Class 8 : Science – ( English ) : Lesson 9. The Amazing World of Solutes, Solvents, and Solutions
EXPLANATION AND ANALYSIS
🧭 Many substances around us do not exist in a pure form.
Drinking water 💧, lemonade 🍋🥤, salt water 🌊, medicines 💊, and even air 🌬️ are solutions. These mixtures look uniform, but inside them, particles are interacting continuously.
🧠 Understanding solutes, solvents, and solutions helps explain dissolving, mixing, and separation in daily life and science.
🔍 This lesson reveals how substances combine at the particle level.
🧪 A solution is a homogeneous mixture in which one substance is completely dissolved in another.
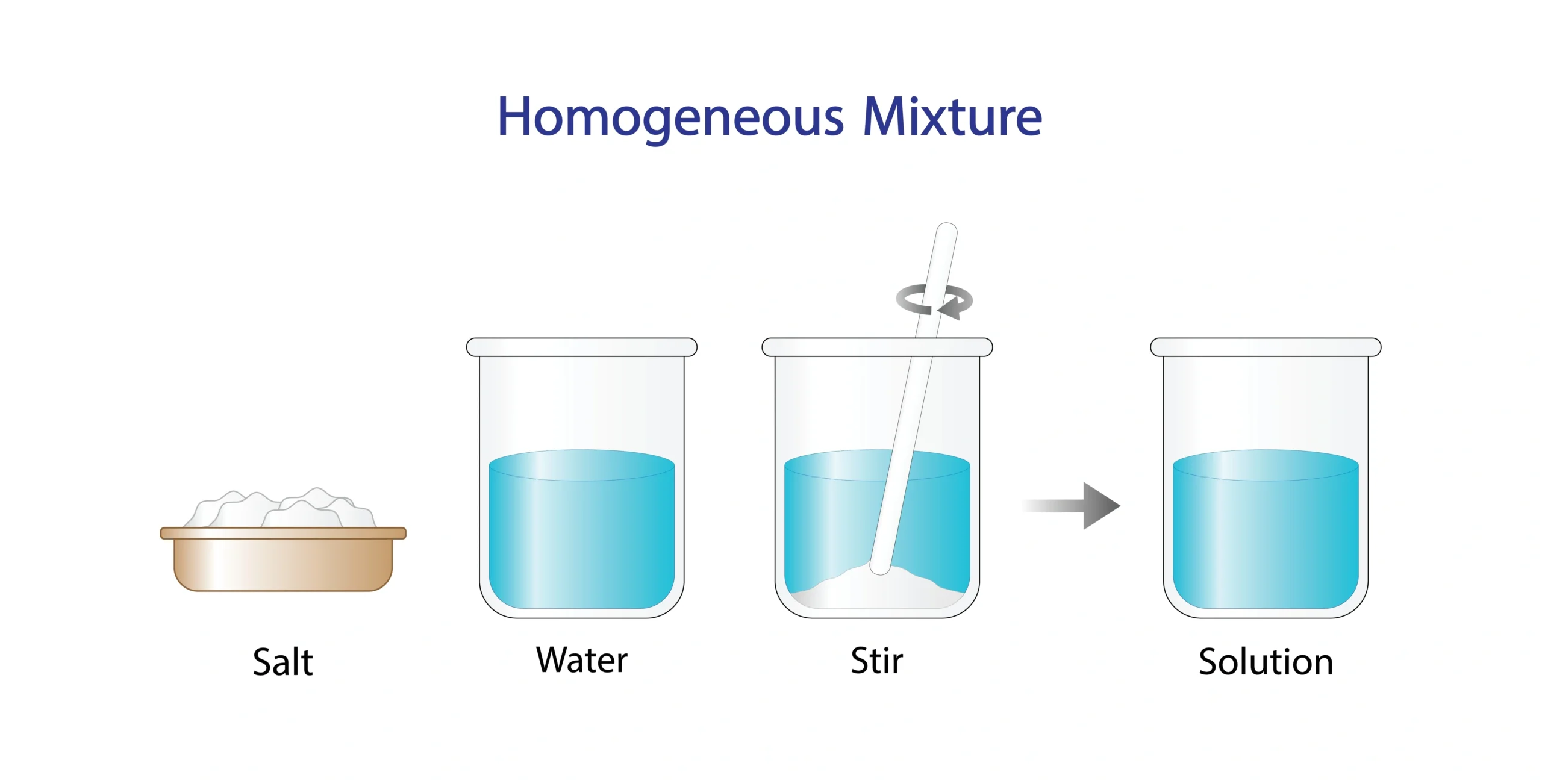
It looks the same throughout and does not show separate components 👀❌.
🧠 Every solution has two main parts.
Solute 🧂
Solvent 💧
🧭 The solute is the substance that gets dissolved.
Sugar in tea 🍬➡️🍵
Salt in water 🧂➡️💧
🧠 Solutes are usually present in smaller amounts.
🧭 The solvent is the substance that dissolves the solute.
Water 💧 is the most common solvent.
🧠 Because it dissolves many substances, water is called a universal solvent.
🧬 When a solute dissolves, its particles spread evenly among solvent particles.
They occupy the spaces between solvent particles 🧩.
🧠 This explains why the solution remains clear and uniform.
