Class 6 : Science – ( English ) : Lesson 8. A Journey through States of Water
EXPLANATION AND ANALYSIS
🌍 Introduction: Water as a Living Companion of Earth
Water is one of the most familiar substances around us, yet it behaves in fascinating ways. We drink it, bathe in it, cook with it, and see it flowing in rivers and falling as rain. At the same time, water can appear as solid ice on mountain peaks, liquid water in lakes and oceans, and invisible water vapour in the air. This ability of water to exist in different forms makes it unique among everyday substances. In this lesson, we explore how water changes its state, why these changes occur, and how these processes shape weather, climate, and life on Earth.
🔵 States of Water: One Substance, Three Forms
Water exists in three main states under normal conditions on Earth.
💧 Liquid water
Liquid water is the most common form we encounter. It flows, takes the shape of its container, and can be poured easily. Rivers, lakes, ponds, seas, and oceans all contain water in this state. Liquid water is essential for plants, animals, and humans because it dissolves nutrients and supports life processes.
❄️ Solid water (ice)
When water is cooled sufficiently, it freezes and becomes ice. Ice has a fixed shape and volume. Snow, hail, frost, and glaciers are all forms of solid water. Interestingly, ice is lighter than liquid water, which is why it floats. This property protects aquatic life during winters, as lakes freeze only on the surface while water below remains liquid.
🌫️ Gaseous water (water vapour)
When water gains enough heat, it changes into water vapour. Water vapour is invisible and mixes with air. Although we cannot see it directly, it is always present around us. Water vapour plays a major role in cloud formation, rainfall, and weather changes.
🔥 Effect of Heat on Water
Changes in the state of water are mainly caused by heating and cooling.
🔥 Heating water
When water is heated, it gains energy. This energy makes water particles move faster and spread apart. As a result, liquid water changes into water vapour. This process does not require boiling; even water in an open bowl slowly changes into vapour.
❄️ Cooling water
When water is cooled, it loses energy. The movement of water particles slows down, and they come closer together. Liquid water changes into ice when cooled enough. Further cooling makes ice colder, but its state remains solid.
Thus, heat energy controls the movement of particles and decides whether water exists as a solid, liquid, or gas.
🔄 Interconversion of States of Water
The continuous change of water from one state to another is called interconversion of states.
🌡️ Melting
When ice is heated, it changes into water. This process is called melting. Ice melts at a fixed temperature known as the melting point.
🌡️ Freezing
When water is cooled, it changes into ice. This process is called freezing. Freezing and melting occur at the same temperature but in opposite directions.
🌫️ Evaporation
Evaporation is the slow change of liquid water into water vapour at any temperature. Wet clothes drying in the sun is a common example. Evaporation causes cooling because water takes heat from its surroundings.
☁️ Condensation
When water vapour cools, it changes back into liquid water. This process is called condensation. Tiny droplets formed during condensation combine to form clouds, fog, or dew.
🌧️ Precipitation
When condensed water droplets in clouds become heavy, they fall to the Earth as rain, snow, or hail. This process is called precipitation.
☀️ Evaporation: A Silent but Powerful Process
Evaporation happens everywhere, all the time.
🌬️ Factors affecting evaporation
Evaporation increases when:
The temperature is high
The surface area of water is large
The air is dry
The wind speed is high
This is why clothes dry faster on a hot, windy day.
❄️ Cooling effect of evaporation
Evaporation causes cooling because water absorbs heat from its surroundings. Sweating cools our body as sweat evaporates from the skin. Earthen pots keep water cool due to evaporation through tiny pores.
