Class 11 : Physics (In English) – Chapter 11: Thermodynamics
EXPLANATION & SUMMARY
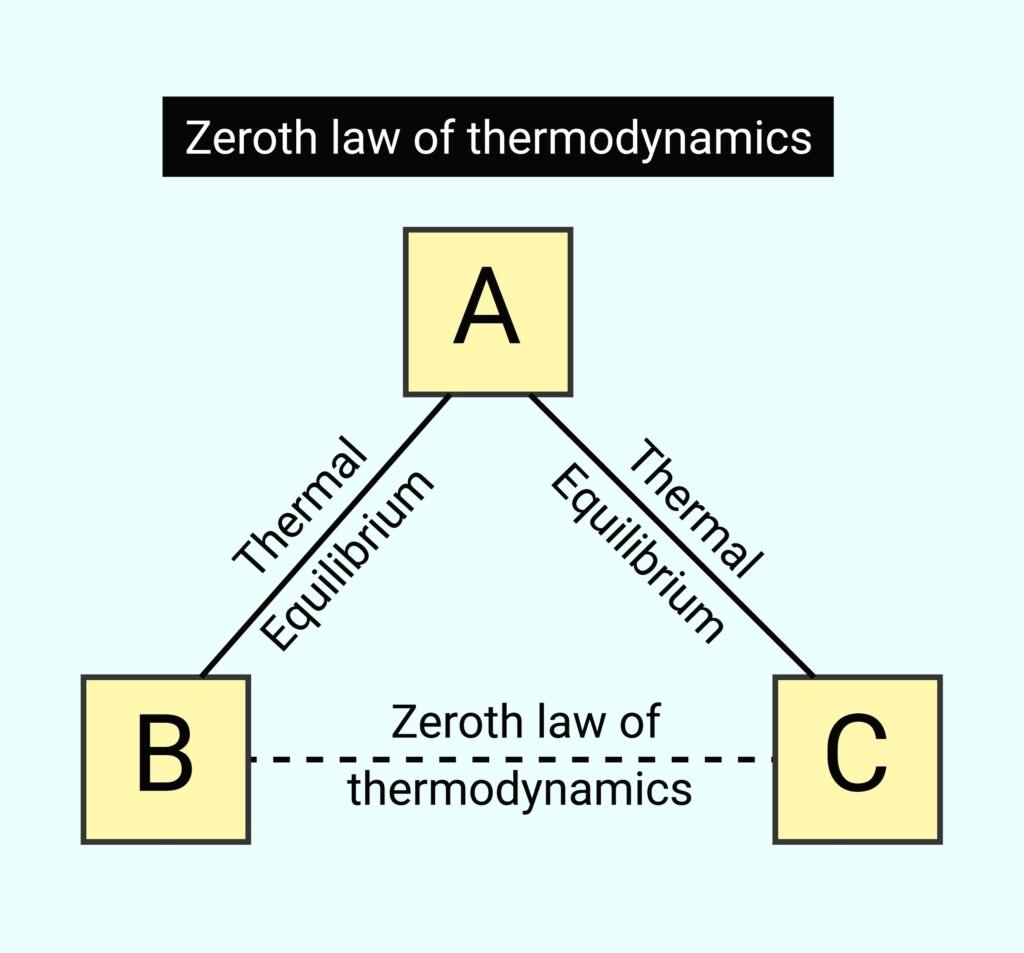
🔵 Introduction to Thermodynamics
Thermodynamics is the branch of physics that deals with the study of heat, work, and internal energy, and the interconversion between these forms of energy. It describes the macroscopic behaviour of systems in equilibrium without referring to molecular details.
💡 The term Thermodynamics originates from Greek:
Thermo → heat
Dynamics → power or motion
Hence, thermodynamics is the science of energy transformation involving heat and mechanical work.
🟢 System and Surroundings
A system is the specific part of the universe chosen for study, while everything else forms the surroundings.
✏️ Types of Systems:
🔹 Open System: Exchanges both matter and energy with surroundings (example: boiling water in an open pan).
🔹 Closed System: Exchanges energy but not matter (example: gas in a sealed cylinder).
🔹 Isolated System: Exchanges neither matter nor energy (example: thermos flask).
💡 The boundary separating the system and surroundings may be real or imaginary, fixed or movable.
🔴 State Variables and State Functions
The macroscopic state of a system is defined by state variables such as pressure (P), volume (V), temperature (T), and internal energy (U).
✔️ A state function depends only on the present state, not on the path followed.
Examples: U, P, V, T, H, entropy (S).
❌ A path function depends on the process path.
Examples: heat (Q), work (W).
🟡 Thermodynamic Processes
A process shows how a system changes from one state to another.
✏️ Important Processes:
🔹 Isothermal Process (ΔT = 0):
Temperature remains constant.
For an ideal gas, PV = constant ⇒ P₁V₁ = P₂V₂
Work done: W = nRT ln(V₂/V₁)
🔹 Adiabatic Process (Q = 0):
No heat exchange.