🧪 Not all substances dissolve in the same solvent.
Salt dissolves in water ✔️.
Sand does not dissolve ❌.
🧠 Solubility depends on the nature of solute and solvent.
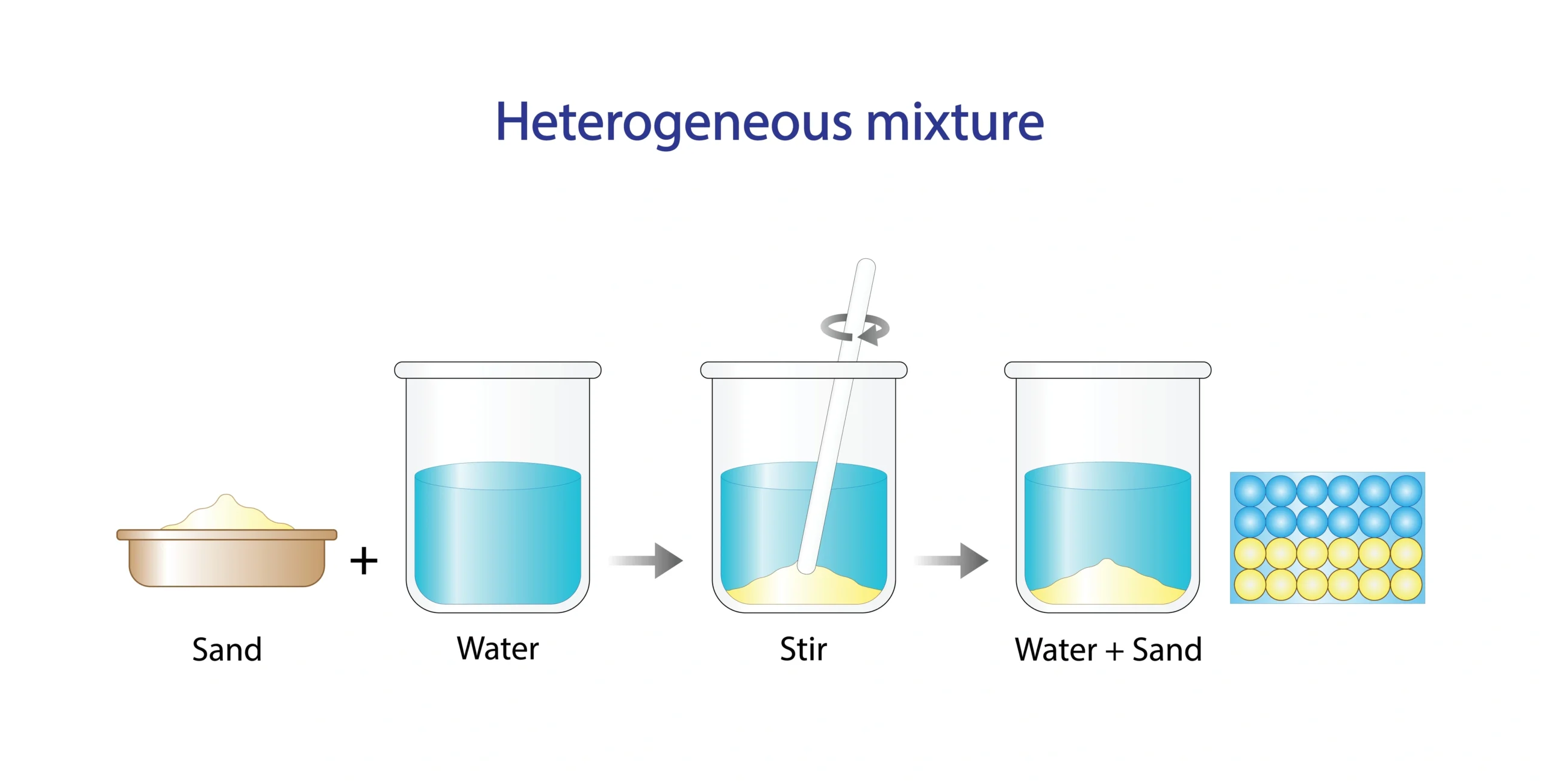
🧭 Solubility is the ability of a solute to dissolve in a solvent.
Some substances are highly soluble 🟢.
Some are partially soluble 🟡.
Some are insoluble 🔴.
🧠 Solubility helps decide how solutions are prepared.
🌡️ Temperature affects solubility.
Heating increases particle movement 🔥➡️⚡.
🧠 Many solids dissolve faster and in larger amounts in warm solvents.
🧭 Stirring also helps dissolving.
Movement spreads solute particles evenly 🔄.
🧠 Stirring increases contact between solute and solvent.
🧪 Size of solute particles matters.
Powdered sugar dissolves faster than sugar crystals 🍬.
🧠 Smaller particles have more surface area.
🧭 Solutions can be classified based on amount of solute.
Dilute solution 💧
Concentrated solution 🧪
🧠 Dilute solutions have less solute.
Concentrated solutions have more solute.
🧪 A saturated solution cannot dissolve more solute at a given temperature.
An unsaturated solution can dissolve more solute.
🧠 This concept is important in chemistry and industries.
🧭 Solutions are not limited to solids in liquids.
