO + NH₂OH → CH₃–CH=NOH
🟢 (vii) Ketal
Compound formed by ketone reacting with two equivalents of alcohol.
➡ Example: CH₃COCH₃ + 2C₂H₅OH → (C₂H₅O)₂C(CH₃)₂
🟠 (viii) Imine
Product formed when carbonyl compound reacts with primary amine.
➡ Example: CH₃CHO + C₂H₅NH₂ → CH₃–CH=NC₂H₅
🔴 (ix) 2,4-DNP derivative
Aldehydes/ketones react with 2,4-dinitrophenylhydrazine to give crystalline derivative.
➡ Example: CH₃CHO + 2,4-DNP → CH₃CH=NNH–C₆H₃(NO₂)₂
🟡 (x) Schiff’s base
Aldehyde/ketone reacts with aromatic primary amine to form –C=N– compound.
➡ Example: CH₃CHO + C₆H₅NH₂ → CH₃CH=NC₆H₅
Question 8.2
Name the following compounds according to IUPAC system of nomenclature:
Answer
🔵 (i) CH₃CH₂CH₂CH₂CHO → Pentanal
🟢 (ii) CH₃CH₂COCH₂CH₂CH₂Cl → 6-Chlorohexan-2-one
🟠 (iii) CH₃CH=CHCHO → But-2-enal
🔴 (iv) CH₃COCH₂COCH₃ → Pentane-2,4-dione
🟡 (v) CH₃CH₂CH₂CH₂COCH₃ → Hexan-2-one
🔵 (vi) (CH₃)₃C–COOH → 2,2-Dimethylpropanoic acid
🟢 (vii) OHCC₆H₄CHO-p → Benzene-1,4-dicarbaldehyde
Question 8.3
Draw the structures of the following compounds.
Answer
🔵 (i) 3-Methylbutanal → CH₃–CH(CH₃)–CH₂–CHO
🟢 (ii) p-Nitropropiophenone → O₂N–C₆H₄–CO–CH₂–CH₃
🟠 (iii) p-Methylbenzaldehyde → CH₃–C₆H₄–CHO (para-position)
🔴 (iv) 4-Methylpent-3-en-2-one → CH₃–C(=O)–CH=C(CH₃)–CH₃
🟡 (v) 4-Chloropentan-2-one → CH₃–CO–CHCl–CH₂–CH₃
🔵 (vi) 3-Bromo-4-phenylpentanoic acid → C₆H₅–CH₂–CH(Br)–CH₂–COOH
🟢 (vii) p,p′-Dihydroxybenzophenone → HO–C₆H₄–CO–C₆H₄–OH (para-para)
🟠 (viii) Hex-2-en-4-ynoic acid → CH₃–CH=CH–C≡C–COOH
Question 8.4
Write the IUPAC names of the following ketones and aldehydes. Wherever possible, give also common names.
Answer
🔵 (i) (CH₃)₂CH–CH₂–CHO → 3-Methylbutanal (common: isovaleraldehyde)
🟢 (ii) CH₃CH₂CH₂–CHO → Butanal (common: butyraldehyde)
🟠 (iii) CH₃(CH₂)₃CHO → Pentanal (common: valeraldehyde)
🔴 (iv) Ph–CH=CH–CHO → 3-Phenylprop-2-enal (common: cinnamaldehyde)
🟡 (v) CH₃COCH₂CH₂CH₃ → Pentan-2-one (common: methyl propyl ketone)
🔵 (vi) Ph–CO–Ph → Diphenylmethanone (common: benzophenone)
Question 8.5
Draw structures of the following derivatives:
Answer
🔵 (i) 2,4-Dinitrophenylhydrazone of benzaldehyde: C₆H₅–CH=N–NH–C₆H₃(NO₂)₂
🟢 (ii) Cyclopropanone oxime: C₃H₄=NOH (oxime form)
🟠 (iii) Acetaldehyde dimethylacetal: CH₃–CH(OMe)₂
🔴 (iv) Semicarbazone of cyclobutanone: cyclic C=NNHCONH₂
🟡 (v) Ethylene ketal of hexan-3-one: cyclic ketal structure with O–CH₂–CH₂–O
🔵 (vi) Methyl hemiacetal of formaldehyde: HO–CH₂–OCH₃
Question 8.6
Predict the products formed when cyclohexanecarbaldehyde reacts with following reagents:
Answer
🔵 (i) PhMgBr → Ph–CH(OH)–C₆H₁₁ (Grignard addition product)
🟢 (ii) Tollen’s reagent → C₆H₁₁–COOH (carboxylic acid, silver mirror test)