PV^γ = constant (γ = C_p / C_v)
Work done: W = (P₁V₁ − P₂V₂) / (γ − 1)
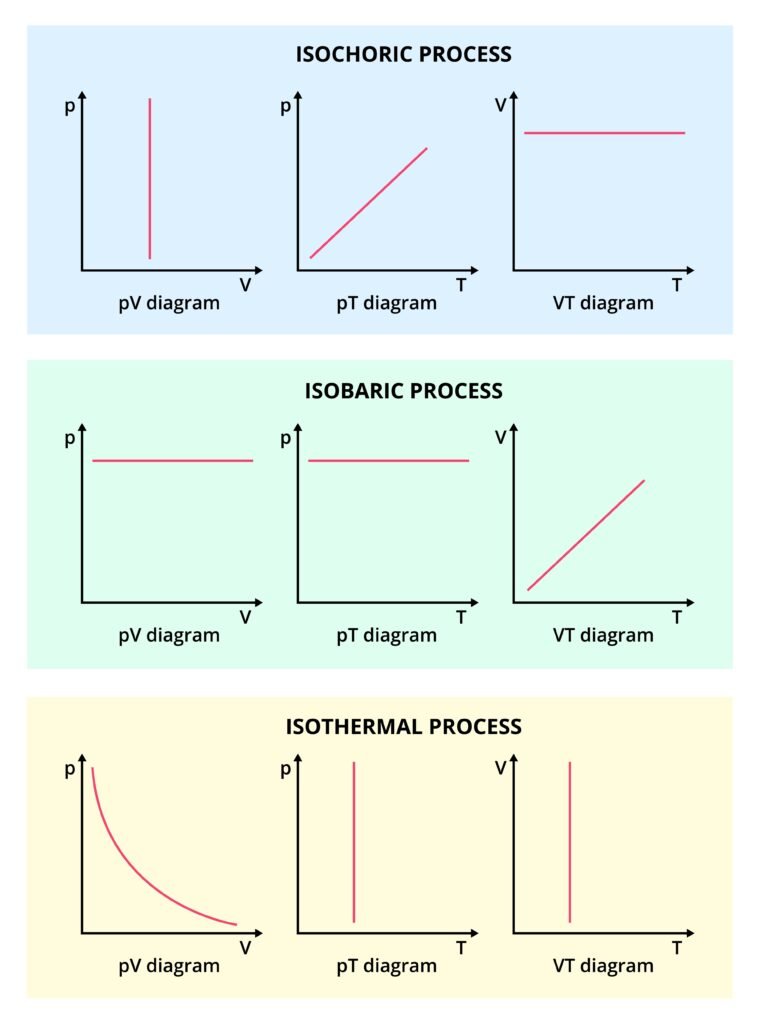
🔹 Isochoric Process (ΔV = 0):
Volume remains constant ⇒ W = 0
Heat supplied = Change in internal energy.
🔹 Isobaric Process (ΔP = 0):
Pressure constant ⇒ W = P(V₂ − V₁)
🔹 Cyclic Process:
System returns to the initial state ⇒ ΔU = 0 ⇒ Q = W.
🔵 Internal Energy (U)
Internal energy is the total microscopic energy of a system due to molecular motion and interactions.
It is a state function and for an ideal gas, depends only on temperature.
For a small change:
➡️ dU = δQ − δW
This represents the First Law of Thermodynamics.
🟢 Work Done by a Gas
When a gas expands against an external pressure P_ext,
➡️ δW = P_ext dV
For a reversible infinitesimal expansion,
➡️ W = ∫ P dV
For isothermal reversible process:
➡️ W = nRT ln(V₂/V₁)
For adiabatic process:
➡️ W = (P₁V₁ − P₂V₂) / (γ − 1)
🔴 Heat (Q)
Heat is energy transfer due to temperature difference between system and surroundings.
It is not a state function and depends on the path of the process.
Positive Q → heat absorbed by the system
Negative Q → heat released by the system
🟡 First Law of Thermodynamics
💡 Statement:
Energy can neither be created nor destroyed; it can only change forms.
➡️ ΔU = Q − W
where,
ΔU → change in internal energy
Q → heat supplied to the system
W → work done by the system
✔️ It expresses energy conservation in thermodynamics.
🔵 Specific Heat and Molar Specific Heat
Specific heat (c) = heat required to raise temperature of 1 kg of a substance by 1 K.
For gases, two molar specific heats are defined:
➡️ C_v → heat at constant volume
➡️ C_p → heat at constant pressure
Relation between them:
➡️ C_p − C_v = R
and
➡️ γ = C_p / C_v
For diatomic gases like N₂ or O₂, γ ≈ 1.4.
🟢 Work Done in Various Processes
✏️ For isothermal process: W = nRT ln(V₂/V₁)
✏️ For adiabatic process: W = (P₁V₁ − P₂V₂) / (γ − 1)
✏️ For isochoric process: W = 0
✏️ For isobaric process: W = P(V₂ − V₁)
💡 The area under the P–V curve represents the work done by the gas.