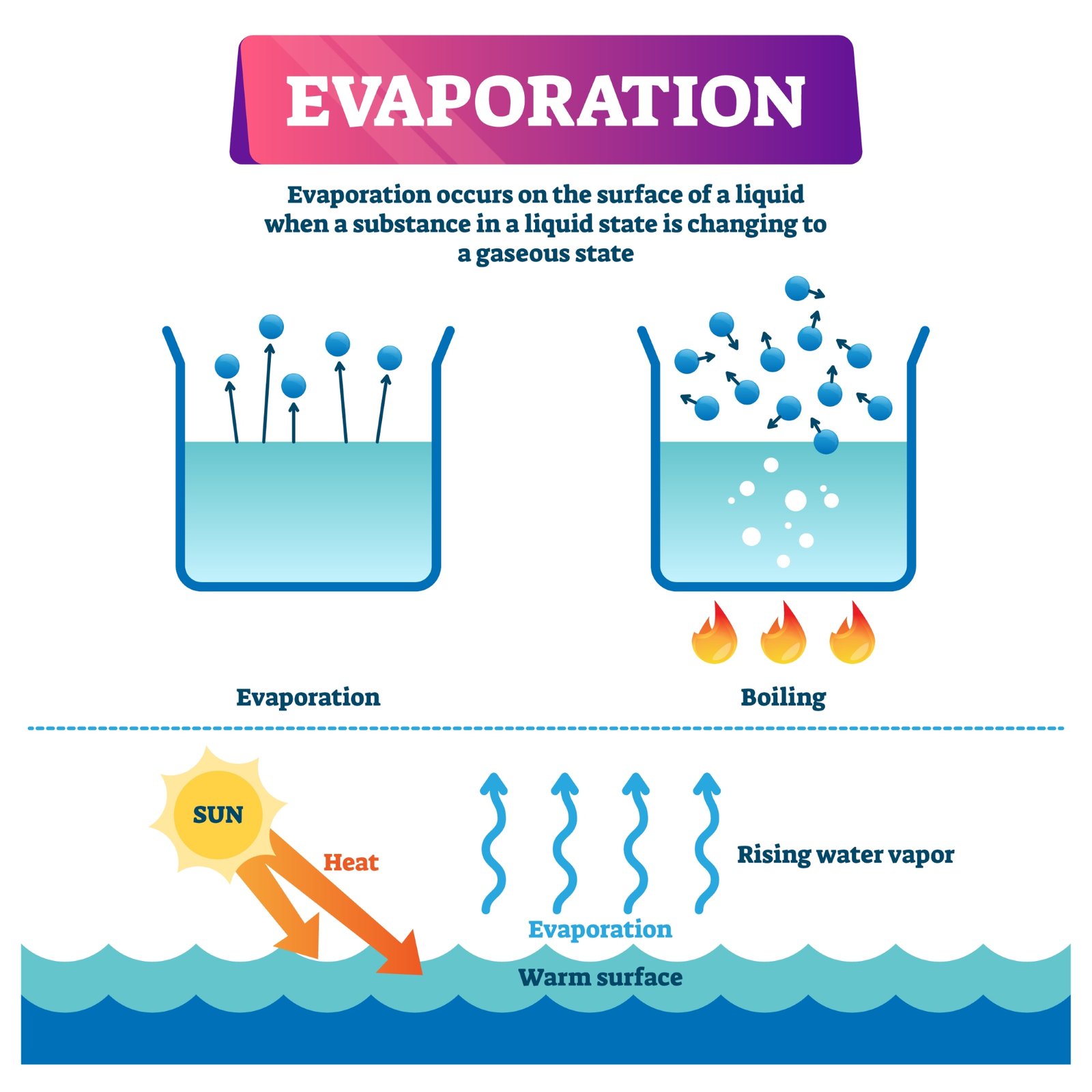
☁️ Condensation and Cloud Formation
Water vapour rises into the atmosphere due to heat from the Sun. As it rises higher, it cools down. Cooling causes water vapour to condense into tiny droplets. Millions of such droplets form clouds. When droplets grow larger and heavier, gravity pulls them down as rain.
This continuous movement of water between Earth and atmosphere forms a natural cycle.
🌎 The Water Cycle: Nature’s Recycling System
The water cycle is the continuous circulation of water on Earth.
🔄 Steps of the water cycle
Evaporation of water from seas, rivers, and lakes
Condensation of water vapour into clouds
Precipitation as rain, snow, or hail
Collection of water in rivers, lakes, and oceans
The water cycle ensures that water is reused again and again, maintaining balance in nature.
