Class 11 : Chemistry (In English) – Chapter 3: Classification of elements and Periodicity in properties
EXPLANATION & SUMMARY
🔵 Introduction
Chemistry is the science of elements 🧪. By the 18th century, chemists discovered more than 30 elements, and by the 19th century, the number increased rapidly 🌍. As the list grew, arranging them in a logical way became necessary. Imagine learning chemistry if all 118 known elements today were memorized randomly without order ❌. That is why scientists searched for patterns among elements, leading to the development of the Periodic Table — one of the most powerful tools in chemistry.
This chapter focuses on the classification of elements and the periodic properties that recur in predictable patterns.
🟢 Early Attempts of Classification
1️⃣ Dobereiner’s Triads (1829)
Johann Dobereiner grouped elements in sets of three called triads.
The atomic mass of the middle element was roughly the average of the other two.
Example: Li (7), Na (23), K (39).
💡 Limitation: Could not include all elements.
2️⃣ Newlands’ Law of Octaves (1864)
John Newlands arranged elements by increasing atomic mass.
Every 8th element had similar properties, like notes in music 🎶.
Example: Li, Na, K showed similarity.
❌ Limitation: Worked only up to calcium, failed for heavier elements.
3️⃣ Mendeleev’s Periodic Law (1869)
Dmitri Mendeleev arranged elements in increasing atomic mass.
Elements with similar properties were placed in same groups.
Left gaps for undiscovered elements and predicted their properties accurately (e.g., Eka-aluminium → Gallium).
Law: “Properties of elements are periodic function of their atomic masses.”
❌ Limitation: Anomalies in order (Co, Ni), isotopes not explained.
🔵 Modern Periodic Law (1913)
Proposed by Henry Moseley using X-ray spectra.
Law: “Properties of elements are periodic function of their atomic numbers.”
Atomic number (Z = number of protons) became the basis.
Modern Periodic Table is arranged on this principle.
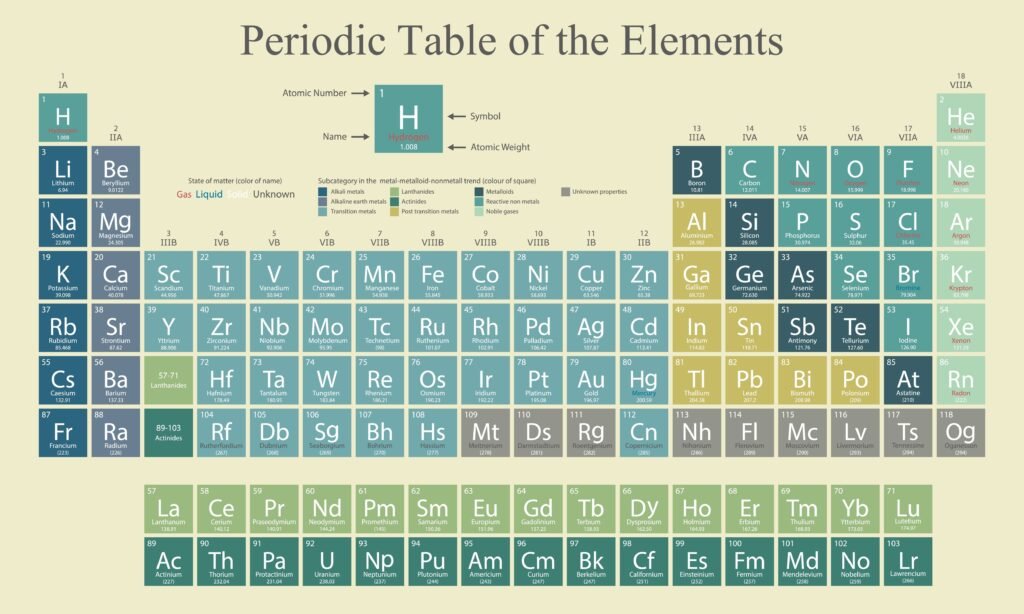
🟢 Structure of the Modern Periodic Table
✔ Arranged in 18 groups (vertical columns) and 7 periods (horizontal rows).