🟠 (iii) Semicarbazide + weak acid → Cyclohexane–CH=NNHCONH₂ (semicarbazone)
🔴 (iv) Excess ethanol + acid → Cyclohexane–CH(OEt)₂ (acetal)
🟡 (v) Zn–Hg + HCl → Cyclohexylmethane (Wolff–Kishner/Clemmensen reduction)
Question 8.7
Which of the following compounds would undergo aldol condensation, which the Cannizzaro reaction and which neither? Write the structures of the expected products:
(i) Methanal
(ii) 2-Methylpentanal
(iii) Benzaldehyde
(iv) Cyclohexanone
(v) Benzophenone
(vi) Cyclohexanone
(vii) Phenylacetaldehyde
(viii) Butan-1-ol
(ix) 2,2-Dimethylbutanal
Answer
🟦 Aldol condensation (compounds having α-H):
2-Methylpentanal, Cyclohexanone, Phenylacetaldehyde → give aldol products.
🟩 Cannizzaro reaction (no α-H atom):
Methanal, Benzaldehyde, 2,2-Dimethylbutanal → disproportionation (alcohol + acid).
🟨 Neither reaction:
Benzophenone (no aldehydic H, stable), Butan-1-ol (alcohol, not carbonyl).
Question 8.8
How will you convert ethanol into the following compounds?
(i) Butane-1,3-diol
(ii) But-2-enal
(iii) But-2-enoic acid
Answer
🔵 (i) Ethanol → Butane-1,3-diol
➡ Ethanol → Ethanal (oxidation with PCC)
➡ Ethanal + HCN → CH₃CH(OH)CN
➡ Hydrolysis → HOCH₂–CH₂–CH₂–OH
🟢 (ii) Ethanol → But-2-enal
➡ Ethanol → Ethanal (oxidation)
➡ Aldol condensation of 2CH₃CHO → CH₃–CH=CH–CHO
🟠 (iii) Ethanol → But-2-enoic acid
➡ Ethanol → Ethanal
➡ Aldol condensation → CH₃–CH=CH–CHO
➡ Oxidation → CH₃–CH=CH–COOH
Question 8.9
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. Indicate which aldehyde acts as nucleophile and which as electrophile.
Answer
🟦 Aldol condensation possible pairs:
Propanal + Propanal
➡ CH₃–CH₂–CH=CH–CHO (Pent-2-enal)
Butanal + Butanal
➡ CH₃–CH₂–CH=CH–CH₂–CHO (Hept-2-enal)
Propanal (nucleophile) + Butanal (electrophile)
➡ CH₃–CH₂–CH=CH–CH₂–CHO (Hex-2-enal)
Butanal (nucleophile) + Propanal (electrophile)
➡ CH₃–CH₂–CH=CH–CH₂–CHO (Isomeric Hex-2-enal)
✅ In each case, –CH₂– next to carbonyl acts as nucleophile.
Question 8.10
An organic compound with molecular formula C₇H₆O forms 2,4-DNP derivative, reduces Tollen’s reagent and undergoes Cannizzaro reaction. On vigorous oxidation, it gives 1,2-benzenedicarboxylic acid. Identify the compound.
Answer
🟦 Conditions:
C₇H₆O → aromatic aldehyde.
2,4-DNP positive → carbonyl present.
Tollen’s + Cannizzaro → aldehyde without α-H.
Oxidation → 1,2-benzenedicarboxylic acid → ortho-disubstituted aromatic.
🎯 Compound = o-Phthalaldehyde (benzene-1,2-dicarbaldehyde).
Question 8.11
An organic compound (A) (C₉H₁₀O) was hydrolysed with dilute H₂SO₄ to give a carboxylic acid (B) and alcohol (C). Oxidation of (C) with chromic acid gave (B) again. (C) on dehydration gave but-1-ene. Write equations.