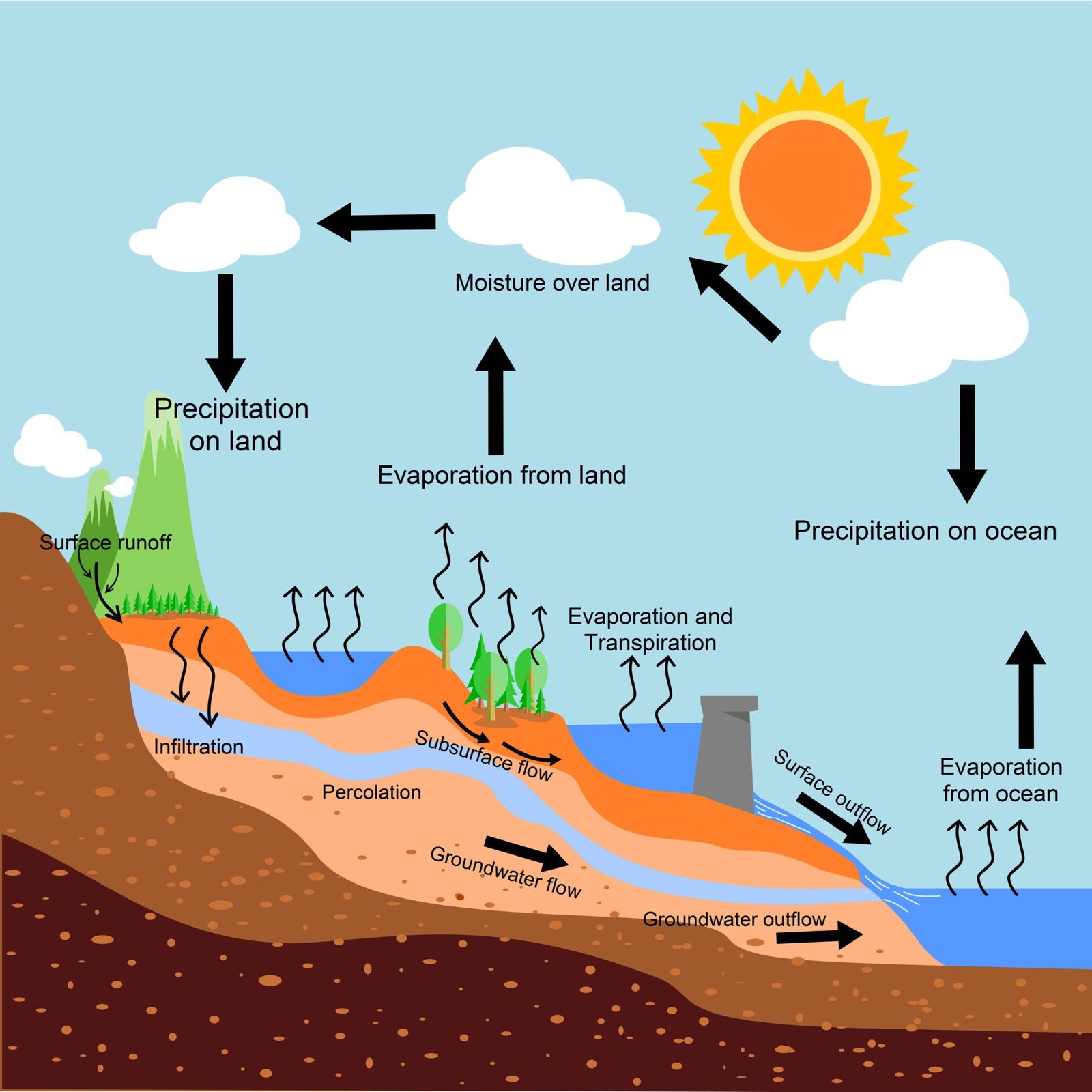
🌱 Importance of States of Water in Daily Life
Different states of water support life and natural processes.
🌾 For plants and animals
Plants absorb water from the soil in liquid form. Animals drink liquid water and depend on rain for survival.
❄️ For climate regulation
Ice caps and glaciers regulate Earth’s temperature. Water vapour controls humidity and rainfall.
🏠 In daily activities
We use ice for cooling, water for cleaning and cooking, and steam for cooking and power generation.
⚠️ Why Water is Special
Water expands on freezing, unlike most substances. This unusual property helps aquatic life survive in cold regions. Water also dissolves many substances, making it a universal solvent. These properties make water essential for life on Earth.