🔴 Reversible and Irreversible Processes
✔️ Reversible Process:
Occurs infinitely slowly through equilibrium states; can be reversed with no change in system or surroundings.
✔️ Irreversible Process:
Occurs rapidly, involves friction or turbulence, and cannot be reversed exactly.
Examples:
Free expansion of gas → irreversible
Slow isothermal expansion → reversible
🟡 Adiabatic Relation for Ideal Gas
For adiabatic process:
➡️ PV^γ = constant
Also,
➡️ T₁V₁^(γ−1) = T₂V₂^(γ−1)
and
➡️ T₁P₁^(1−γ) = T₂P₂^(1−γ)
🔵 Second Law of Thermodynamics
While the first law tells us energy is conserved, the second law tells us the direction of natural processes and introduces entropy.
💡 Kelvin–Planck Statement:
It is impossible to make a cyclic engine that converts all heat absorbed into work without any loss.
💡 Clausius Statement:
Heat cannot flow spontaneously from a colder body to a hotter body.
✔️ All spontaneous processes are irreversible and accompanied by increase in entropy.
🟢 Heat Engine
A heat engine converts heat into work.
✏️ Essential parts:
Heat source at high temperature T₁
Sink at low temperature T₂
Working substance (gas) operating cyclically
➡️ W = Q₁ − Q₂
➡️ η = W / Q₁ = 1 − (Q₂ / Q₁)
For a Carnot Engine:
➡️ η = 1 − (T₂ / T₁)
💡 Efficiency depends only on temperatures, not on the substance used.
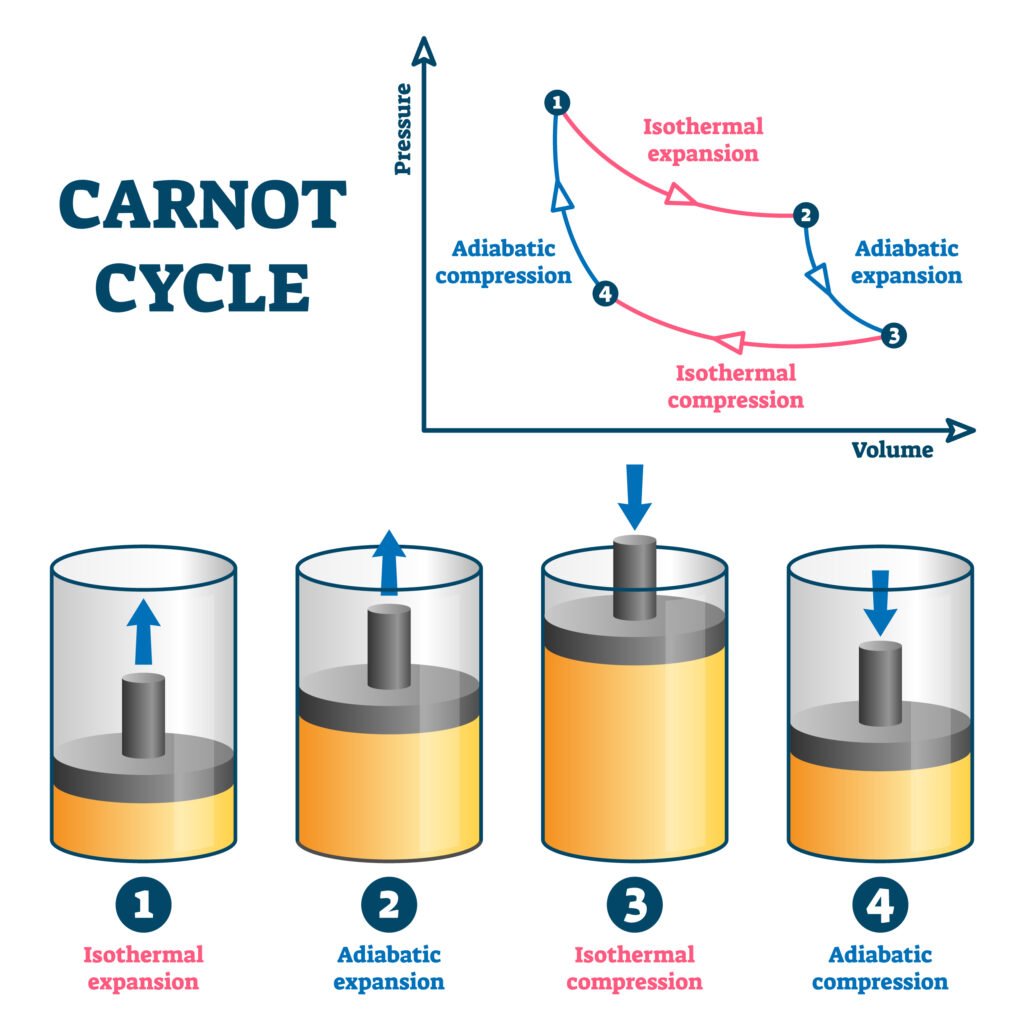
🔴 Refrigerator and Heat Pump
A refrigerator transfers heat from cold to hot region by using external work.