Answer
🟦 Step 1: Hydrolysis → ester breakdown
A = Ester → C₄H₉–OOC–C₆H₅
🟩 Step 2: Products →
B = C₆H₅COOH (benzoic acid)
C = C₄H₉OH (butan-1-ol)
🟨 Step 3: Oxidation → C₄H₉OH → C₄H₉COOH (butanoic acid)
But in question, given B again → so (C) is actually CH₃CH₂CH₂CH₂OH (butanol).
🟧 Step 4: Dehydration → CH₃–CH₂–CH=CH₂ (but-1-ene).
✅ Hence, A = Butyl benzoate.
Question 8.12
Arrange the following compounds in increasing order of their property as indicated:
(i) Acetaldehyde, Acetone, Di-tert-butyl ketone, Methyl tert-butyl ketone (Reactivity towards HCN)
(ii) CH₃CH₂COOH, CH₃CH(Br)COOH, CH₃CH(Cl)COOH, (CH₃)₃CCOOH (Acid strength)
(iii) Benzoic acid, 4-Nitrobenzoic acid, 3,4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (Acid strength)
Answer
🔵 (i) Reactivity towards HCN (depends on steric hindrance):
Di-tert-butyl ketone < Methyl tert-butyl ketone < Acetone < Acetaldehyde
🟢 (ii) Acid strength (–I effect of substituents):
(CH₃)₃CCOOH < CH₃CH₂COOH < CH₃CH(Cl)COOH < CH₃CH(Br)COOH
🟠 (iii) Acid strength (–I/–R effect):
4-Methoxybenzoic acid < Benzoic acid < 4-Nitrobenzoic acid < 3,4-Dinitrobenzoic acid
Question 8.13
Give simple chemical tests to distinguish between the following pairs of compounds:
Answer
🔵 (i) Propanal and Propanone
Propanal: Gives positive Tollen’s (silver mirror) and Fehling’s test.
Propanone: Negative to both.
🟢 (ii) Acetophenone and Benzophenone
Acetophenone: Positive iodoform test (methyl ketone).
Benzophenone: Negative.
🟠 (iii) Phenol and Benzoic acid
Phenol: Neutral FeCl₃ → violet colour.
Benzoic acid: Effervescence with NaHCO₃.
🔴 (iv) Pentan-2-one and Pentan-3-one
Pentan-2-one: Positive iodoform test.
Pentan-3-one: Negative.
Question 8.14
How will you bring about the following conversions?
Answer
🔵 (i) Benzoic acid → Benzaldehyde
RCOOH → (RCOCl using SOCl₂) → Rosenmund reduction → RCHO.
🟢 (ii) Ethanol → 3-Hydroxybutanal
Ethanol → Ethanal (oxidation) → Aldol condensation → CH₃–CH(OH)–CH₂–CHO.
🟠 (iii) Propanal → 2-Methylpropan-2-ol
CH₃CH₂CHO + CH₃MgBr → CH₃CH₂–C(OH)(CH₃)₂.
🔴 (iv) Benzoic acid → m-Nitrobenzoic acid
Benzoic acid → Nitration (HNO₃/H₂SO₄) → m-nitrobenzoic acid (–COOH = m-directing).
🟡 (v) Benzaldehyde → Benzophenone
Benzaldehyde → Benzoin condensation → Benzoin → Oxidation → Benzil → Friedel–Crafts acylation → Benzophenone.
Question 8.15
How will you bring about the following conversions?
Answer
🔵 (i) Benzoic acid → Benzaldehyde → Benzyl alcohol
Benzoic acid → Benzaldehyde (Rosenmund reduction).
Benzaldehyde → Benzyl alcohol (reduction with NaBH₄).
🟢 (ii) Acetophenone → Benzoyl chloride
Acetophenone → Oxidation → Benzoic acid → SOCl₂ → Benzoyl chloride.
🟠 (iii) Propanal → 2-Methylpropan-2-ol
Same as Q8.14(iii).
Question 8.16
Describe the following reactions:
Answer
🔵 (i) Cannizzaro reaction
Disproportionation of aldehydes without α-H in conc. alkali.
➡ 2HCHO + NaOH → HCOONa + CH₃OH
🟢 (ii) Cross aldol condensation
Aldehydes/ketones with α-H react with another carbonyl compound.