🟣 Summary
Water is a unique substance that exists naturally in three states—solid, liquid, and gas. Ice, water, and water vapour are different forms of the same substance, and the change from one form to another depends on heat energy. Processes like melting, freezing, evaporation, condensation, and precipitation allow water to move continuously between Earth and the atmosphere. Evaporation plays a key role in cooling, while condensation leads to cloud formation and rainfall. The water cycle ensures the constant recycling of water, supporting plants, animals, climate, and human activities. The special properties of water, such as expansion on freezing and ability to dissolve substances, make it vital for sustaining life on our planet.
🟢 Quick Recap
Water exists in solid, liquid, and gaseous states
Heating and cooling cause changes in states
Evaporation occurs at all temperatures
Evaporation causes cooling
Condensation forms clouds
Rainfall is called precipitation
The water cycle recycles water continuously
Ice floats on water and protects aquatic life
Water vapour affects weather and climate
——————————————————————————————————————————————————————————————————————————–
TEXTBOOK QUESTIONS
🔒 ❓ Question 1.
Which of the following best describes condensation?
🟢 1️⃣ The conversion of water into its vapour state
🔵 2️⃣ The process of water changing from a liquid into gaseous state
🟡 3️⃣ The formation of clouds from tiny water droplets
🟣 4️⃣ The conversion of water vapour into its liquid state
📌 Answer: 🟣 4️⃣ The conversion of water vapour into its liquid state
Condensation is the process in which water vapour cools down and changes back into liquid water.
🔒 ❓ Question 2.
Identify in which of the given processes evaporation is very important—
(i) Colouring with
🟢 1️⃣ crayons
🔵 2️⃣ water colours
🟡 3️⃣ acrylic colours
🟣 4️⃣ pencil colours
📌 Answer: 🔵 2️⃣ water colours
Water colours dry when water present in them evaporates, making evaporation essential.
(ii) Writing on paper with
🟢 1️⃣ pencil
🔵 2️⃣ ink pen
🟡 3️⃣ ball point pen
📌 Answer: 🔵 2️⃣ ink pen
Ink dries on paper due to evaporation of the solvent present in the ink.
🔒 ❓ Question 3.
We see green coloured plastic grass at many places these days. Space around natural grass feels cooler than space around plastic grass. Why?
📌 Answer:
Natural grass contains water and shows evaporation. During evaporation, heat is absorbed from the surroundings, producing a cooling effect. Plastic grass does not contain water, so evaporation does not occur and cooling is absent.
🔒 ❓ Question 4.
Give examples of liquids other than water that evaporate.
📌 Answer:
Liquids such as alcohol, petrol, kerosene, acetone and perfume evaporate easily. These liquids change into vapour at room temperature.
🔒 ❓ Question 5.
Fans move air around, creating a cooling sensation. Why are fans used to dry wet clothes even though evaporation requires heat?
📌 Answer:
Fans increase the movement of air around wet clothes. This removes water vapour from the surface quickly and increases the rate of evaporation. The heat required for evaporation is taken from the surroundings, producing a cooling effect and helping clothes dry faster.
🔒 ❓ Question 6.
Sludge removed from drains is left in heaps for 3–4 days before being used as manure. How does this help?
📌 Answer:
When sludge is left in open heaps, water present in it evaporates. Drying reduces bad smell, weight and harmful microbes, making it safer to handle and suitable for use as manure.
🔒 ❓ Question 7.
Observe activities in your house for a day. Identify activities involving evaporation. How does understanding evaporation help us?
📌 Answer:
Activities like drying clothes, cooling of water in earthen pots, sweating and mopping floors involve evaporation. Understanding evaporation helps us dry clothes faster, stay cool in summer and conserve water efficiently.
🔒 ❓ Question 8.
How is water present in the solid state in nature?
📌 Answer:
Water exists in solid state as ice, snow, glaciers and hailstones. These forms are commonly found in cold regions and high mountains.
🔒 ❓ Question 9.
“Water is our responsibility before it is our right.” Share your thoughts.
📌 Answer:
Water is limited and essential for life. We must use it carefully, avoid wastage and protect water sources. Responsible use ensures availability of water for future generations.
🔒 ❓ Question 10.
The seat of a two-wheeler parked on a sunny day becomes very hot. How can you cool it down?