➡️ Q₂ = heat absorbed from cold region
➡️ Q₁ = heat rejected to hot region
➡️ W = Q₁ − Q₂
Coefficient of Performance (COP):
➡️ β = Q₂ / W = Q₂ / (Q₁ − Q₂)
💡 Heat pump works similarly but for heating rooms.
🟡 Entropy
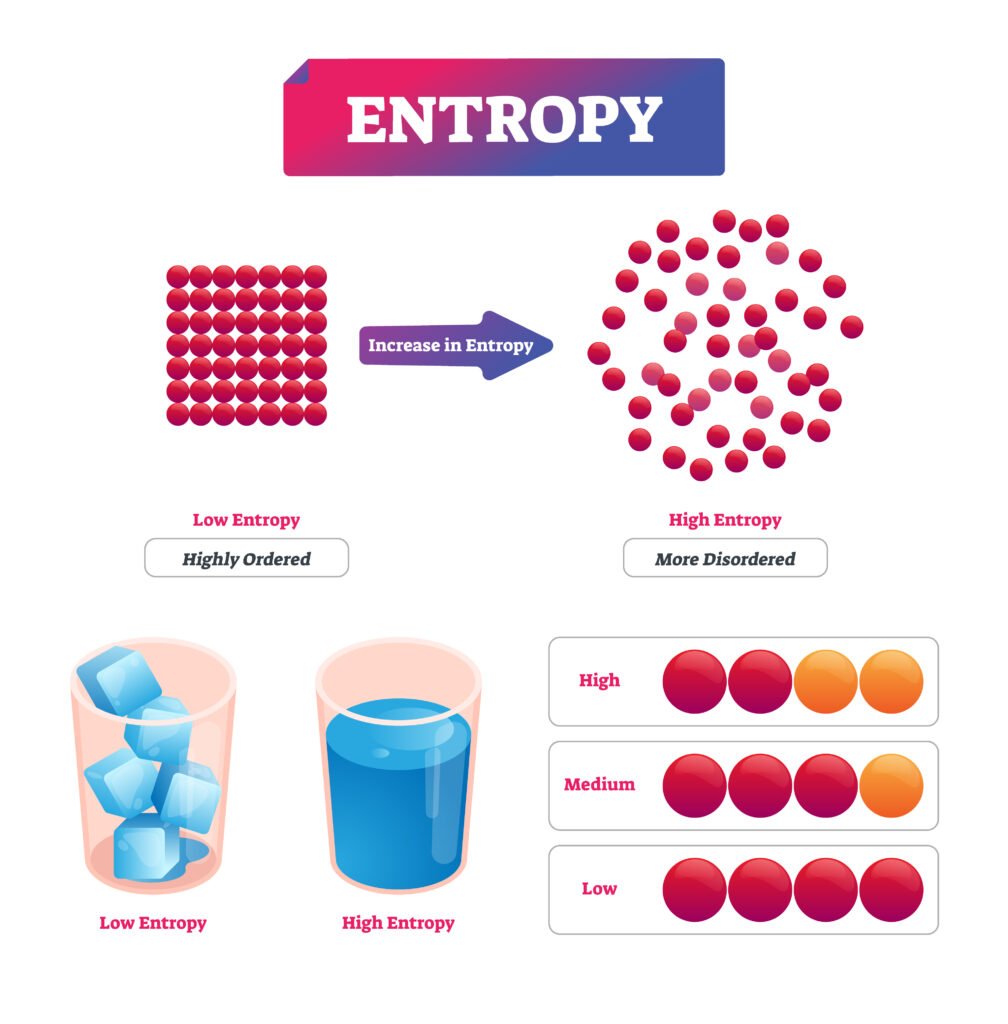
Entropy (S) is a measure of disorder or randomness.
For a reversible process:
➡️ dS = δQ_rev / T
✔️ In all natural processes, entropy of the universe increases (ΔS_total ≥ 0).
✔️ This is the entropy form of the second law.
🔵 Applications of Thermodynamics
✔️ Design of engines, refrigerators, and air conditioners.
✔️ Understanding energy conversion and efficiency.
✔️ Predicting feasibility of physical and chemical processes.
✔️ Explaining equilibrium and spontaneous changes in nature.
🌿 (2) Chapter Summary (~300 words)
Thermodynamics studies heat, work, and internal energy.
A system may be open, closed, or isolated.
The First Law: ΔU = Q − W expresses energy conservation.
For gases, C_p − C_v = R and γ = C_p / C_v.
In adiabatic process: PV^γ = constant.
Reversible processes are ideal; irreversible processes occur naturally.
The Second Law defines direction of heat flow and introduces entropy (S).
Efficiency of a Carnot engine: η = 1 − (T₂ / T₁).
Refrigerators and heat pumps transfer heat using external work.