✔ Groups: Elements with similar valence shell electronic configuration.
✔ Periods: Elements with same principal quantum number (n).
✔ Classification:
Group 1 → Alkali metals.
Group 2 → Alkaline earth metals.
Group 17 → Halogens.
Group 18 → Noble gases.
d-block → Transition metals.
f-block → Inner transition elements (lanthanides + actinides).
🌟 Periodicity arises due to repetition of similar valence shell configuration.
🔵 Important Periodic Properties
Now let us explore properties that change periodically across periods and groups.
1️⃣ Atomic Radius (size of atom)
Distance from nucleus to outermost electron.
Across period (→): decreases due to increased nuclear charge pulling electrons closer.
Down group (↓): increases due to addition of shells.
2️⃣ Ionic Radius
Cations (loss of electron) → smaller radius ⚡.
Anions (gain of electron) → larger radius.
3️⃣ Ionization Enthalpy (IE)
Minimum energy to remove an electron.
Across period: increases due to higher nuclear charge.
Down group: decreases because outer electrons are farther, weaker attraction.
Exceptions: Filled/half-filled subshell stability (e.g., O vs N).
4️⃣ Electron Gain Enthalpy (EGE)
Energy change when an electron is added.
Across period: generally becomes more negative (favorable).
Down group: becomes less negative.
Halogens have high negative values. Noble gases ~0.
5️⃣ Electronegativity (EN)
Tendency to attract shared electrons in bond.
Across period: increases.
Down group: decreases.
Fluorine is most electronegative (3.98 Pauling).
6️⃣ Valency
Combining capacity of atom.
Depends on number of valence electrons.
Shows periodicity with group.
🟢 Trends and Anomalies
Diagonal Relationship: Elements of second period show similarities with diagonally placed elements of third period (e.g., Li–Mg, Be–Al).
Inert Pair Effect: Heavier p-block elements show lower oxidation states due to reluctance of s-electrons to participate.
d-block properties: Show variable oxidation states, form colored ions, good catalysts.
f-block elements: Lanthanide contraction influences periodic trends.
🔵 Significance of Periodic Table
✔ Provides systematic arrangement of all known elements.
✔ Predicts chemical and physical properties.
✔ Helps in understanding bonding, reactivity, trends.
✔ Basis of modern chemical research and discovery of new elements.
✨ Conclusion
From Dobereiner’s triads to Moseley’s atomic number law, the periodic table is a triumph of scientific classification 🌍. Periodicity arises from repeating patterns in electronic configuration, which governs properties. This makes the table not just a chart, but a powerful predictive model of matter.
🔹 II. Lesson Summary
📌 Classification of Elements and Periodicity – Key Takeaways
🔵 Early Classification:
Dobereiner’s Triads → average atomic mass rule.
Newlands’ Octaves → repetition every 8th element.
Mendeleev → periodic law of atomic masses, predicted new elements.
🟢 Modern Periodic Law:
Moseley: atomic number as basis.
Periodic properties depend on electronic configuration.
🔴 Structure of Modern Table:
18 groups, 7 periods.
s, p, d, f blocks.
Group trends: alkali metals, halogens, noble gases.
🟡 Periodic Properties:
Atomic radius → decreases across, increases down.
Ionization enthalpy → increases across, decreases down.
Electron gain enthalpy → more negative across, less negative down.
Electronegativity → increases across, decreases down.
Valency → periodic repetition.
🌟 Anomalies:
Diagonal relationships.
Inert pair effect in heavy p-block.
d-block variability, colored ions, catalytic behavior.
Lanthanide contraction.
📚 Importance:
Explains order among elements.
Predicts unknown elements.