Gas in liquid — oxygen in water 🐟💧.
Liquid in liquid — alcohol in water 🍶➡️💧.
Gas in gas — air 🌬️.
🧠 Solutions exist in different physical forms.
🧪 Some mixtures look like solutions but are not true solutions.
Suspensions 🧺
Colloids 🌫️
🧠 These have particles that do not dissolve completely.
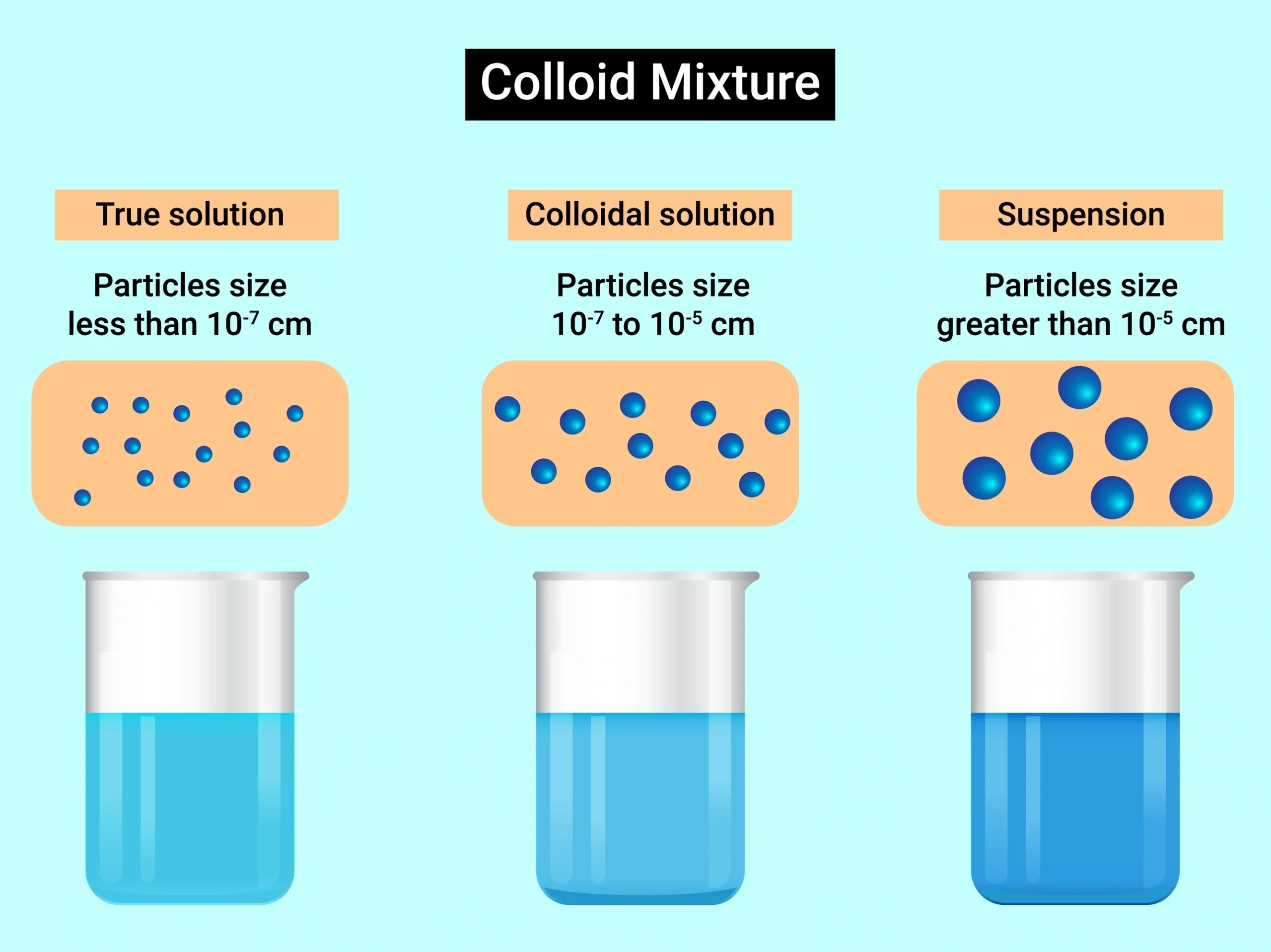
🧭 Suspensions have large particles.
They settle down after some time ⬇️.
🧠 Muddy water 🌊🟤 is a suspension.
🧭 Colloids have particles smaller than suspensions.
They remain evenly spread.
🧠 Milk 🥛 and fog 🌫️ are colloids.
🧪 Solutions play a major role in daily life.
Cooking 🍳
Medicines 💊
Cleaning 🧼
Industry 🏭
🧠 Life processes depend on solutions.
🧠 In the human body, blood 🩸 is a solution-like mixture.
Digestion 🫀
Transport 🚚
Excretion ♻️
🧠 Solutions support biological functions.