📌 Answer:
Sprinkling water on the seat cools it down. As water evaporates, it absorbs heat from the seat, lowering its temperature.
——————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS
SECTION 1 — MCQs (5 Questions)
🔒 ❓ Q1. Which change of state occurs when ice changes into water?
🟢 1️⃣ Freezing
🔵 2️⃣ Melting
🟡 3️⃣ Condensation
🟣 4️⃣ Evaporation
✔️ Answer: 🔵 2️⃣ Melting
🔒 ❓ Q2. Which state of water has a fixed shape and volume?
🟢 1️⃣ Solid
🔵 2️⃣ Liquid
🟡 3️⃣ Gas
🟣 4️⃣ Vapour
✔️ Answer: 🟢 1️⃣ Solid
🔒 ❓ Q3. What is the process of change of water vapour into liquid water called?
🟢 1️⃣ Evaporation
🔵 2️⃣ Melting
🟡 3️⃣ Condensation
🟣 4️⃣ Freezing
✔️ Answer: 🟡 3️⃣ Condensation
🔒 ❓ Q4. Which factor increases the rate of evaporation?
🟢 1️⃣ Low temperature
🔵 2️⃣ High humidity
🟡 3️⃣ Increased surface area
🟣 4️⃣ Closed container
✔️ Answer: 🟡 3️⃣ Increased surface area
🔒 ❓ Q5. In which state do water particles move most freely?
🟢 1️⃣ Ice
🔵 2️⃣ Water
🟡 3️⃣ Water vapour
🟣 4️⃣ Snow
✔️ Answer: 🟡 3️⃣ Water vapour
SECTION 2 — Very Short Answer (5 Questions)
(One or two words only)
🔒 ❓ Q6. Name the solid state of water.
📌 ✅ Answer: Ice
🔒 ❓ Q7. What is water vapour?
📌 ✅ Answer: Gas
🔒 ❓ Q8. Name the process of liquid changing into gas.
📌 ✅ Answer: Evaporation
🔒 ❓ Q9. Which change of state releases heat?
📌 ✅ Answer: Condensation
🔒 ❓ Q10. Name one factor that affects evaporation.
📌 ✅ Answer: Temperature
SECTION 3 — Short Answer (3 Questions)
(About 40–50 words)
🔒 ❓ Q11. Why does evaporation cause cooling?
📌 ✅ Answer:
🔹 During evaporation, faster moving particles escape from the liquid surface.
🔸 This removes heat energy from the remaining liquid.
🔹 As a result, the temperature of the liquid falls and cooling is felt.
🔒 ❓ Q12. How is boiling different from evaporation?
📌 ✅ Answer:
🔹 Evaporation occurs at all temperatures and only from the surface.
🔸 Boiling occurs at a fixed temperature and throughout the liquid.
🔹 Evaporation is slow, while boiling is a rapid process.
🔒 ❓ Q13. Why do wet clothes dry faster on a windy day?
📌 ✅ Answer:
🔹 Wind removes water vapour from the surface of clothes.
🔸 This increases the rate of evaporation.
🔹 Hence, clothes dry faster when air is moving.
SECTION 4 — Long Answer (1 Question)
(About 70–80 words)
🔒 ❓ Q14. Explain the three states of water and their characteristics.
📌 ✅ Answer:
🔹 Water exists in three states: solid, liquid and gas.
🔸 In solid state as ice, water has a fixed shape and volume.
🔹 In liquid state, water has fixed volume but no fixed shape.
🔸 In gaseous state as water vapour, it has neither fixed shape nor volume.
——————————————————————————————————————————————————————————————————————————–
ADVANCE KNOWLEDGE
🌍 Water: The Most Extraordinary Ordinary Substance
Water looks simple—transparent, tasteless, flowing—but in reality, it is one of the strangest substances in the universe. Scientists believe that without water behaving in its unique way, life on Earth would not exist at all.
🧠 For curious minds:
Water is one of the very few substances that naturally exists on Earth in all three states—solid, liquid, and gas—within the temperature range suitable for life.
🧪 States of Water: Same Substance, Different Behaviours
Water does not change its identity when it changes state.
It remains H₂O, but the arrangement and motion of molecules change.