Essential tool for chemistry and periodic trends.
📝 Quick Recap:
✔ Classification of elements evolved from triads → octaves → Mendeleev → modern periodic table.
✔ Modern law: properties depend on atomic number, not mass.
✔ Periodic table: 18 groups, 7 periods, 4 blocks (s, p, d, f).
✔ Periodic properties: atomic size, ionization enthalpy, electron gain enthalpy, electronegativity, valency.
✔ Trends explained by nuclear charge and shell structure.
✔ Special features: diagonal relationship, inert pair effect, lanthanide contraction.
✔ Periodic table remains the backbone of chemistry, predicting properties and guiding discovery.
————————————————————————————————————————————————————————————————————————————
QUESTIONS FROM TEXTBOOK
🔵 Question 3.1
What is the basic theme of organisation in the periodic table?
🟢 Answer:
✔ The periodic table is based on the Modern Periodic Law:
“The physical and chemical properties of elements are the periodic functions of their atomic numbers.”
✔ Therefore, atomic number (Z) decides an element’s position.
✔ Elements are arranged in increasing Z across periods (rows) and similar-property groups (columns).
➡ This allows prediction of chemical behaviour and trends.
🔵 Question 3.2
Which important property did Mendeleev use to classify the elements in his periodic table and did he stick to that?
🟢 Answer:
✔ He arranged elements by increasing atomic mass.
✔ But, to keep similar properties in one column, he interchanged some (e.g. Co & Ni).
➡ Thus he did not strictly follow mass order—chemical similarity was given priority.
🔵 Question 3.3
What is the basic difference in approach between the Mendeleev’s Periodic Law and the Modern Periodic Law?
🟢 Answer:Mendeleev Modern
Basis Atomic mass Atomic number
Statement Properties are periodic functions of mass Properties are periodic functions of number
✔ Shift to atomic number removed anomalies like Co–Ni.
🔵 Question 3.4
On the basis of quantum numbers, justify that the sixth period of the periodic table should have 32 elements.
🟢 Answer:
Electrons fill:
6s → 2 - 4f → 14 - 5d → 10 - 6p → 6
Total = 2 + 14 + 10 + 6 = 32 elements ✅
🔵 Question 3.5
In terms of period and group, where would you locate the element with Z = 114?
🟢 Answer:
Configuration = [Rn] 5f¹⁴ 6d¹⁰ 7s² 7p²
→ Outer 7s² 7p² = Group 14 (p-block)
→ Highest n = 7 = Period 7
➡ Position: Period 7, Group 14, p-block
🔵 Question 3.6
Write the atomic number of the element present in the third period and seventeenth group of the periodic table.
🟢 Answer:
3rd period range = Z 11–18; Group 17 = halogens → Cl (Z = 17) ✅
🔵 Question 3.7
Which element do you think would have been named by
(i) Lawrence Berkeley Laboratory (ii) Seaborg’s group?
🟢 Answer:
(i) Berkelium (Bk) (ii) Seaborgium (Sg) ✅
🔵 Question 3.8
Why do elements in the same group have similar physical and chemical properties?
🟢 Answer:
✔ Same valence-shell configuration ⇒ same valence electrons
➡ Hence similar bonding & chemical behaviour (e.g. Group 1: ns¹).
🔵 Question 3.9
What does atomic radius and ionic radius really mean to you?
🟢 Answer:
Atomic radius: half the distance between nuclei of two identical atoms joined by a covalent bond.
Ionic radius: distance from nucleus to outermost e⁻ in an ion.
💡 Cations < parent atom; Anions > parent atom.
🔵 Question 3.10
How do atomic radii vary in a period and in a group? Explain.
🟢 Answer:
Across a period: ↓ radius (↑ Z → stronger attraction)
Down a group: ↑ radius (new shells added)
🔵 Question 3.11
What do you understand by isoelectronic species? Name one isoelectronic species with each:
(i) F⁻ (ii) Ar (iii) Mg²⁺ (iv) Rb⁺
🟢 Answer:
Isoelectronic = same total electrons.