Entropy of an isolated system never decreases.
Thermodynamics explains engines, power plants, chemical reactions, and atmospheric changes.
⚡ (3) Quick Recap (Key Points)
🔵 ΔU = Q − W → First Law of Thermodynamics
🟢 PV = nRT → Ideal Gas Law
🟡 PV^γ = constant → Adiabatic process
🔴 η = 1 − T₂/T₁ → Carnot efficiency
💡 dS = δQ_rev / T → Entropy change
✔️ In any spontaneous process, total entropy increases.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
Question 11.1
A geyser heats water flowing at the rate of 3.0 litres per minute from 27 °C to 77 °C. If the geyser operates on a gas burner, what is the rate of consumption of the fuel if its heat of combustion is 4.0 × 10⁴ J g⁻¹?
Answer
🔵 Given:
➡️ Volume flow rate = 3.0 L min⁻¹ ≡ 3.0 kg min⁻¹ (ρ₍water₎ ≈ 1 kg L⁻¹)
➡️ ΔT = 77 − 27 = 50 K
➡️ c₍water₎ ≈ 4.2 × 10³ J kg⁻¹ K⁻¹
➡️ Heat of combustion of fuel = 4.0 × 10⁴ J g⁻¹
🟢 Step 1: Heat supplied per minute
➡️ Q̇ (per minute) = ṁ × c × ΔT
➡️ = (3.0 kg min⁻¹) × (4.2 × 10³ J kg⁻¹ K⁻¹) × (50 K)
➡️ = 6.3 × 10⁵ J min⁻¹
🟡 Step 2: Convert to per second (power input as heat)
➡️ P = (6.3 × 10⁵ J) / 60 s = 1.05 × 10⁴ J s⁻¹
🔴 Step 3: Fuel consumption rate
➡️ ṁ₍fuel₎ = P / (heat of combustion)
➡️ = (1.05 × 10⁴ J s⁻¹) / (4.0 × 10⁴ J g⁻¹)
➡️ = 2.625 × 10⁻¹ g s⁻¹
✔️ Final: Fuel consumption rate ≈ 0.26 g s⁻¹ (≈ 15.8 g min⁻¹).
Question 11.2
What amount of heat must be supplied to 2.0 × 10² kg of nitrogen (at room temperature) to raise its temperature by 45 °C at constant pressure? (Molecular mass of N₂ = 28; R = 8.3 J mol⁻¹ K⁻¹.)
Answer
🔵 Given:
➡️ m = 2.0 × 10² kg, ΔT = 45 K
➡️ M(N₂) = 28 × 10⁻³ kg mol⁻¹
➡️ For diatomic gas (at room T): C_p = (7/2)R = 3.5R
🟢 Step 1: Number of moles
➡️ n = m / M = (200 kg) / (0.028 kg mol⁻¹) = 7.142857 × 10³ mol
🟡 Step 2: Heat at constant pressure
➡️ Q = n C_p ΔT = n × (3.5R) × ΔT
➡️ = (7.142857 × 10³) × (3.5 × 8.3 J mol⁻¹ K⁻¹) × (45 K)
🔴 Step 3: Compute
➡️ 3.5 × 8.3 = 29.05 J mol⁻¹ K⁻¹
➡️ n × C_p = (7.142857 × 10³) × 29.05 ≈ 2.075 × 10⁵ J K⁻¹
➡️ Q = (2.075 × 10⁵) × 45 ≈ 9.34 × 10⁶ J
✔️ Final: Q ≈ 9.3 × 10⁶ J (≈ 9.3 MJ).
Question 11.3
Explain why
(a) Two bodies at different temperatures T₁ and T₂ if brought in thermal contact do not necessarily settle to the mean temperature (T₁ + T₂)/2.
(b) The coolant in a chemical or a nuclear plant (i.e., the liquid used to prevent the different parts of a plant from getting too hot) should have high specific heat.
(c) Air pressure in a car tyre increases during driving.
(d) The climate of a harbour town is more temperate than that of a town in a desert at the same latitude.
Answer
(a) 🔵 Bodies approach a common final temperature that conserves energy:
➡️ m₁c₁(T_f − T₁) + m₂c₂(T_f − T₂) = 0 ⟹ T_f depends on masses & specific heats, not simply the arithmetic mean.
✔️ Hence, T_f ≠ (T₁ + T₂)/2 in general.
(b) 🟢 A high specific heat coolant can absorb large amounts of heat with small rise in temperature (Q = mcΔT).
✔️ This keeps plant components within safe temperature limits.
(c) 🟡 During driving, tyre air is compressed and heated due to flexing and friction; temperature rises ⇒ by ideal gas behaviour (p ∝ T at ~constant V), pressure increases.
(d) 🔴 Harbour towns have large water bodies nearby; water has high specific heat and provides thermal buffering (sea breeze/land breeze), reducing extremes.
✔️ Hence, more temperate climate than deserts at the same latitude.