❄️ Solid State — Ice
Molecules vibrate slowly
Fixed positions
Strong attraction between particles
⭐ Amazing fact:
Ice is less dense than liquid water, which is why it floats.
If ice sank, oceans would freeze from the bottom, killing aquatic life.
💧 Liquid State — Water
Molecules move freely but remain close
No fixed shape
Can flow and take container’s shape
🧠 This state supports:
Life processes
Transport of nutrients
Chemical reactions in cells
☁️ Gaseous State — Water Vapour
Molecules move very fast
Very weak attraction
Invisible to our eyes
⭐ Water vapour controls:
Clouds
Rain
Climate balance
⚠️ Common Misconception vs Reality
⚠️ Misconception: Steam is just hot air
✅ Reality: Steam is gaseous water, carrying enormous energy
⚠️ Misconception: Ice melts only at 0°C
✅ Reality: Pressure and impurities can change melting point
🧠 Molecular Secret Behind State Change
The change of state is not magic—it is energy exchange.
🧪 Heating water:
Energy enters
Molecules move faster
Attraction weakens
❄️ Cooling water:
Energy leaves
Molecules slow down
Attraction strengthens
⭐ The temperature may remain constant during state change, yet energy is absorbed or released.
This hidden energy is called latent heat.
⏳ Historical Curiosity: When Scientists Were Confused
⏳ In ancient times:
People believed steam was “smoke of water”
Ice was thought to be a different substance
🧠 It was only after molecular theory developed that scientists understood:
State change is physical, not chemical
No new substance is formed
🌍 Water Cycle: Earth’s Greatest Recycling System
Water never gets used up—it only changes form and location.
🌍 Key processes:
🌞 Evaporation
☁️ Condensation
🌧️ Precipitation
🌊 Collection
⭐ The water you drink today may have:
Fallen as rain thousands of years ago
Been part of a glacier
Flowed through ancient rivers
🧠 Evaporation: Cooling Without Cold
Evaporation happens at all temperatures, not only at boiling point.
🧪 Faster evaporation occurs when:
Temperature is high
Surface area is large
Wind speed is high
⭐ Evaporation causes cooling because:
High-energy molecules escape
Remaining molecules slow down
🧠 This is why:
Sweating cools the body
Wet clothes feel cold
🚀 Boiling vs Evaporation — Not the Same
⚠️ Misconception: Boiling and evaporation are identical
✅ Reality: They are very different
🔥 Boiling:
Occurs at a fixed temperature
Happens throughout the liquid
🌬️ Evaporation:
Happens at all temperatures
Occurs only at the surface
🌡️ Pressure: The Invisible Controller
Water does not behave the same everywhere.
🧠 At high mountains:
Air pressure is low
Water boils at lower temperature
🧠 In pressure cookers:
Pressure increases
Boiling point rises
⭐ This is why food cooks faster in a pressure cooker.
🚀 Water Beyond Earth
🌌 Scientists search for water to find life.
🚀 Water discovered:
On Mars (ice caps)
On Moon (craters)
On Europa (moon of Jupiter)
🧠 Liquid water means:
Possible chemical reactions
Possible life-friendly conditions
⚠️ Climate Change and States of Water
Small temperature changes cause huge effects.
🌍 Rising temperatures lead to:
Glacier melting
Sea level rise
Extreme weather
🧠 States of water act as climate regulators.
⭐ Amazing Water Facts
⭐ Water expands when it freezes
⭐ Clouds are made of tiny water droplets and ice crystals
⭐ Human body is about 60 percent water
⭐ One molecule of water can participate in thousands of reactions
🌍 Water and Life: A Deep Connection
Every living cell depends on:
Liquid water for reactions
Water balance for survival
🧠 Without water’s unusual properties:
Blood could not flow
Plants could not stand upright
Earth would be lifeless
🧠 Why Curious Minds Must Think Deeper
Water is not just something we drink.
It is a cosmic substance, shaping planets, climate, and life.
🚀 Understanding water deeply prepares young minds to:
Think scientifically
Connect physics, chemistry, and biology
See nature as an intelligent system
🌟 Final Thought
Water teaches us a powerful lesson:
🧠 The smallest particles, when arranged differently, can change the entire world.
——————————————————————————————————————————————————————————————————————————–