(i) F⁻ (10 e⁻) → Ne
(ii) Ar (18 e⁻) → K⁺
(iii) Mg²⁺ (10 e⁻) → Na⁺
(iv) Rb⁺ (36 e⁻) → Kr
🔵 Question 3.12
Species: N³⁻, O²⁻, F⁻, Na⁺, Mg²⁺, Al³⁺
(a) Common feature (b) Order of increasing ionic radius
🟢 Answer:
(a) All have 10 e⁻ ⇒ isoelectronic.
(b) Higher Z ⇒ smaller size
Order: Al³⁺ < Mg²⁺ < Na⁺ < F⁻ < O²⁻ < N³⁻
🔵 Question 3.13
Explain why cations are smaller and anions larger than their parent atoms.
🟢 Answer:
Cations: e⁻ lost → repulsion ↓ → effective Z ↑ → radius ↓
Anions: e⁻ gained → repulsion ↑ → effective Z ↓ → radius ↑ ✅
🔵 Question 3.14
What is the significance of the terms “isolated gaseous atom” and “ground state” while defining ionization enthalpy and electron gain enthalpy?
🟢 Answer:
✔ Isolated: Atom not influenced by neighbours → true intrinsic energy.
✔ Gaseous: No intermolecular forces; easy to compare.
✔ Ground state: Lowest-energy configuration; ensures standard reference.
➡ These conditions make the values comparable for all elements.
🔵 Question 3.15
Energy of electron in ground-state H-atom = –2.18 × 10⁻¹⁸ J. Find ionization enthalpy (kJ mol⁻¹).
🟢 Answer:
✳ For 1 atom → 2.18 × 10⁻¹⁸ J
✳ For 1 mol → 2.18 × 10⁻¹⁸ × 6.022 × 10²³ = 1.31 × 10⁶ J mol⁻¹
➡ = 1312 kJ mol⁻¹ ✅
🔵 Question 3.16
Order of actual first ionization enthalpies (kJ mol⁻¹):
Li < B < Be < C < O < N < F < Ne. Explain: (i) Be > B (ii) O < N, F
🟢 Answer:
(i) Be: 2s² (filled) → stable → harder to remove e⁻ → IE ↑
(ii) O: 2p⁴ has paired e⁻ → repulsion → easier removal → IE ↓
✔ Hence given order is correct.
🔵 Question 3.17
Why is IE₁(Na) < IE₁(Mg) but IE₂(Na) ≫ IE₂(Mg)?
🟢 Answer:
Na: 1s²2s²2p⁶3s¹ → removing 3s¹ gives noble-gas core → IE₁ small, IE₂ huge.
Mg: 1s²2s²2p⁶3s² → first e⁻ from 3s², second from 3s¹ → both relatively easy.
➡ Therefore IE₂(Na) ≫ IE₂(Mg). ✅
🔵 Question 3.18
Why does ionization enthalpy decrease down a group?
🟢 Answer:
✔ Atomic size ↑
✔ Shielding ↑
✔ Effective nuclear attraction on valence e⁻ ↓
➡ Easier removal → IE ↓
🔵 Question 3.19
IE₁ (kJ mol⁻¹): B 801, Al 577, Ga 579, In 558, Tl 589. Explain irregularity.
🟢 Answer:
d-/f-electrons in Ga, In, Tl give poor shielding → Z_eff ↑ → IE slightly higher than smooth trend → deviation observed.
🔵 Question 3.20
Which pair has more negative electron gain enthalpy?
(i) O or F (ii) F or Cl
🟢 Answer (corrected):
(i) F < O → F has more negative ΔegH.
(ii) Cl has more negative ΔegH than F (lower repulsion). ✅
🔵 Question 3.21
Second electron-gain enthalpy of O — positive or negative?