➡ CH₃CHO + PhCHO → CH₃–CH(OH)–CH₂–PhCHO
🟠 (iii) Decarboxylation
Carboxylates heated with soda lime release CO₂.
➡ RCOONa + NaOH → RH + Na₂CO₃
Question 8.17
Give possible explanation for each of the following observations:
Answer
🔵 (i) Aldehydes are more reactive than ketones towards nucleophilic addition.
Less steric hindrance and greater +I effect in ketones → aldehydes are more electrophilic.
🟢 (ii) The boiling points of aldehydes and ketones are lower than those of alcohols of comparable molecular mass.
Aldehydes/ketones: Dipole–dipole interactions only.
Alcohols: Strong intermolecular H-bonding → higher boiling point.
🟠 (iii) Carboxylic acids have higher boiling points than aldehydes, ketones and alcohols of comparable molecular mass.
Carboxylic acids: Dimerise via strong H-bonding → very high boiling point.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
Section A (Q1–Q16) — MCQs (16 × 1 = 16 marks)
Options for Assertion–Reason Questions:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Question 1. The IUPAC name of CH₃–CH₂–CHO is:
🔵 (A) Propanal
🟢 (B) Propanone
🟠 (C) Acetone
🔴 (D) Ethanal
Answer: 🔵 (A) Propanal
Question 2. Which of the following undergoes Cannizzaro reaction?
🔵 (A) Acetone
🟢 (B) Formaldehyde
🟠 (C) Propanal
🔴 (D) Butanone
Answer: 🟢 (B) Formaldehyde
Question 3. Aldol condensation involves:
🔵 (A) Nucleophilic substitution
🟢 (B) Free radical substitution
🟠 (C) Enolate ion attack
🔴 (D) Electrophilic substitution
Answer: 🟠 (C) Enolate ion attack
Question 4. The product of iodoform test is yellow ppt of:
🔵 (A) CHI₃
🟢 (B) AgI
🟠 (C) KI
🔴 (D) I₂
Answer: 🔵 (A) CHI₃
Question 5. Oxidation of acetaldehyde gives:
🔵 (A) Acetic acid
🟢 (B) Ethanol
🟠 (C) Methanoic acid
🔴 (D) Propanoic acid
Answer: 🔵 (A) Acetic acid
Question 6. Which compound gives silver mirror test?
🔵 (A) Acetone
🟢 (B) Benzaldehyde
🟠 (C) Acetophenone
🔴 (D) 2-Butanone
Answer: 🟢 (B) Benzaldehyde
Question 7. Assertion (A): Carboxylic acids have higher boiling points than aldehydes/ketones.
Reason (R): They form dimers by hydrogen bonding.
Answer: 1
Question 8. Assertion (A): Acetone gives iodoform test.
Reason (R): It has –CO–CH₃ group.
Answer: 1
Question 9. The strongest acid among the following is:
🔵 (A) Formic acid
🟢 (B) Acetic acid
🟠 (C) Propanoic acid
🔴 (D) Benzoic acid
Answer: 🔵 (A) Formic acid
Question 10. Which of the following is not a nucleophilic addition reaction?
🔵 (A) Addition of HCN to aldehyde
🟢 (B) Aldol condensation
🟠 (C) Cannizzaro reaction
🔴 (D) Reduction to alcohol
Answer: 🟠 (C) Cannizzaro reaction
Question 11. The product of Clemmensen reduction of acetophenone is:
🔵 (A) Ethylbenzene
🟢 (B) Styrene
🟠 (C) Benzene
🔴 (D) Toluene
Answer: 🔵 (A) Ethylbenzene
Question 12. Which compound gives positive Fehling’s test?
🔵 (A) Benzaldehyde
🟢 (B) Acetaldehyde
🟠 (C) Acetone
🔴 (D) Formaldehyde
Answer: 🟢 (B) Acetaldehyde and 🔴 (D) Formaldehyde (both acceptable, aldehydes with α-H)
Question 13. Ethyl formate hydrolyses to give:
🔵 (A) Ethanol + Formic acid
🟢 (B) Methanol + Acetic acid
🟠 (C) Ethanol + Acetic acid
🔴 (D) Methanol + Formic acid
Answer: 🔵 (A) Ethanol + Formic acid
Question 14. Which of the following does not undergo decarboxylation easily?