Question 11.4
A cylinder with a movable piston contains 3 moles of hydrogen at standard temperature and pressure. The walls of the cylinder are made of a heat insulator, and the piston is insulated by a polythene sheet. So no heat can pass from the gas to the surroundings. The gas is compressed by pushing the piston inward, till its volume is halved: (a) what is the final temperature of the gas? (b) what is the work done on the gas? (c) what is the change in internal energy?
Answer
🔵 Given:
➡️ n = 3 mol H₂ (diatomic), initial at STP: T₁ = 273 K
➡️ Adiabatic (Q = 0), V₂ = V₁/2
➡️ γ (H₂ at room T) = 7/5 = 1.4; C_v = (5/2)R
(a) 🟢 Final temperature (adiabatic): T V^{γ−1} = const.
➡️ T₂ = T₁ (V₁/V₂)^{γ−1} = 273 × 2^{(1.4−1)} = 273 × 2^{0.4}
➡️ 2^{0.4} ≈ 1.32 ⇒ T₂ ≈ 360 K
(b) 🟡 For adiabatic: Q = 0 ⇒ ΔU = W_on (work done on gas)
➡️ ΔU = n C_v (T₂ − T₁) = 3 × (5/2 R) × (360 − 273)
➡️ = 3 × (2.5 × 8.3) × 87 = 3 × 20.75 × 87 ≈ 5.42 × 10³ J
➡️ Work done on gas W_on = ΔU ≈ 5.4 × 10³ J
(c) 🔴 Change in internal energy
➡️ ΔU ≈ +5.4 × 10³ J (increase)
✔️ Final: T₂ ≈ 360 K, W_on ≈ 5.4 kJ, ΔU ≈ +5.4 kJ.
Question 11.5
In changing the state of a gas adiabatically from an equilibrium state A to another equilibrium state B, an amount of work equal to 22.3 J is done on the system. If the gas is taken from state A to B via a process in which the net heat absorbed by the system is 9.35 cal, how much is the net work done by the system in the latter case? (Take 1 cal = 4.19 J)
Answer
🔵 Same initial and final states ⇒ ΔU is path independent.
➡️ Adiabatic path (Q = 0): work done on system = 22.3 J ⇒ ΔU = +22.3 J.
🟢 For the second path: Q = 9.35 cal = 9.35 × 4.19 J = 39.2 J (approx.)
➡️ First law (sign convention: W_by is work done by system): Q = ΔU + W_by
➡️ W_by = Q − ΔU = 39.2 − 22.3 = 16.9 J
✔️ Final: Net work done by the system ≈ 16.9 J.
Question 11.6
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
(a) What is the final pressure of the gas in A and B?
(b) What is the change in internal energy of the gas?
(c) What is the change in the temperature of the gas?
(d) Do the intermediate states of the system (before settling to the final equilibrium state) lie on its P–V–T surface?
Answer
🔵 Free expansion into vacuum; ideal gas; insulated.
(a) 🟢 Final volume becomes double, temperature (for ideal gas) unchanged ⇒ p_final = p_initial/2 = ½ atm.
(b) 🟡 For ideal gas, U = U(T) only; Joule expansion with Q = 0 and W_by = 0 ⇒ ΔU = 0.
(c) 🔴 Since ΔU = 0 for ideal gas ⇒ ΔT = 0.
(d) 💡 The opening is sudden (non-quasi-static); system is not in equilibrium during transition.
✔️ Intermediate states do not lie on the P–V–T surface.
Question 11.7
An electric heater supplies heat to a system at a rate of 100 W. If system performs work at a rate of 75 joules per second. At what rate is the internal energy increasing?
Answer
🔵 Given: Q̇ = 100 W = 100 J s⁻¹, work done by system Ẇ_by = 75 J s⁻¹.
🟢 First law (rate form): dU/dt = Q̇ − Ẇ_by
➡️ dU/dt = 100 − 75 = 25 J s⁻¹
✔️ Final: Internal energy increases at 25 W (25 J s⁻¹).
Question 11.8
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in Fig. (11.11).
Its volume is then reduced to the original value from E to F by an isobaric process. Calculate the total work done by the gas from D to E to F.
Answer
🔵 From the P–V graph (Fig. 11.11):
➡️ D( V = 2.0 m³, P = 600 kPa ), E( V = 5.0 m³, P = 300 kPa ), F( V = 2.0 m³, P = 300 kPa ).
➡️ Path: D → E (linear) then E → F (isobaric at 300 kPa).
(Use SI: 600 kPa = 6.0 × 10⁵ Pa; 300 kPa = 3.0 × 10⁵ Pa.)