🟢 Answer:
Adding e⁻ to O⁻ → O²⁻ faces repulsion → energy absorbed → positive value ✅
🔵 Question 3.22
Difference between electron-gain enthalpy and electronegativity?
🟢 Answer:
Property Electron-gain enthalpy Electronegativity
Definition Energy change on gaining e⁻ Tendency to attract shared e⁻
Type Thermodynamic quantity (kJ mol⁻¹) Relative scale (dimensionless)
Depends on Isolated atom Bonded atom
🔵 Question 3.23
Electronegativity of N is 3.0 in all compounds — true?
🟢 Answer: ❌ False
Electronegativity varies with hybridization, oxidation state, environment; hence not constant.
🔵 Question 3.24
Effect on radius when atom
(a) gains e⁻ (b) loses e⁻
🟢 Answer:
(a) Gain → anion → e–e repulsion ↑ → radius ↑
(b) Loss → cation → Z_eff ↑ → radius ↓ ✅
🔵 Question 3.25
Will isotopes have same first IE?
🟢 Answer:
✔ Same Z and electronic structure → nearly same IE; tiny difference due to mass negligible.
🔵 Question 3.26
Main differences between metals and non-metals:
🟢 Answer:
Metals: lustrous, good conductors, low IE, form cations, basic oxides.
Non-metals: dull, poor conductors, high IE, form anions, acidic oxides.
🔵 Question 3.27
Use periodic table to find:
(a) element with five e⁻ in outer subshell
(b) element tending to lose two e⁻
(c) element tending to gain two e⁻
(d) group containing metal, non-metal, liquid, gas
🟢 Answer (corrected):
(a) F or Cl → outer p⁵
(b) Mg (loses 2 e⁻)
(c) O (gains 2 e⁻)
(d) Group 17:
- Gases F₂, Cl₂ - Liquid Br₂ - Solid I₂ - At shows metallic behaviour ✅
🔵 Question 3.28
Explain why the reactivity order is:
Group 1: Li < Na < K < Rb < Cs Group 17: F > Cl > Br > I
🟢 Answer:
✔ Group 1 (alkali metals) lose one electron to form M⁺.
➡ Down the group:
Atomic size ↑
Ionization enthalpy ↓
→ Electron loss easier → reactivity increases
✅ Order: Li < Na < K < Rb < Cs
✔ Group 17 (halogens) gain one electron to form X⁻.
➡ Down the group:
Size ↑
Electron-gain enthalpy becomes less negative
→ Harder to gain e⁻ → reactivity decreases
✅ Order: F > Cl > Br > I
🔵 Question 3.29
Give the general outer electronic configuration of each block.
🟢 Answer:
s-block: (n)s¹–²
p-block: (n)s² (n)p¹–⁶
d-block: (n–1)d¹–¹⁰ (n)s¹–²
f-block: (n–2)f¹–¹⁴ (n–1)d⁰–¹ (n)s²
Block name depends on where the last electron enters.
🔵 Question 3.30
Assign positions for elements having:
(i) ns²np¹ (ii) (n–1)d¹ns² (n = 4) (iii) (n–2)f⁷(n–1)d¹ns² (n = 6)
🟢 Answer:
(i) ns²np¹ → Group 13, p-block (B family)
(ii) 3d¹4s² → Group 3, Period 4, d-block (Sc)
(iii) 4f⁷5d¹6s² → Group 3, Period 6, f-block (Gd)
🔵 Question 3.31
From the data:
Element ΔH₁ ΔH₂ ΔegH
I 520 7300 –60
II 419 3051 –48
III 1681 3374 –328
IV 1008 1846 –295
V 2372 5251 +48
VI 738 1451 –40
Identify:
(a) least reactive (b) most reactive metal (c) most reactive non-metal
(d) least reactive non-metal (e) metal forming ionic MX (f) metal forming covalent MX
🟢 Answer:
(a) V – high ΔH₁, +ΔegH → inert → least reactive
(b) II – lowest ΔH₁ → most reactive metal
(c) III – very negative ΔegH → most reactive non-metal
(d) V again
(e) II forms stable ionic MX
(f) III forms stable covalent MX
🔵 Question 3.32
Predict formulas of stable binary compounds:
(a) Li & O (b) Mg & N (c) Al & I (d) Si & O (e) P & F (f) Element 71 & F
🟢 Answer:
(a) Li₂O (b) Mg₃N₂ (c) AlI₃ (d) SiO₂ (e) PF₅ (f) LuF₃
🔵 Question 3.33
Period number corresponds to:
(a) atomic number (b) atomic mass (c) principal quantum number (d) azimuthal number
🟢 Answer: (c) ✅
🔵 Question 3.34
Which statement is incorrect?