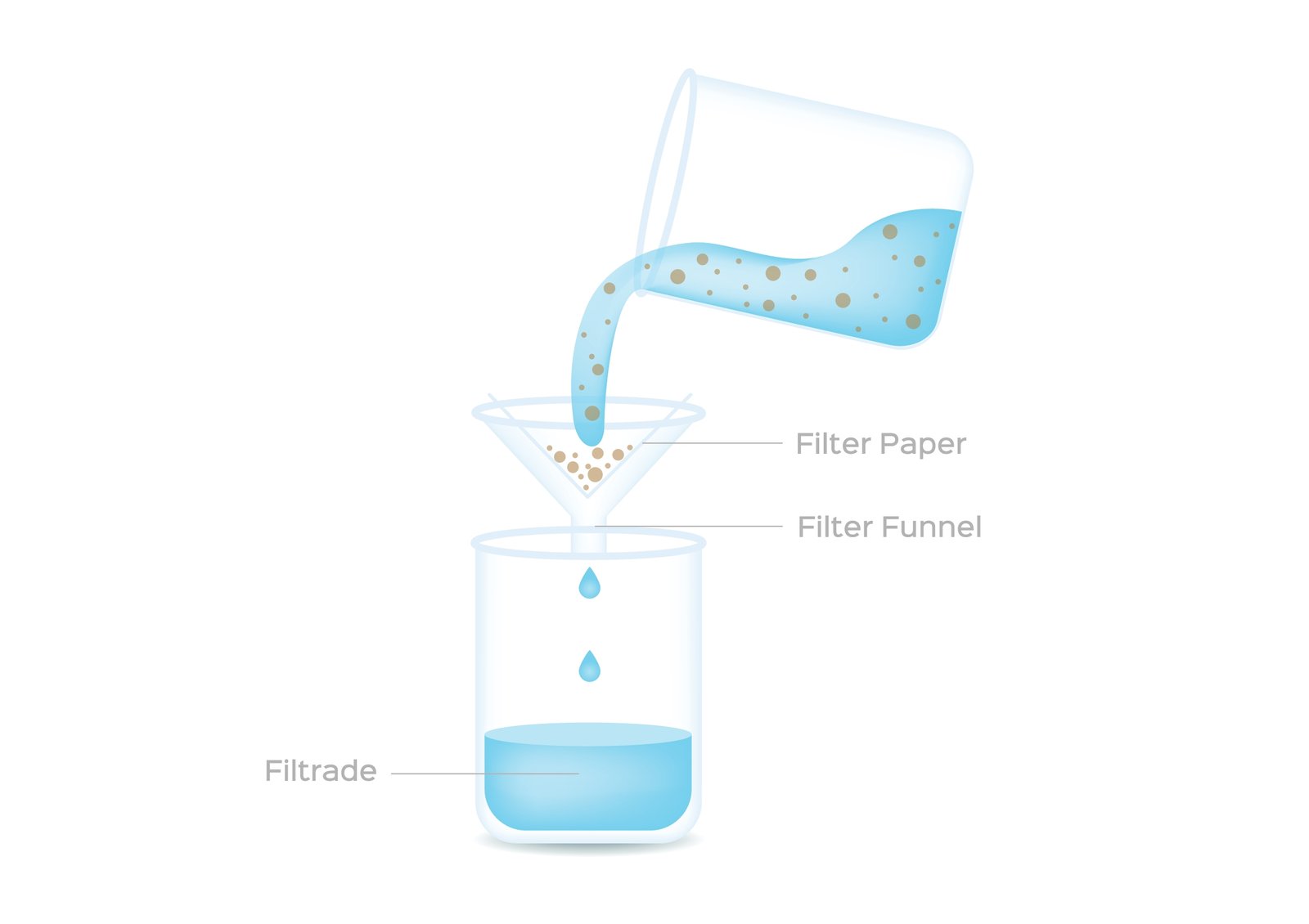
🧭 Separation of solutions is useful.
Evaporation 🌬️
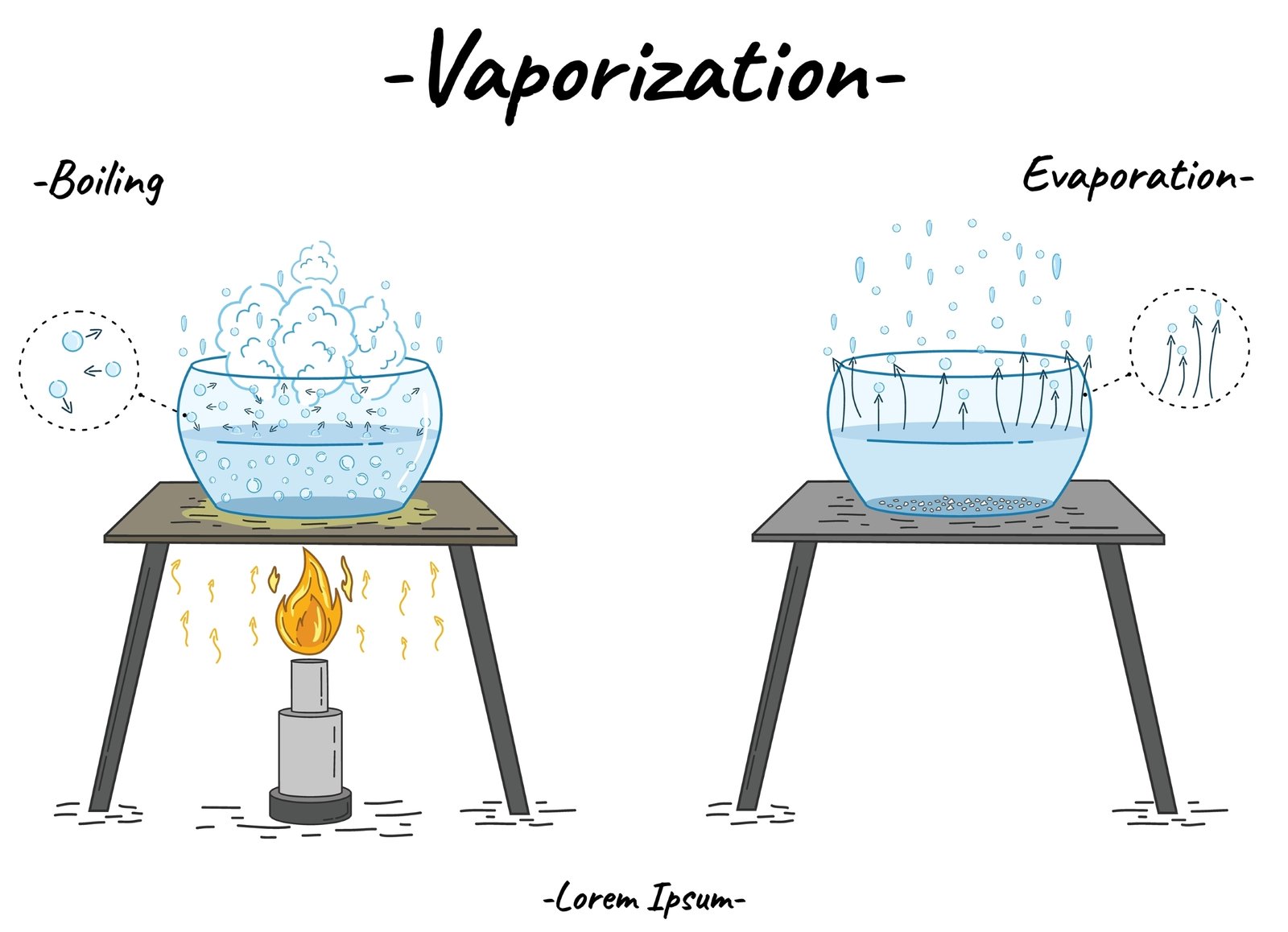