🔵 (A) Sodium acetate
🟢 (B) Sodium formate
🟠 (C) Sodium benzoate
🔴 (D) Sodium oxalate
Answer: 🔴 (D) Sodium oxalate
Question 15. The mechanism of esterification involves:
🔵 (A) Electrophilic substitution
🟢 (B) Nucleophilic addition–elimination
🟠 (C) Free radical
🔴 (D) Oxidation
Answer: 🟢 (B) Nucleophilic addition–elimination
Question 16. Reduction of carboxylic acids with LiAlH₄ gives:
🔵 (A) Aldehydes
🟢 (B) Alcohols
🟠 (C) Ketones
🔴 (D) Hydrocarbons
Answer: 🟢 (B) Alcohols
Section B (Q17–Q21) — Very Short Answer (2 marks each)
Question 17. Write one difference between aldol condensation and Cannizzaro reaction.
Answer
🟦 Aldol: Requires α-H; gives β-hydroxy carbonyl compounds.
🟩 Cannizzaro: No α-H; gives alcohol + carboxylate.
Question 18. Write IUPAC name of the product formed in iodoform test with acetone.
Answer
🟦 Product: Triiodomethane (CHI₃).
🟩 IUPAC name: Triiodomethane.
Question 19. Write the structure of the compound formed when ethanol reacts with CH₃COOH in presence of H₂SO₄.
Answer
🟦 Reaction: CH₃CH₂OH + CH₃COOH ⇌ CH₃COOCH₂CH₃ + H₂O
🟩 Product: Ethyl ethanoate (Ester).
Question 20. Write the equation for decarboxylation of sodium acetate.
Answer
🟦 Reaction: CH₃COONa + NaOH → CH₄ + Na₂CO₃ (Δ, CaO).
Question 21. Why are aldehydes more reactive than ketones towards nucleophilic addition?
Answer
🟦 Aldehydes: Less steric hindrance, carbonyl C more δ⁺.
🟩 Ketones: Greater +I effect of two alkyl groups reduces reactivity.
Section C (Q22–Q28) — Short Answer (3 marks each)
Question 22. Write structures of aldol products from:
(i) CH₃CHO
(ii) CH₃COCH₃
Answer
🟦 (i) 2CH₃CHO → CH₃–CH(OH)–CH₂–CHO (3-Hydroxybutanal).
🟩 (ii) 2CH₃COCH₃ → CH₃–C(OH)–CH₂–COCH₃ (4-Hydroxy-4-methylpentan-2-one).
Question 23. What happens when benzaldehyde is treated with conc. NaOH?
Answer
🟦 No α-H → undergoes Cannizzaro reaction.
🟩 Products: Benzyl alcohol + Sodium benzoate.
Question 24. Give one chemical test to distinguish between:
(i) Acetone and Acetaldehyde
(ii) Formic acid and Acetic acid
Answer
🟦 (i) Acetaldehyde gives silver mirror with Tollen’s; acetone does not.
🟩 (ii) Formic acid decolourises alkaline KMnO₄ (as it can be oxidised further); acetic acid does not.
Question 25. Write a short note on esterification reaction.
Answer
🟦 Carboxylic acid + Alcohol → Ester + Water (in presence of H₂SO₄).
🟩 Example: CH₃COOH + CH₃CH₂OH ⇌ CH₃COOCH₂CH₃ + H₂O.
🟧 Mechanism: Protonation → nucleophilic attack → elimination.
Question 26. Write the mechanism of nucleophilic addition of HCN to a carbonyl group.
Answer
🟦 Step 1: CN⁻ attacks C=O carbon → tetrahedral alkoxide.
🟩 Step 2: Protonation of alkoxide → cyanohydrin (R₂C(OH)–CN).
Question 27. Arrange in increasing order of acidity: Formic acid, Acetic acid, Propanoic acid. Give reason.
Answer
🟦 Order: Propanoic acid < Acetic acid < Formic acid.
🟩 Reason: –I effect decreases with chain length; no alkyl in formic acid → strongest acid.
Question 28. Write the equation for Rosenmund reduction.
Answer
🟦 RCOCl + H₂ (Pd/BaSO₄, quinoline) → RCHO.
Section D (Q29–Q30) — Case-Based Questions (2 × 4 = 8 marks)
Question 29.