🟢 Step 1: Work along D → E (area under straight line = trapezium)
➡️ W_DE = [(P_D + P_E)/2] × (V_E − V_D)
➡️ = [(6.0 × 10⁵ + 3.0 × 10⁵)/2] × (5.0 − 2.0)
➡️ = (4.5 × 10⁵) × 3.0 = 1.35 × 10⁶ J
🟡 Step 2: Work along E → F (isobaric compression)
➡️ W_EF = P × (V_F − V_E) = (3.0 × 10⁵) × (2.0 − 5.0)
➡️ = −9.0 × 10⁵ J (negative since volume decreases; work done by gas is negative)
🔴 Step 3: Total work (D → E → F)
➡️ W_total = W_DE + W_EF = (1.35 × 10⁶) + (−9.0 × 10⁵)
➡️ = 4.5 × 10⁵ J
✔️ Final: Total work done by the gas = 4.5 × 10⁵ J.
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
🌡️ SECTION A – Multiple Choice Questions (Q1–Q18)
Q1. The First Law of Thermodynamics is a statement of:
🔵 (A) Conservation of energy
🟢 (B) Conservation of mass
🟠 (C) Conservation of heat only
🔴 (D) Conservation of momentum
Answer: (A) Conservation of energy
Q2. In an adiabatic process, there is:
🔵 (A) No change in pressure
🟢 (B) No heat exchange
🟠 (C) No change in temperature
🔴 (D) Constant volume
Answer: (B) No heat exchange
Q3. For an isothermal expansion of an ideal gas, the work done is given by:
🔵 (A) W = nRT ln(V₂/V₁)
🟢 (B) W = nC_vΔT
🟠 (C) W = P(V₂ − V₁)
🔴 (D) W = 0
Answer: (A) W = nRT ln(V₂/V₁)
Q4. For an ideal gas, the internal energy depends only on:
🔵 (A) Volume
🟢 (B) Pressure
🟠 (C) Temperature
🔴 (D) Density
Answer: (C) Temperature
Q5. In a cyclic process, the change in internal energy (ΔU) is:
🔵 (A) Maximum
🟢 (B) Minimum
🟠 (C) Zero
🔴 (D) Infinite
Answer: (C) Zero
Q6. Work done in an isochoric process is:
🔵 (A) Zero
🟢 (B) Positive
🟠 (C) Negative
🔴 (D) Infinite
Answer: (A) Zero
Q7. The ratio of molar specific heats (C_p / C_v) is called:
🔵 (A) γ (Gamma)
🟢 (B) β
🟠 (C) η
🔴 (D) α
Answer: (A) γ (Gamma)
Q8. The efficiency of a Carnot engine working between temperatures T₁ and T₂ is:
🔵 (A) 1 + T₂/T₁
🟢 (B) 1 − T₂/T₁
🟠 (C) T₁/T₂
🔴 (D) T₂/T₁
Answer: (B) 1 − T₂/T₁
Q9. For an ideal gas, C_p − C_v equals:
🔵 (A) γ
🟢 (B) nR
🟠 (C) R
🔴 (D) P
Answer: (C) R
Q10. During free expansion of an ideal gas:
🔵 (A) Work is done on the gas
🟢 (B) Work is done by the gas
🟠 (C) No work is done
🔴 (D) Temperature increases
Answer: (C) No work is done
Q11. Which of the following is not a state function?
🔵 (A) Pressure
🟢 (B) Volume
🟠 (C) Heat
🔴 (D) Internal energy
Answer: (C) Heat
Q12. If a system absorbs 100 J of heat and does 40 J of work, its internal energy change is:
🔵 (A) 60 J
🟢 (B) 140 J
🟠 (C) 40 J
🔴 (D) 100 J
Answer: (A) 60 J
Q13. The process PV = constant is:
🔵 (A) Adiabatic
🟢 (B) Isothermal
🟠 (C) Isochoric
🔴 (D) Isobaric
Answer: (B) Isothermal
Q14. In a reversible process:
🔵 (A) System passes through equilibrium states
🟢 (B) System changes abruptly
🟠 (C) Heat loss is maximum
🔴 (D) Entropy decreases
Answer: (A) System passes through equilibrium states
Q15. Entropy remains constant in:
🔵 (A) Isothermal process
🟢 (B) Adiabatic reversible process
🟠 (C) Irreversible process
🔴 (D) Isochoric process
Answer: (B) Adiabatic reversible process
Q16. A Carnot engine operates between 400 K and 300 K. Its efficiency is:
🔵 (A) 0.75
🟢 (B) 0.25
🟠 (C) 0.5
🔴 (D) 0.33
Answer: (B) 0.25
Q17. For an ideal gas, internal energy is directly proportional to:
🔵 (A) Pressure only
🟢 (B) Volume only
🟠 (C) Temperature only
🔴 (D) Heat supplied
Answer: (C) Temperature only
Q18. Second law of thermodynamics gives:
🔵 (A) Direction of heat flow
🟢 (B) Conservation of heat
🟠 (C) Conservation of mass
🔴 (D) None of these
Answer: (A) Direction of heat flow
⚙️ SECTION B – Very Short / Short Answers (Q19–Q23)
Q19. Define the First Law of Thermodynamics.
Answer:
✏️ It states that the total energy of an isolated system remains constant; energy can neither be created nor destroyed but can change form.