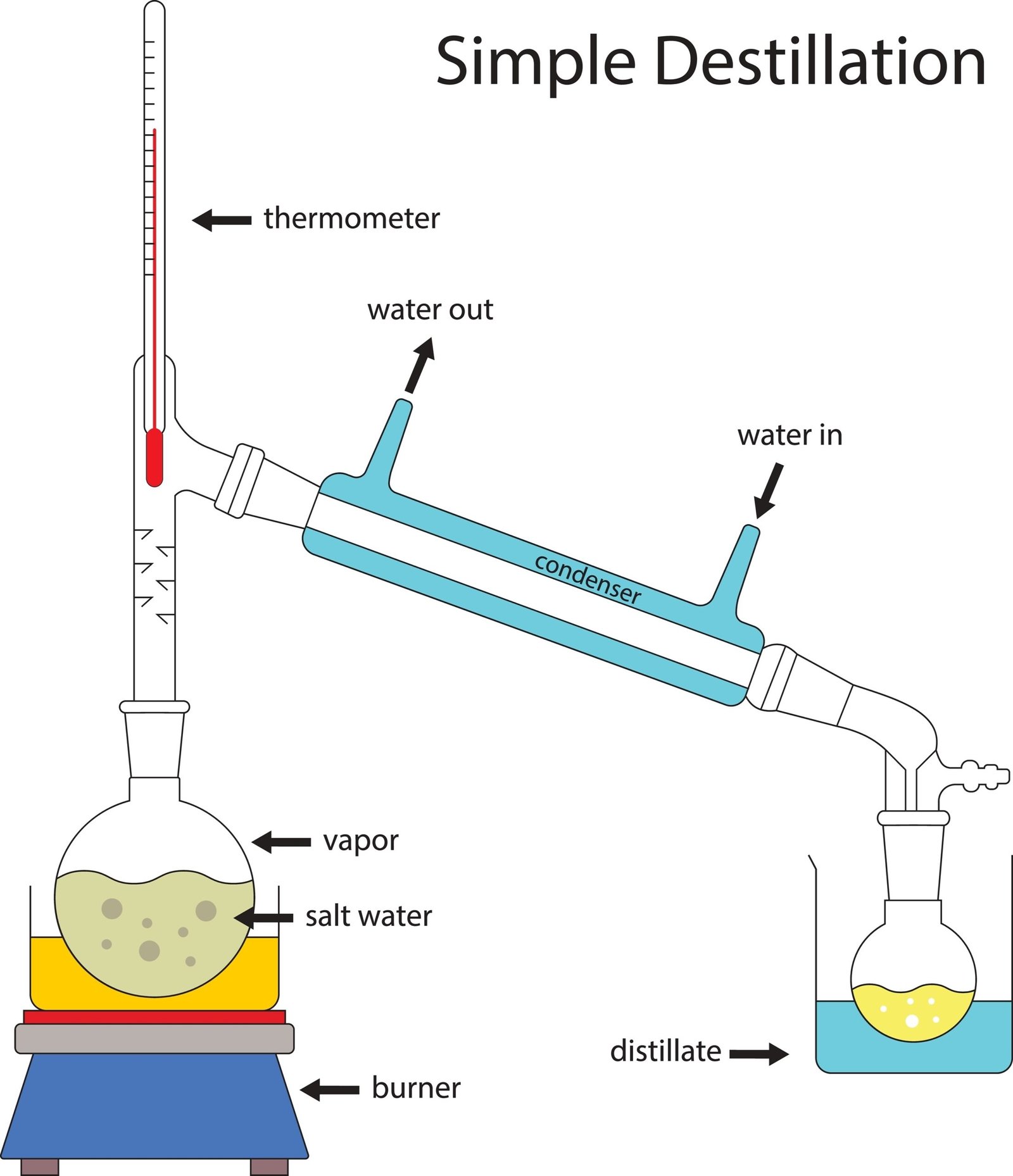
Distillation ⚗️
Crystallization 💎
🧠 These methods help obtain pure substances.