(a) p-block has 6 columns
(b) d-block has 8 columns
(c) Columns = electrons filling subshell
(d) Block shows value of l
🟢 Answer: (b) ❌ (d-block has 10 columns)
🔵 Question 3.35
Which factor does NOT affect valence shell?
(a) principal quantum number (b) nuclear charge (c) nuclear mass (d) core electrons
🟢 Answer: (c) ✅
🔵 Question 3.36
Size order of F⁻, Ne, Na⁺ depends mainly on:
🟢 Answer: (a) Nuclear charge
All have 10 e⁻; higher Z → smaller radius
➡ Order: Na⁺ < Ne < F⁻ ✅
🔵 Question 3.37
Which statement is incorrect about ionization enthalpy?
(a) Each successive IE is higher
(b) Big rise after valence electrons removed
(c) Large jump marks start of core
(d) Removal from lower n is easier
🟢 Answer: (d) ❌ (Lower-n electrons are harder to remove)
🔵 Question 3.38
Metallic character order for B, Al, Mg, K:
🟢 Answer: K > Mg > Al > B ✅
🔵 Question 3.39
Non-metallic character order for B, C, N, F, Si:
🟢 Answer: F > N > C > B > Si ✅
🔵 Question 3.40
Oxidizing power order for F, Cl, O, N:
🟢 Answer: F > O > Cl > N ✅
————————————————————————————————————————————————————————————————————————————
OTHER IMPORTANT QUESTIONS FOR EXAMS
(CBSE MODEL QUESTIONS PAPER)
ESPECIALLY MADE FROM THIS LESSON ONLY
✳ Section A (Q1–Q16) – MCQs (1 mark each, 16 × 1 = 16 marks)
Options:
Both Assertion (A) and Reason (R) are true, and R is the correct explanation of A
Both A and R are true, but R is not the correct explanation of A
A is true, but R is false
A is false, but R is true
Question 1. The modern periodic law is based on:
Atomic mass
Atomic number
Valency
Isotopes
Answer: 2
Question 2. Who is called the “Father of Modern Periodic Table”?
Dobereiner
Newlands
Mendeleev
Moseley
Answer: 4
Question 3. Which of the following is an alkali metal?
Ca
Na
Al
Mg
Answer: 2
Question 4. Which of the following belongs to Group 17?
O
Cl
Na
Ca
Answer: 2
Question 5. In moving from left to right in a period, atomic radius:
Increases
Decreases
Remains same
First increases then decreases
Answer: 2
Question 6. Ionization enthalpy increases across a period because:
Atomic size increases
Atomic size decreases
Shielding effect increases
Nuclear charge decreases
Answer: 2
Question 7. Which element has the highest electronegativity?
O
N
F
Cl
Answer: 3
Question 8. The first noble gas in periodic table is:
He
Ne
Ar
Kr
Answer: 1
Question 9. Which block contains transition elements?
s-block
p-block
d-block
f-block
Answer: 3
Question 10. Lanthanides belong to:
Period 5
Period 6
Period 7
Period 4
Answer: 2
Question 11. Mendeleev classified elements based on:
Atomic number
Atomic mass
Electronic configuration
Isotopes
Answer: 2
Question 12. Which property shows periodic variation?