➡️ Mathematically: ΔU = Q − W
Q20. What is an adiabatic process?
Answer:
💡 A thermodynamic process where no heat exchange takes place (Q = 0).
➡️ Relation: PV^γ = constant
Q21. What are state variables?
Answer:
🔵 Quantities that define the thermodynamic state of a system such as P, V, T, U.
✔️ They depend only on state, not on path.
Q22. Write relation between C_p and C_v for an ideal gas.
Answer:
➡️ C_p − C_v = R
💡 Here, R = universal gas constant.
Q23. Give one example each of reversible and irreversible processes.
Answer:
✔️ Reversible → Isothermal expansion of gas in equilibrium.
✔️ Irreversible → Free expansion of gas in vacuum.
🔬 SECTION C – Mid-Length Questions (Q24–Q28)
Q24. Derive work done in an isothermal expansion of an ideal gas.
Answer:
🔵 For isothermal process (T constant): PV = nRT
🟢 Work done, W = ∫ P dV = nRT ∫(dV/V)
🟡 On integration, W = nRT ln(V₂/V₁)
✔️ Hence, work done by gas during isothermal expansion = nRT ln(V₂/V₁)
Q25. Derive relation between pressure and volume in an adiabatic process.
Answer:
✏️ For adiabatic process: δQ = 0 ⇒ dU = −δW
For ideal gas, U ∝ T ⇒ dU = nC_v dT
Also, δW = P dV
➡️ nC_v dT = −P dV
Using ideal gas law (PV = nRT):
P dV + V dP = nR dT
Eliminating dT, we get:
C_v (P dV + V dP)/R = −P dV
Simplify to obtain: PV^γ = constant
✔️ where γ = C_p / C_v.
Q26. Define isobaric, isochoric and isothermal processes.
Answer:
🔹 Isobaric: Pressure constant (ΔP = 0)
🔹 Isochoric: Volume constant (ΔV = 0)
🔹 Isothermal: Temperature constant (ΔT = 0)
Q27. Explain why work done in an adiabatic process is less than in an isothermal process.
Answer:
➡️ In adiabatic process, no heat enters; system uses its own internal energy to do work, so temperature falls quickly.
➡️ Hence, total work output (area under curve) is smaller than in isothermal expansion.
Q28. Write Kelvin–Planck and Clausius statements of Second Law of Thermodynamics.
Answer:
🔵 Kelvin–Planck: No engine can convert all absorbed heat into work.
🟢 Clausius: Heat cannot flow spontaneously from a colder body to a hotter body.
🧠 SECTION D – Long Answer Questions (Q29–Q31)
Q29. Explain the working of a Carnot engine and derive its efficiency.
Answer:
🔵 Carnot Engine: Ideal heat engine working reversibly between two reservoirs.
🟢 Steps:
1️⃣ Isothermal expansion at T₁ (absorbs Q₁)
2️⃣ Adiabatic expansion (temperature falls to T₂)
3️⃣ Isothermal compression at T₂ (rejects Q₂)
4️⃣ Adiabatic compression (returns to T₁)
Efficiency:
η = 1 − (T₂ / T₁)
✔️ Depends only on source and sink temperatures, not on working substance.
Q30. What is entropy? Derive expression for change in entropy for reversible process.
Answer:
💡 Entropy (S) is a measure of disorder of a system.
For a reversible process:
dS = δQ_rev / T
Integrating,
ΔS = Q_rev / T (if T constant)
✔️ It quantifies energy dispersal; ΔS ≥ 0 in all real processes.
Q31. Describe refrigerator as an application of Second Law of Thermodynamics.
Answer:
🔵 Principle: Heat is transferred from cold to hot region by external work.
🟢 Heat absorbed: Q₂; Rejected: Q₁; Work: W = Q₁ − Q₂
Coefficient of Performance (COP):
β = Q₂ / (Q₁ − Q₂)
✔️ It operates by cyclic compression and expansion of refrigerant, based on Clausius statement.
🌍 SECTION E – Case / Application Based (Q32–Q33)
Q32. A gas expands isothermally from 2.0 L to 6.0 L at 300 K. Calculate the work done. (Given: 1 mol gas, R = 8.3 J mol⁻¹ K⁻¹)
Answer:
🔵 W = nRT ln(V₂/V₁)
➡️ W = 1 × 8.3 × 300 × ln(6/2)
➡️ = 2490 × ln(3) = 2490 × 1.0986
➡️ W ≈ 2736 J
✔️ Work done = 2.74 × 10³ J
Q33. A Carnot engine operates between 500 K and 300 K. If it absorbs 800 J heat, calculate work done and heat rejected.
Answer:
🟢 η = 1 − T₂/T₁ = 1 − 300/500 = 0.4
➡️ W = η × Q₁ = 0.4 × 800 = 320 J
➡️ Q₂ = Q₁ − W = 800 − 320 = 480 J
✔️ Work done = 320 J, Heat rejected = 480 J.
————————————————————————————————————————————————————————————————————————————