🌍 Understanding solutions helps protect environment.
Water purification 🚰
Waste treatment ♻️
Chemical safety ⚠️
🧠 Knowledge improves responsible use of materials.
🧭 Studying solutions builds scientific thinking.
Particle concept 🧬
Observation 👀
Reasoning 🧠
🧠 It prepares students for advanced chemistry.
📜 Solutions may look simple, but they show deep particle behavior.
Invisible interactions create visible uniformity.
🚀 Learning about solutes and solvents reveals the hidden structure of matter.
📝 Summary of the Lesson
A solution is a homogeneous mixture made of a solute and a solvent. The solute dissolves in the solvent, spreading its particles evenly. Water is the most common solvent. Solubility depends on the nature of substances, temperature, stirring, and particle size. Solutions may be dilute, concentrated, saturated, or unsaturated. Solutions can exist in different forms such as solid in liquid, liquid in liquid, and gas in liquid. Suspensions and colloids differ from true solutions. Solutions are important in daily life, industries, and biological processes.
⚡ Quick Recap
⭐ Solution is a homogeneous mixture
⭐ Solute dissolves in solvent
⭐ Water is a universal solvent
⭐ Solubility depends on conditions
⭐ Solutions can be dilute or concentrated
⭐ Saturated solution has maximum solute
⭐ Solutions are vital in daily life
——————————————————————————————————————————————————————————————————————————–
TEXTBOOK QUESTIONS
🔒 ❓ Question 1
State whether the statements are True [T] or False [F]. Correct the false statements.
📌 Answer:
(i) False
🔵 Oxygen gas is more soluble in cold water than in hot water.
🔴 As temperature increases, the solubility of gases in liquids decreases.
(ii) False
🔵 A mixture of sand and water is not a solution.
🟣 It is a heterogeneous mixture because sand does not dissolve in water.
(iii) False
🔵 The amount of space occupied by an object is called its volume, not mass.
🟢 Mass refers to the quantity of matter present.
(iv) False
🔵 An unsaturated solution contains less solute than a saturated solution at the same temperature.
🟣 It can still dissolve more solute.
(v) True
🔵 Air is a homogeneous mixture of gases and hence is also considered a solution.
🔒 ❓ Question 2
Fill in the blanks.
📌 Answer:
(i) The volume of a solid can be measured by the method of displacement, where the solid is immersed in water and the rise in water level is measured.
(ii) The maximum amount of solute dissolved in solvent at a particular temperature is called solubility at that temperature.
(iii) Generally, the density decreases with increase in temperature.
(iv) The solution in which glucose has completely dissolved in water and no more glucose can dissolve is called a saturated solution of glucose.
🔒 ❓ Question 3
You pour oil into a glass containing water. The oil floats on top. What does this tell you?
📌 Answer:
(ii) Water is denser than oil
🔵 Oil floats because it has lower density than water.
🔴 A substance with lower density floats on a substance with higher density.
🔒 ❓ Question 4
A stone sculpture weighs 225 g and has a volume of 90 cm³. Calculate its density and predict whether it will float or sink in water.
📌 Answer:
🔵 Density = Mass / Volume
🔴 Density = 225 g / 90 cm³
🟣 Density = 2.5 g/cm³
🔵 Since the density of water is 1 g/cm³,
🟢 The sculpture is denser than water and will sink.
🔒 ❓ Question 5
Which one of the following statements is the most appropriate? Why are the others not appropriate?
📌 Answer:
(iii) No more solute can be dissolved into the saturated solution at that temperature.
🔵 This correctly defines a saturated solution.
🔴 (i) is incorrect because a saturated solution cannot dissolve more solute.
🟣 (ii) is incorrect because it describes an unsaturated solution, not saturated.
🟢 (iv) is incorrect because saturation depends on amount of solute, not only temperature.
🔒 ❓ Question 6
You have a bottle with a volume of 2 litres. You pour 500 mL of water into it. How much more water can the bottle hold?