Melting point
Atomic radius
Density
Colour
Answer: 2
Question 13. Which of the following is isoelectronic with Ne?
Na⁺
O²⁻
F⁻
All of these
Answer: 4
Question 14. (Assertion–Reason)
Assertion (A): Ionization enthalpy increases across a period.
Reason (R): Effective nuclear charge increases across a period.
Answer: 1
Question 15. (Assertion–Reason)
Assertion (A): Atomic radius increases down a group.
Reason (R): New electron shells are added.
Answer: 1
Question 16. Which element has lowest ionization enthalpy?
Cs
Li
Na
K
Answer: 1
⚡ Section B (Q17–Q21) – Very Short Answer (2 marks each, 5 × 2 = 10 marks)
Q17. State modern periodic law.
🟦 Properties of elements are periodic functions of their atomic numbers.
🟩 Periodicity arises due to repetition of similar electronic configuration.
Q18. Why is fluorine more electronegative than chlorine?
🟦 Atomic size of F is smaller → stronger attraction for electrons.
🟩 Effective nuclear charge is greater in F.
Q19. Define Dobereiner’s triads. Give one example.
🟦 A group of 3 elements with similar properties, arranged in increasing atomic mass.
🟩 Example: Li, Na, K → Na has average atomic mass of Li and K.
Q20. Why does atomic radius increase down a group?
🟦 Number of shells increases.
🟩 Nuclear charge increases but shielding effect dominates.
Q21. Calculate effective nuclear charge (Z_eff) for 2p electron of oxygen atom (Z = 8) using Slater’s rule.
➤ Formula: Z_eff = Z – σ
➤ For O (1s² 2s² 2p⁴), shielding constant σ = 2 (from 1s) + 3.4 (from 2s,2p) = 5.4
➤ Z_eff = 8 – 5.4 = 2.6
✅ Final Answer: Z_eff = 2.6
🧪 Section C (Q22–Q28) – Short Answer (3 marks each, 7 × 3 = 21 marks)
Q22. Write three limitations of Mendeleev’s periodic table.
🟦 Position of isotopes not justified.
🟨 Irregularities in atomic mass order (e.g., Co, Ni).
🟩 Position of hydrogen not fixed.
Q23. Explain the trends of ionization enthalpy in the periodic table.
🟦 Increases across a period due to higher nuclear charge and smaller size.
🟨 Decreases down a group due to larger size and shielding effect.
🟩 Exceptions observed (e.g., O < N).
Q24. Write a note on electron gain enthalpy. Give its periodic trend.
🟦 Energy change when an electron is added to neutral atom in gaseous state.
🟨 Across period: becomes more negative due to increase in nuclear charge.
🟩 Down group: becomes less negative due to size increase.
Q25. State Newlands’ law of octaves. Why was it rejected?
🟦 When arranged by atomic mass, every 8th element has similar properties.
🟨 Failed after calcium; only applicable up to first 20 elements.
🟩 Could not accommodate new elements.
Q26. Why noble gases have very high ionization enthalpy?
🟦 They have stable ns²np⁶ configuration.
🟨 Small atomic radius.
🟩 Extra stability requires more energy to remove electron.
Q27. Differentiate between d-block and f-block elements.
🟦 d-block: transition metals, partially filled d-orbitals, groups 3–12.
🟨 f-block: inner transition metals, partially filled f-orbitals, lanthanides/actinides.
🟩 d-block: period 4–7, f-block: period 6–7.
Q28. Explain diagonal relationship with one example.
🟦 Certain elements of period 2 show similarity with diagonally placed period 3 elements.
🟨 Example: Li (period 2, group 1) similar to Mg (period 3, group 2).