Read the passage and answer the questions:
Ethanal (CH₃CHO) undergoes self-condensation in presence of dilute alkali to give β-hydroxy aldehyde. On heating, it loses water to form an α,β-unsaturated aldehyde.
(i) Write the equation for the reaction.
(ii) What is the IUPAC name of the β-hydroxy aldehyde?
(iii) What is the IUPAC name of the α,β-unsaturated aldehyde formed?
Answer
🟦 (i) CH₃CHO + CH₃CHO ⇌ CH₃–CH(OH)–CH₂–CHO → (Δ) → CH₃–CH=CH–CHO
🟩 (ii) Product: 3-Hydroxybutanal
🟧 (iii) Product: But-2-enal
Question 30.
Read the passage and answer:
Formic acid behaves differently from other carboxylic acids. It is easily oxidised and also gives positive Tollen’s test.
(i) Write the oxidation product of formic acid.
(ii) Write the Tollen’s test reaction for formic acid.
(iii) Explain why formic acid shows this behaviour.
Answer
🟦 (i) Oxidation: HCOOH + [O] → CO₂ + H₂O
🟩 (ii) HCOOH + 2[Ag(NH₃)₂]⁺ + 3OH⁻ → 2Ag (mirror) + 2NH₃ + CO₂ + 2H₂O
🟧 (iii) Reason: Formic acid has –CHO group (aldehydic), hence acts like aldehyde.
Section E (Q31–Q33) — Long Answer Questions (3 × 5 = 15 marks)
Question 31.
Explain the mechanism of nucleophilic addition reaction of HCN to aldehydes and ketones.
OR
Describe the mechanism of esterification reaction of carboxylic acids with alcohols.
Answer
🟦 Mechanism of HCN addition
CN⁻ ion generated from HCN (in base) acts as nucleophile.
CN⁻ attacks carbonyl carbon → tetrahedral alkoxide intermediate.
Protonation of alkoxide (by HCN) → cyanohydrin (R₂C(OH)–CN).
✅ Final Product: Cyanohydrin.
🟩 OR – Mechanism of esterification
Protonation of C=O oxygen of R–COOH by H⁺.
Nucleophilic attack of alcohol (R′OH) on carbonyl carbon.
Proton transfer and elimination of water.
Deprotonation → Ester (RCOOR′).
Question 32.
Account for the following:
(i) Carboxylic acids have higher boiling points than aldehydes, ketones and alcohols of comparable mass.
(ii) Aldehydes are more reactive than ketones towards nucleophilic addition.
(iii) Aldehydes and ketones undergo nucleophilic addition reactions.
OR
Discuss the acidic strength of carboxylic acids. How is it affected by substituents?
Answer
🟦 (i) Carboxylic acids form strong H-bonded dimers → very high b.p.
🟩 (ii) Aldehydes: less steric hindrance, more δ⁺ carbonyl C.
🟧 (iii) Due to polar C=O bond, nucleophiles attack easily.
🟩 OR – Acidic strength
Carboxylic acids ionise: RCOOH ⇌ RCOO⁻ + H⁺.
Strength increases with –I substituents (Cl, NO₂).
Decreases with +I substituents (alkyl, –OCH₃).
Example order: CH₃COOH < ClCH₂COOH < NO₂CH₂COOH.
Question 33.
Explain with equations:
(i) Rosenmund reduction
(ii) Clemmensen reduction
(iii) Wolff–Kishner reduction
OR
Describe the following with examples:
(i) Aldol condensation
(ii) Cannizzaro reaction
(iii) Haloform test
Answer
🟦 (i) Rosenmund: RCOCl + H₂ (Pd/BaSO₄, quinoline) → RCHO
🟩 (ii) Clemmensen: RCOR′ + Zn(Hg)/HCl → RCH₂R′
🟧 (iii) Wolff–Kishner: RCOR′ + NH₂NH₂/KOH → RCH₂R′
🟩 OR
(i) Aldol: 2CH₃CHO → CH₃–CH(OH)–CH₂–CHO → CH₃–CH=CH–CHO
(ii) Cannizzaro: 2HCHO + NaOH → CH₃OH + HCOONa
(iii) Haloform: CH₃COCH₃ + I₂/NaOH → CHI₃ (yellow ppt) + salt.
————————————————————————————————————————————————————————————————————————————