📌 Answer:
🔵 Total volume = 2 litres = 2000 mL
🔴 Water poured = 500 mL
🟣 Remaining capacity = 2000 − 500
🟢 Remaining water = 1500 mL
🔒 ❓ Question 7
An object has a mass of 400 g and a volume of 40 cm³. What is its density?
📌 Answer:
🔵 Density = Mass / Volume
🔴 Density = 400 g / 40 cm³
🟢 Density = 10 g/cm³
🔒 ❓ Question 8
Analyse Fig. 9.25(a) and 9.25(b). Why does the unpeeled orange float, while the peeled one sinks?
📌 Answer:
🔵 The unpeeled orange contains air trapped in its peel.
🔴 This trapped air lowers its average density.
🟣 The peeled orange loses the air layer.
🟢 Its density becomes greater than water, so it sinks.
🔒 ❓ Question 9
Object A has a mass of 200 g and a volume of 40 cm³.
Object B has a mass of 240 g and a volume of 60 cm³.
Which object is denser?
📌 Answer:
🔵 Density of A = 200 / 40 = 5 g/cm³
🔴 Density of B = 240 / 60 = 4 g/cm³
🟢 Object A is denser than Object B
🔒 ❓ Question 10
Reema has a piece of modelling clay weighing 120 g. She first moulds it into a cube of volume 60 cm³, then flattens it into a thin sheet. What happens to its density?
📌 Answer:
🔵 Density depends on mass and volume, not shape.
🔴 Mass remains the same in both cases.
🟣 Volume also remains unchanged.
🟢 Therefore, the density remains the same.
🔒 ❓ Question 11
A block of iron has a mass of 600 g and a density of 7.9 g/cm³. What is its volume?
📌 Answer:
🔵 Volume = Mass / Density
🔴 Volume = 600 / 7.9
🟣 Volume ≈ 75.9 cm³
🔒 ❓ Question 12
In the given experimental setup, when the test tube is kept in hot water, the water level in the glass tube rises. How does it affect density?
📌 Answer:
🔵 Heating causes water to expand.
🔴 Volume increases while mass remains constant.
🟣 Density = Mass / Volume therefore decreases.
🟢 Hence, density decreases on heating.
——————————————————————————————————————————————————————————————————————————–
OTHER IMPORTANT QUESTIONS
SECTION 1 — MCQs (5 Questions)
🔒 ❓ Q1. Which component dissolves in a solution?
🟢 1️⃣ Solvent
🔵 2️⃣ Solute
🟡 3️⃣ Residue
🟣 4️⃣ Suspension
✔️ Answer: 🔵 2️⃣ Solute
🔒 ❓ Q2. Which substance is called the universal solvent?
🟢 1️⃣ Alcohol
🔵 2️⃣ Oil
🟡 3️⃣ Water
🟣 4️⃣ Kerosene
✔️ Answer: 🟡 3️⃣ Water
🔒 ❓ Q3. Which solution allows light to pass without scattering?
🟢 1️⃣ Suspension
🔵 2️⃣ Colloid
🟡 3️⃣ True solution
🟣 4️⃣ Emulsion
✔️ Answer: 🟡 3️⃣ True solution
🔒 ❓ Q4. What happens when more solute is added beyond solubility limit?
🟢 1️⃣ Dissolves completely
🔵 2️⃣ Settles down
🟡 3️⃣ Evaporates
🟣 4️⃣ Reacts chemically
✔️ Answer: 🔵 2️⃣ Settles down
🔒 ❓ Q5. Which factor increases solubility of a solid in liquid?
🟢 1️⃣ Cooling
🔵 2️⃣ Heating
🟡 3️⃣ Freezing
🟣 4️⃣ Standing
✔️ Answer: 🔵 2️⃣ Heating
SECTION 2 — Very Short Answer (5 Questions)
🔒 ❓ Q6. What is a substance that dissolves called?
📌 ✅ Answer: Solute
🔒 ❓ Q7. What do we call the substance that dissolves solute?
📌 ✅ Answer: Solvent
🔒 ❓ Q8. Name a saturated solution condition.
📌 ✅ Answer: No more solute dissolves
🔒 ❓ Q9. Which solution scatters light?
📌 ✅ Answer: Colloid
🔒 ❓ Q10. Name the solution of salt in water.
📌 ✅ Answer: True solution
SECTION 3 — Short Answer (3 Questions)
🔒 ❓ Q11. What is a solution?
📌 ✅ Answer:
🔹 A solution is a homogeneous mixture of solute and solvent.
🔸 The solute dissolves completely in the solvent.
🔹 Solutions have uniform composition throughout.
🔒 ❓ Q12. Why is water called a universal solvent?
📌 ✅ Answer:
🔹 Water dissolves many substances easily.
🔸 Most solids, liquids and gases dissolve in it.
🔹 Hence, water is called a universal solvent.
🔒 ❓ Q13. Differentiate between saturated and unsaturated solutions.
📌 ✅ Answer:
🔹 Saturated solution cannot dissolve more solute at a given temperature.
🔸 Unsaturated solution can dissolve more solute.
🔹 Temperature affects both types.
SECTION 4 — Long Answer (1 Question)
🔒 ❓ Q14. Explain types of solutions based on solubility and size of particles.
📌 ✅ Answer:
🔹 Solutions are classified as true solutions, colloids and suspensions.