🟩 Reason: similar ionic sizes and charge densities.
🧭 Section D (Q29–Q30) – Case-Based Questions (4 marks each, 2 × 4 = 8 marks)
Q29. Read the following passage and answer the questions:
Henry Moseley showed that atomic number is a more fundamental property of elements than atomic mass. This led to the modern periodic law, and the arrangement of elements in increasing atomic number explained anomalies in Mendeleev’s table.
(a) Who gave modern periodic law? (1 mark)
(b) State the modern periodic law. (1 mark)
(c) Explain how Moseley’s work resolved the position of cobalt and nickel. (2 marks)
🧪 Answer:
(a) Henry Moseley.
(b) Properties of elements are periodic functions of their atomic numbers.
(c) Although Co (atomic mass 58.9) is greater than Ni (58.7), atomic numbers are 27 and 28. Moseley’s arrangement by atomic number correctly placed Co before Ni.
Q30. Read the following passage and answer the questions:
Across a period, atomic radius decreases, ionization enthalpy increases, and electron gain enthalpy becomes more negative. These trends arise from increasing nuclear charge with nearly constant shielding.
(a) Why does atomic radius decrease across a period? (1 mark)
(b) Why does ionization enthalpy increase across a period? (1 mark)
(c) Explain the trend in electron gain enthalpy across a period. (2 marks)
🧪 Answer:
(a) Effective nuclear charge increases, pulling electrons closer.
(b) Higher nuclear charge and smaller size require more energy to remove electron.
(c) Addition of electron is easier due to stronger attraction → electron gain enthalpy becomes more negative.
⚡ Section E (Q31–Q33) – Long Answer (5 marks each, 3 × 5 = 15 marks)
Q31. (a) Discuss the main features of modern periodic table.
OR
(b) Write electronic configurations of the first three transition elements of period 4. Explain why they are placed in d-block.
🧪 Answer (a):
🟦 Elements arranged in order of increasing atomic number.
🟨 7 periods (horizontal rows) and 18 groups (vertical columns).
🟩 Groups divided into s, p, d, f blocks based on orbital filling.
🧪 Periodicity explained by repeating electronic configuration.
🎯 Provides logical positions for isotopes, transition, and inner transition elements.
Answer (b):
Sc (Z = 21): [Ar] 3d¹ 4s²
Ti (Z = 22): [Ar] 3d² 4s²
V (Z = 23): [Ar] 3d³ 4s²
🧪 All have partially filled d-orbitals → placed in d-block.
Q32. (a) Explain factors affecting ionization enthalpy.
OR
(b) Define electronegativity. Compare the electronegativity trends in periods and groups.
🧪 Answer (a):
🔷 Atomic size — larger size → lower IE.
🔶 Nuclear charge — higher charge → higher IE.
🧪 Shielding effect — more shielding → lower IE.
🎯 Subshell type — fully/half-filled orbitals → extra stability, higher IE.
Answer (b):
🟦 Electronegativity = tendency of atom to attract shared electrons.
🟨 Across a period: increases (size decreases, nuclear charge increases).
🟩 Down a group: decreases (size increases, shielding dominates).
🎯 Highest value = Fluorine.
Q33. (a) Discuss anomalous behaviour of first element in each group with examples.
OR
(b) Explain diagonal relationship with reasons and examples.
🧪 Answer (a):
🟦 First element of a group (e.g., Li, Be, B, C) shows different properties due to:
➤ Small size
➤ High ionization enthalpy
➤ High electronegativity
🟨 Examples: Li forms Li₃N unlike other alkali metals; Be differs from other alkaline earth metals (covalent compounds).
Answer (b):
🟦 Diagonal relationship: Similarities between period 2 element and diagonally placed period 3 element.
🟨 Example: Li ~ Mg; Be ~ Al.
🟩 Reason: Comparable sizes and charge densities, similar polarising power.
————————————————————————————————————————————————————————————————————————————