🔸 True solutions have very small particles and are transparent.
🔹 Colloids scatter light and have medium-sized particles.
🔸 Suspensions have large particles that settle on standing.
🔹 Solubility and particle size decide the type of solution.
——————————————————————————————————————————————————————————————————————————–
ADVANCE KNOWLEDGE
🌍 When Matter Disappears but Still Exists
When sugar dissolves in water, it seems to vanish. When salt mixes with water, it becomes invisible. But nothing disappears. Matter only spreads, hides, and reorganizes itself at the microscopic level. This mysterious behaviour gives birth to one of the most powerful ideas in science: solutions.
🧠 Big idea:
A solution looks simple, but inside it is a perfectly mixed microscopic world.
🧠 What Is a Solution Really?
A solution is a homogeneous mixture where one substance is evenly spread throughout another.
🧪 Every solution has two main parts:
Solvent → the substance that does the dissolving
Solute → the substance that gets dissolved
⭐ The solvent controls the environment; the solute adapts to it.
⚠️ Misconception vs Reality
⚠️ Misconception: Dissolved substances disappear
✅ Reality: Solute particles spread between solvent particles
⚠️ Misconception: Only solids can dissolve in liquids
✅ Reality: Solids, liquids, and gases can all form solutions
💧 Solvent: The Silent Controller
The solvent decides the nature of a solution.
🧠 Common solvents:
Water (most universal solvent)
Alcohol
Acetone
⭐ Water is called the universal solvent because it dissolves more substances than any other liquid.
🧠 But water cannot dissolve everything—oil resists it.
🧂 Solute: The Guest Particle
The solute becomes part of the solution.
🧠 Solutes can be:
Solids (salt, sugar)
Liquids (alcohol in water)
Gases (carbon dioxide in water)
⭐ Soft drinks are gas-in-liquid solutions.
🔄 Dissolving: A Microscopic Dance
Dissolving is not melting.
🧠 What really happens:
Solvent particles surround solute particles
Attractions pull solute particles apart
Solute spreads evenly
⭐ This process is called solvation.
🧠 Energy and attraction decide whether dissolving will occur.
🌡️ Factors Affecting Solubility
Not everything dissolves equally.
🧠 Solubility depends on:
Temperature
Nature of solute and solvent
Pressure (for gases)
⭐ Hot water dissolves more sugar than cold water.
🧠 Gas solubility decreases when temperature increases—this is why warm soda goes flat.
⚗️ Saturated, Unsaturated, and Supersaturated
Solutions have limits.
🧠 Unsaturated
Can dissolve more solute
🧠 Saturated
Cannot dissolve more at that temperature
🧠 Supersaturated
Holds more solute than normal
Unstable and rare
⭐ Supersaturated solutions are chemical overachievers.
🌍 Solutions in Daily Life
Solutions are everywhere.
🧠 Examples:
Blood
Seawater
Air
Medicines
⭐ Air is a solution of gases.
🧠 Life itself runs in aqueous solutions.
🧠 Concentration: Strength of a Solution
Concentration tells how much solute is present.
🧠 High concentration:
More solute
Stronger effect
🧠 Low concentration:
Less solute
Milder effect
⭐ Medicines depend on precise concentration.
🧪 Colloids and Suspensions: Solution Cousins
Not all mixtures are true solutions.
🧠 Colloids
Tiny particles
Do not settle
Example: milk
🧠 Suspensions
Large particles
Settle on standing
Example: muddy water
⭐ Solutions are the most stable form.
🌬️ Gas Solutions and Pressure
Gases dissolve better under pressure.
🧠 Example:
Carbon dioxide in soda
⭐ When pressure is released, gas escapes as bubbles.
🧠 This principle is used in:
Fire extinguishers
Scuba tanks
🧬 Solutions Inside the Human Body
Human life depends on solutions.
🧠 Examples:
Blood plasma
Cell fluids
Digestive juices
⭐ Every cell lives in a carefully balanced solution.
🧠 Imbalance can cause disease.
🌍 Environmental Science and Solutions
Pollution often spreads as solutions.
⚠️ Examples:
Chemicals in rivers
Gases in air
🧠 Cleaning pollution requires understanding:
Solubility
Dilution
Separation
🚀 Modern Science: Advanced Solution Use
🚀 Scientists use solutions in:
Drug delivery
Nanotechnology
Chemical reactions
⭐ Controlled solutions make modern chemistry precise.
🌌 Solutions Beyond Earth
Planetary atmospheres are gas solutions.
🌌 Examples:
Earth’s air
Jupiter’s gas layers
🧠 Studying them helps understand climate and planetary evolution.
⭐ Amazing Facts
⭐ A single drop of ink spreads through litres of water
⭐ Blood is more complex than seawater
⭐ Oceans contain almost every natural element in solution
⭐ Life cannot exist without solutions
🧠 Why Curious Minds Must Master Solutions
This topic teaches:
Microscopic thinking
Balance and limits
Cause-and-effect reasoning
🧠 It shows how invisible arrangements control visible reality.
🌟 Final Thought
A solution looks calm and uniform,
but inside it is constant motion and interaction.
🧠 To understand solutions is to understand how nature mixes simplicity into complexity.
——————————————————————————————————————————————————————————————————————————–
